Chemical equilibria The basis of classical analytical chemistry

Chemical equilibria — The basis of classical analytical chemistry and the basis of all chemical work Acid-base equilibria, complex equilibria, solubility equilibria, redox equilibria Literature: 1. Hulanicki, A (2012) Reakcje kwaso w i zasad w chemii analitycznej. Wydawnictwo Naukowe PWN, 2012 ((1987) Reactions of Acids and Bases in Analytical Chemistry. PWN-Polish Scientific Publ, Warszawa) 2. Buttler JN (1998) Ionic equilibrium. Solubility and p. H calculations. J. Wiley & Sons, New York 3. Šůcha L, Kotrlý S (1972) Solution equilibria in analytical chemistry. Van Nostrand Reinhold Comp. , London 4. Inczedý J (1976) Analytical Applications of complex equilibria. E. Horwood, Chichester; Akadémiai Kiado, Budapest 5. Kahlert H, Scholz F (2013) Acid-Base Diagrams, Springer, Berlin (Säure-Base-Diagramme, Springer Spektrum, Berlin, 2014)

Acid-base equilibria (Równowagi kwasów i zasad) Etymology: Acid: Alkali: Base: from French acide (16 th c. ) or directly from Latin acidus "sour, sharp" late 14 th c. , "soda ash, " from Medieval Latin alkali, from Arabic al-qaliy "the ashes, burnt ashes" (of saltwort, a plant growing in alkaline soils), from qala "to roast in a pan. " The modern chemistry sense is from 1813. "bottom, foundation, pedestal, " early 14 th c. , from Old French bas "depth" (12 c. ), from Latin basis "foundation, " from Greek βαθμός "step, pedestal, " from ἀναβαίνω "to step". The chemical sense (1810) was introduced in French 1754 by French chemist Guillaume-François Rouelle (1703 -1770).


Johann Rudolph Glauber March 30, 1691(Karlstadt) – March 16, 1670 (Amsterdam) Glauber recognized acids and alkalis as two opposing principles. He was the first to state that a salt is the product of an acid–alkali reaction Rouelle explained the formation of salts as a reaction of acids with a class of compounds which he termed bases. Guillaume-François Rouelle Sept. 15, 1703 (Mathieu) – Aug. 3, 1770 (Passy/Paris)

Robert Boyle Jan. 25; 1626 (Lismore) – Dec. 31, 1691 (London) Boyle had stated that acids have a high dissolution power and can be characterized by four chemical reactions: (a) giving a red color to blue vegetable dyes, (b) precipitating sulfur from ‘liver of sulfur’, (c) neutralizing bases, and (d) reacting with chalk to form a gas.

Antoine-Laurent Lavoisier Aug. 26, 1743 (Paris) – guillotined May 8, 1794 (Paris) Lavoisier created the word oxygène (oxygen) from Greek ὀξύς (oxys) “sharp”, i. e. acidic, and γενής (genês) “born” because he supposed that oxygen is responsible for compounds being acids (“oxygen theory of acids”). German: Säure = acid, Sauerstoff = oxygen Dutch: zuur = acid, zuurstof = oxygen Russian: кислота = acid, кислород = oxygen Czech: kyselina = acid, kyslik = oxygen Hebrew: חומצה (chumtzah) = acid, חמצן (chamtzan) = oxygen Polish: kwas = acid, but tlen* = oxygen Arabic: ﺣﺎﻣﺾ (hamidun) = acid, but =ﺃﻜﺴﺠﻴﻦ oxygen * Michał Sędziwój herbu Ostoja, łac. Sendivogius Polonus (ur. 2 lutego 1566 w Łukowicy, zm. między 20 V a 12 VIII 1636 w Krawarzach) – polski alchemik i lekarz. Otrzymywał tlen z rozkładu saletry potasowej podczas jej prażenia, które to doświadczenie opisał w swoim dziele z 1604 r. p. t. "Dwanaście traktatów o kamieniu filozofów". Stwierdzał, że saletra jest ciałem złożonym, zawierającym "ducha świata" (tak nazwał tlen, uznając go za kamień filozoficzny), umożliwiającym życie ludzi i zwierząt. Wiedział więc, że gaz jest składnikiem powietrza i jest niezbędny do życia.

Jöns Jacob Berzelius August 20, 1779, near Linköping, Sweden – August 7, 1848, Stockholm, Sweden Berzelius supposed that the nature of acids and bases is connected with electricity. He has derived this idea from the observation that electrolysis of aqueous solutions leads to an acidification at the plus pole and alkalization at the minus pole. Justus von Liebig (1838) refuted the idea of Lavoisier and defined an acid as a special hydrogen-containing compound in which the hydrogen can be replaced by a metal. This definition of acids served for over 50 years. Justus von Liebig May 12, 1803 (Darmstadt) – April 18, 1873 (München)

Svante August Arrhenius February 19, 1859, Wijk, Sweden – October 2, 1927, Stockholm, Sweden Arrhenius developed theory of the electrolytic dissociation (1883– 1887). According to him, an acid is a substance which delivers hydrogen ions to the solution. A base is a substance which delivers hydroxide ions to the solution. The symbol p stands for „–log“. It has been introduced in 1909 by the Danish chemist Søren Peter Lauritz Sørensen [1]. Initially Sørensen used to write ; however, this was not generally accepted, and people used to write p. H instead. For a long time it has been believed that Sørensen has chosen ‚p’ as a symbol for „power“ („Potenz“ in German); however, recently, a historical study has revealed that he has used it simply as a symbol for a variable [2], writing for the concentration Cp or 10−p. In modern style this is: . [1] Sørensen SPL (1909) Compt Rend Lab Carlsberg 8: 1; and 8: 396 [2] Nørby JG (2000) Trends Biochem Sc 25: 36 -37 Søren Peter Lauritz Sørensen January 9 th, 1868, Havrebjerg, Denmark – February 12 th, 1939, Charlottenlund, Denmark

Brønsted, Johannes Nicolaus 22. 02. 1879, Varde, Denmark – 17. 12. 1947, Copenhagen, Denmark Lowry, Thomas Martin Oct. 26, 1874, Bradford, West Riding, England – Sept. 2, 1936, Cambridge, England According to Brønsted and Lowry an acid is a proton donator and a base is a proton acceptor

Lewis acid-base theory (1923) To describe reactions where no proton transfer occurs, Lewis has proposed a more general acid–base concept where an acid is a molecule or an ion with an incomplete outer electron sphere, i. e. , an acceptor of electron pairs. A base is a molecule or an ion having a free electron pair, i. e. , an electron pair donor. Gilbert Newton Lewis October 23, 1875 (Weymouth, Massachusetts) – March 23, 1946 (Berkeley, California)

Solvo-acid-base theory (1905/1952) Solvo-acids: providing the cations formed in the auto-solvolysis Solvo-bases: providing the anions formed in the auto-solvolysis Examples: 1. Franklin EC (1905) J Am Chem Soc 27: 820 -851; (1924) J Am Chem Soc 46: 2137 -2151 2. Cady HP, Elsey HM (1928) J Chem Educ 5: 1425 -1428 3. Jander G, Mesech H (1939) Z physik Chem A 183: 255 4. Spandau H, Gutmann V (1952) Angew Chem 64: 93 -120 Edward Curtis Franklin March 1, 1862 (Geary County, Kansas) – February 13, 1937 Hamilton Perkins Cady May 2, 1874 - May 26, 1943 The University of Kansas, University Archives Gerhart Jander Oct. 26, 1892, Alt-Döbern, Germany – Dec. 12, 1961, Berlin, Germany Howard M. Elsey 1891(? ) – ?

Lux-Flood oxide-acid-base theory (1939/1947) Oxo-acids: oxide acceptor Oxo-bases: oxide donator 1. Lux H (1939) Z Elektrochem 45: 303 2. Flood H, Förland T (1947) Acta Chem Scand 1: 592 3. Flood H, Förland T (1947) Acta Chem Scand 1: 781 (1947) 4. Flood H, Förland T, B. Roald (1947) Acta Chem Scand 1: 790 Hermann Lux Sept. 3, 1904 (Karlsruhe) – July 8, 1999 Håkon Flood Sept. 25, 1905 (Trondheim) – Oct. 9, 2001 (Trondheim )

Wymiane i równowagi w wytopach wodorotlenków metalów alkaliycznych Lux H, Niedermaier T (1956) Z allg anorg Chem 285: 246– 261 Lux was a master of experimental techniques. As an example, he has developed the method of hanging melts. Lux has written a book on the art of experimenting in inorganic chemistry: “Anorganisch-chemische Experimentierkunst” 1 st edition 1954, 3 rd edition 1970, J. A. Barth, Leipzig “Technika laboratoryjna w chemii”, PWN, 1960

The unified acid-base theory of Usanovich (1938) Acid: cation donator or anion (or electron) aceptor Base: cation acceptor or anion (or electron) donator 1. Usanovich M (1938) Zh obshchey khim 9: 182 -192 2. Yatsimirskii KB (1973) Theoret Exper Chem 6: 376 -380 Михаил Ильич Усанович Mikhail Il’ich Usanovich June 16, 1894, Zhitomir, Russian Empire – June 15, 1981, Alma-Ata, Soviet Union, now Almaty, Kazakhstan

Acid Base
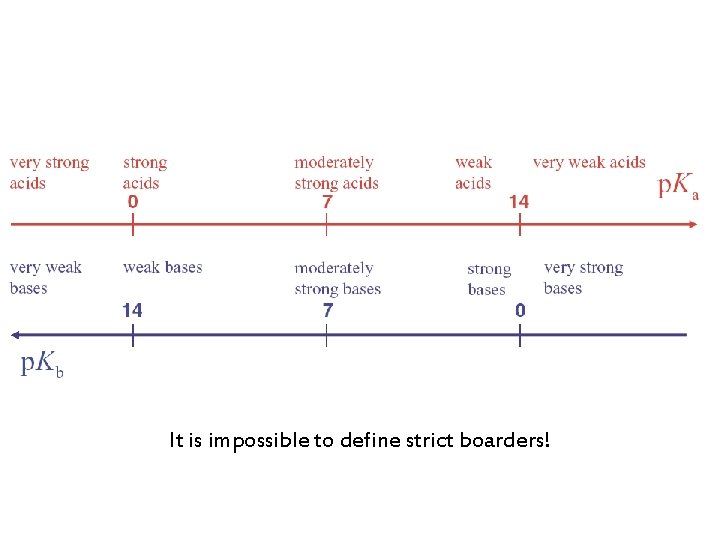
It is impossible to define strict boarders!

The case of hydrofluoric acid The equilibrium constants of (1) and (2) are very large, and the conventionally given p. Ka value refers to the sum of HF and [F−H 3 O+]: Ref: Giguère PA (1979) J Chem Educ 56: 571 -575 SAFETY CONCERNS: Hydrofluoric acid is not so much dangerous because of its acidity, but because fluoride ions very quickly diffuse through human tissue, bind calcium and magnesium ions and lead to fatal enzyme dysfunction. Severe necrosis is also caused! See: (a) Forensic Science International 167 (2007) 49– 52, and (b) Journal of Fluorine Chemistry 79 (1996) 161 -165
- Slides: 17