Chemical Equilbrium Chemistry in Two Directions Frankly Chemistry

Chemical Equilbrium Chemistry in Two Directions Frankly Chemistry (Kc 5 min) https: //www. youtube. com/watch? v=WVREqp 3 g. WLU

Equilibrium is a state in which there are no observable MACROSCOPIOC changes as time goes by. However, the atoms are moving about all the time, back and forth, equally. Chemical equilibrium is achieved when: • the rates of the forward and reverse reactions are equal and • the concentrations of the reactants and products remain constant Physical equilibrium H 2 O (l) H 2 O (g) Chemical equilibrium N 2 O 4 (g) 2 NO 2 (g)
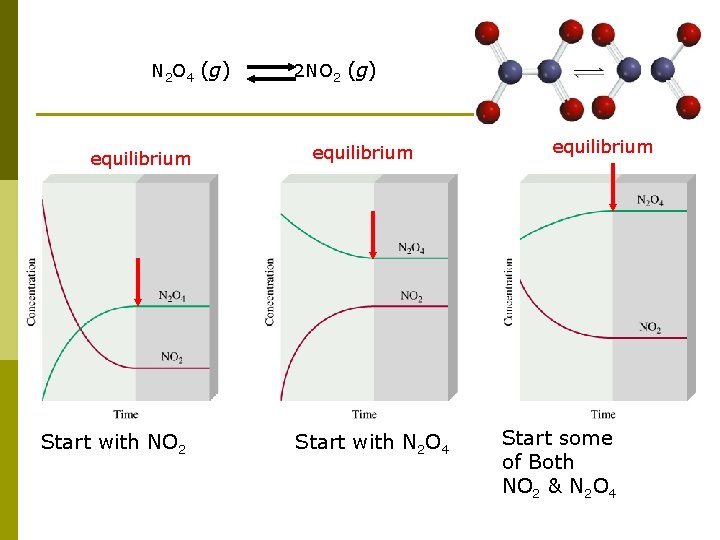
N 2 O 4 (g) 2 NO 2 (g) equilibrium Start with NO 2 equilibrium Start with N 2 O 4 equilibrium Start some of Both NO 2 & N 2 O 4
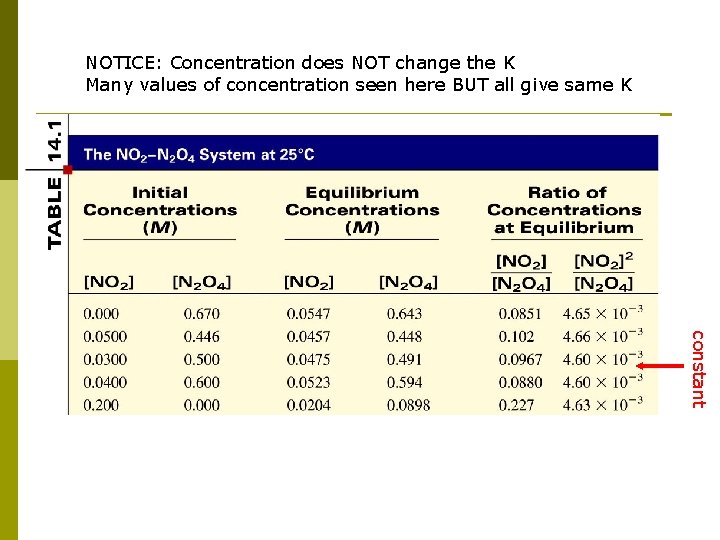
NOTICE: Concentration does NOT change the K Many values of concentration seen here BUT all give same K constant
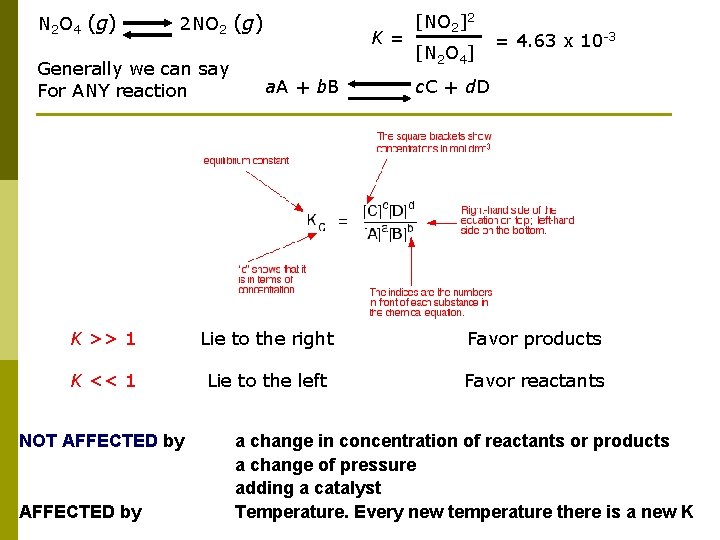
N 2 O 4 (g) 2 NO 2 (g) Generally we can say For ANY reaction K = [NO 2]2 [N 2 O 4] = 4. 63 x 10 -3 a. A + b. B c. C + d. D K >> 1 Lie to the right Favor products K << 1 Lie to the left Favor reactants NOT AFFECTED by a change in concentration of reactants or products a change of pressure adding a catalyst Temperature. Every new temperature there is a new K

Every new temperature there is a new K Equilibrium constants are changed if you change the temperature of the system…. ONLY! The Kp expression (p for pressure) ( c for [conc] ) is: Kp is at 500 K: temperature Kp 500 K 160 Kp is at 700 K: temperature Kp 700 K 54

LET US TRY A QUESTION: Consider the following equilibrium: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) + energy Which of the following will cause the equilibrium constant to increase? A. adding H 2 O(g) [PRODUCTS] B. removing some NO(g) Recall, K = [REACTANTS] C. increasing the temperature D. adding a catalyst E. decreasing the temperature then F. Keq’s are constant they never change! Answer : E Consider the following equilibrium: 2 NO(g) + O 2(g) 2 NO 2(g) + energy When O 2 is increased, equilibrium shifts to the_____ and the K _____ A. left and Keq decreases. B. right and Keq increases. C. left and Keq remains constant. D. right and Keq remains constant. Answer: D P big K big

Homogeneous Equilibrium CH 3 COOH (aq) + H 2 O (l) CH 3 COO- (aq) + H 3 O+ (aq) Make a Kc for this reaction ‘ Kc = [CH 3 COO-][H 3 O+] [CH 3 COOH] [H 2 O] Kc = [H 2 O] = constant at 55. 5 M so it is given a value of 1 [CH 3 COO-][H 3 O+] [CH 3 COOH] ‘ = Kc [H 2 O] You ignore pure liquids and solids – they don’t have a “concentration” and their concentration is considered constant and so it doesn’t contribute

Expressions for Equilibrium Constants Examples: N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g) PCl 5(g) ⇄ PCl 3(g) + Cl 2(g); CH 4(g) + H 2(g) ⇄ CO(g) + 3 H 2(g) Ca. CO 3(s) ⇌Ca. O(s)+CO 2(g) Kc=[CO 2]
![A(aq) + 2 B(aq) ↔ 6 C (aq) Keq = [C]6 [B]2[A] 2 H A(aq) + 2 B(aq) ↔ 6 C (aq) Keq = [C]6 [B]2[A] 2 H](http://slidetodoc.com/presentation_image_h/a4dc55cc9d83e1a90381938753ae16a5/image-10.jpg)
A(aq) + 2 B(aq) ↔ 6 C (aq) Keq = [C]6 [B]2[A] 2 H 2 (g) + O 2 (g) ↔ 2 H 2 O (g) Keq = [H 2 O]2 [H 2]2[O 2] 2 H 2 (g) + O 2 (g) ↔ 2 H 2 O (l) Keq = [H 2 O]2 [H 2]2[O 2]

Write the equilibrium constant expression: 2 HCl(aq) + Ca. CO 3(s) Ca. Cl 2(aq) + H 2 CO 3(aq) A. K = [H 2 CO 3][Ca. Cl 2] [HCl]2[Ca. CO 3] B. K = [H 2 CO 3][Ca. Cl 2] [HCl][Ca. CO 3] C. K = [H 2 CO 3][Ca. Cl 2] [HCl]2 D. K = [HCl]2[Ca. CO 3] [Ca. Cl 2][H 2 CO 3] E. K = [HCl]2 [Ca. Cl 2][H 2 CO 3] ANSWER: C

Different kinds of “K” No matter what the subscript, K IS K! We have Ksp, and Ka, Kb, Kp BUT ALL are K It changes NOTHING. ALL K is done same way, products/reactants (coefficients raised to powers (s and l, not included). VALUE OF Kc AFFECTED by a change of temperature NOT AFFECTED by a change in concentration of reactants or products a change of pressure adding a catalyst

Kc vs Kp p When a reaction occurs in the gas phase, you can use the partial pressure of the gas instead of the concentration. p To separate the 2 different expressions, they are written differently: Kc = equilibrium constant with concentrations of species Kp = equilibrium constant with partial pressures of the species

Equilibrium Problems p There are two main types of problems: n n p You know the concentrations and you are calculating the equilibrium constant You know the equilibrium constant and you are calculating the concentrations Of course, there are nuances to these problems.

Hydrogen and oxygen gases were mixed in a 2 L flask at 450 C. Equilibrium was established & it was determined that there were 3 moles of water, 1. 2 moles of hydrogen and 0. 8 moles of oxygen in the flask. 1 st you need a balanced equation: 2 H 2 (g) + O 2 (g) 2 H 2 O (g) This allows us to immediately write the equilibrium constant expression: Keq = Kc =[H 2 O]2 [H 2]2[O 2] Since we know the equilibrium concentrations: hydrogen: 1. 2 moles/2 L = 0. 6 M oxygen: 0. 8 moles/2 L = 0. 4 M water: 3 moles/2 L = 1. 5 M Kc =(1. 5 M)2 (0. 6 M)2(0. 4 M) Kc =15. 63

A more complicated problem Hydrogen and oxygen gas will react to form steam (gaseous water). 4. 36 g of hydrogen and 28. 6 g of oxygen were mixed in a 2 L flask at 250 C. After equilibrium was established, it was determined that there was 6. 6 g of water What is the equilibrium constant for this reaction at 250 C?

4. 36 g of hydrogen and 28. 6 g of oxygen were mixed in a 2 L flask at 250 C. After equilibrium was established, it was found that there was 6. 6 g of water 4. 36 g hydrogen * 1 mol H 2 = 2. 16 mol H 2 2. 016 g H 2 (INITIAL amount) C=n/v so 2. 16 mol H 2/2 L = 1. 08 M 28. 6 g oxygen * 1 mol O 2 = 0. 894 mol O 2 32. 0 g O 2 (INITIAL amount) 0. 894 mol O 2/2 L = 0. 447 M 6. 6 g H 2 O * 1 mol H 2 O = 0. 366 mol H 2 O 18. 02 g H 2 O (EQUILIBRIUM AMOUNT) 0. 366 mol H 2 O/2 L = 0. 183 M
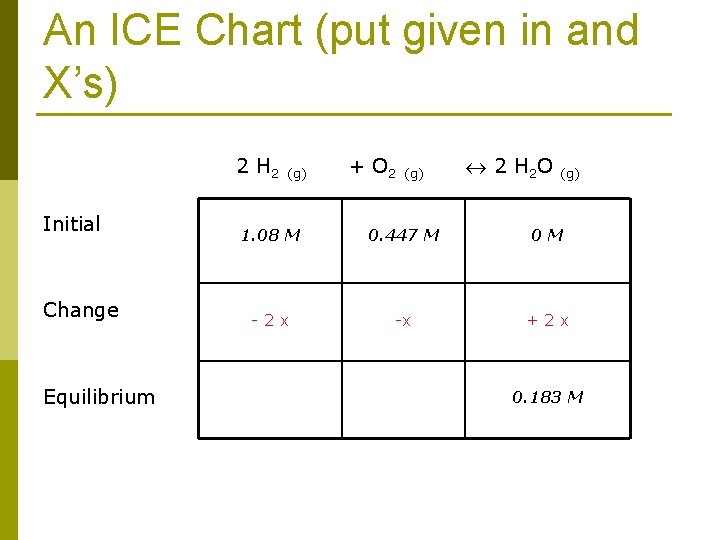
An ICE Chart (put given in and X’s) 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Initial Change Equilibrium 1. 08 M 0. 447 M 0 M - 2 x -x + 2 x 0. 183 M
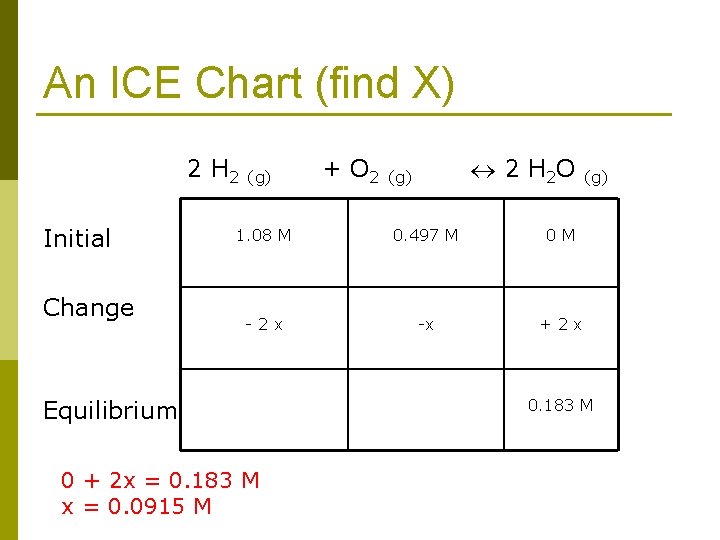
An ICE Chart (find X) 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Initial Change 1. 08 M 0. 497 M 0 M - 2 x -x + 2 x Equilibrium 0 + 2 x = 0. 183 M x = 0. 0915 M 0. 183 M
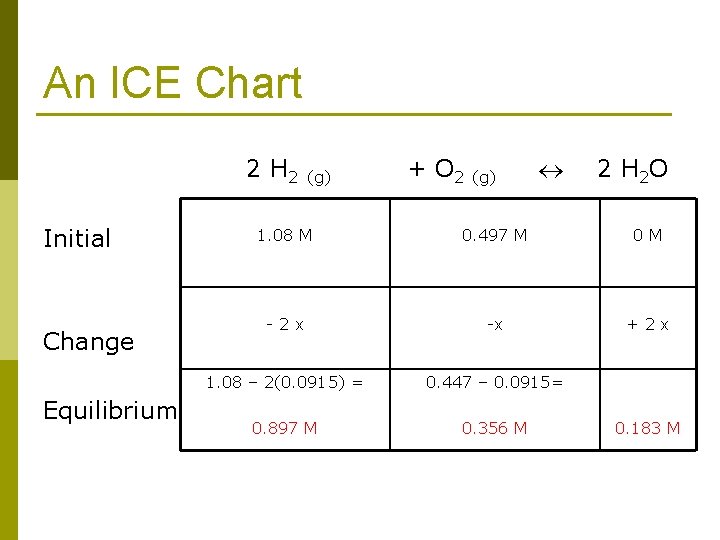
An ICE Chart 2 H 2 (g) + O 2 (g) 2 H 2 O Initial Change Equilibrium 1. 08 M 0. 497 M 0 M - 2 x -x + 2 x 1. 08 – 2(0. 0915) = 0. 447 – 0. 0915= 0. 897 M 0. 356 M 0. 183 M

Now we simply put the concentrations into Kc Keq = Kc = [H 2 O]2 [H 2]2[O 2] Keq = Kc = [0. 183]2 [0. 897]2[0. 356] Kc = 0. 117

p ANOTHER EXAMPLE

A 6. 00 M Fe 3+(aq) and 10. 0 M SCN-(aq) are mixed at a certain temperature and at equilibrium the concentration of Fe. SCN 2+(aq) is 4. 00 M. Fe 3+(aq) + SCN–(aq) Initial Change Equilibrium 6. 00 –x 2. 00 10. 00 –X 6. 00 Fe. SCN 2+(aq) 0. 00 +X 4. 00 x=4. 00 K = 0. 333 23

Calculating Equilibrium Constant p Example: 1. 000 mole of H 2 gas and 1. 000 mole of I 2 vapor are introduced into a 5. 00 -liter sealed flask. The mixture is heated to a certain temperature and the following reaction occurs until equilibrium is established. H 2(g) + I 2(g) ⇄ 2 HI(g) At equilibrium, the mixture is found to contain 1. 580 mole of HI. (a) What are the concentrations of H 2, I 2 and HI at equilibrium? (b) Calculate the equilibrium constant Kc. Initial C = n/v = 1. 000 mol / 5. 00 L = 0. 200 M H 2 and I 2 Equilibrium C = n/v = 1. 580 mol / 5. 00 L = 0. 316 M HI

Calculating Equilibrium Constant for reaction: H 2(g) + I 2(g) ⇄ 2 HI(g) p p —————————————— H 2(g) + I 2(g) ⇄ 2 HI(g) p —————————————— p p Initial [ ], M: 0. 200 Change in [ ], M: -x Equilibrium [ ], M p —————————————— p 0 + 2 x = 0. 316 x = 0. 158 M 0. 200 -x 0. 000 + 2 x 0. 316
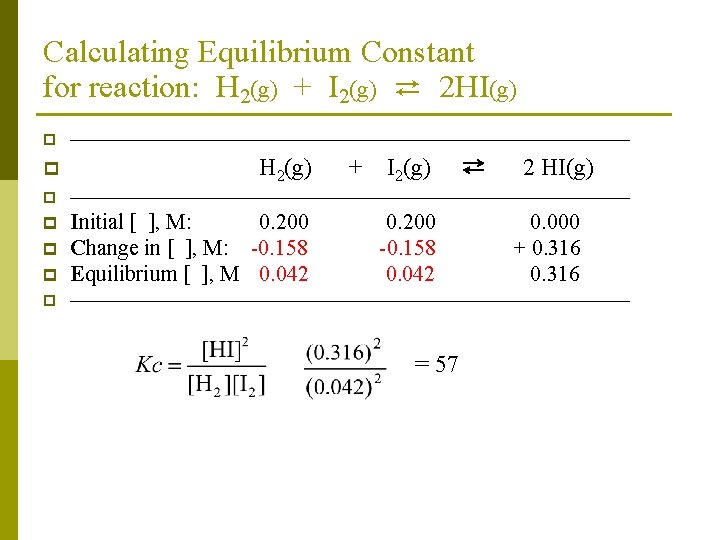
Calculating Equilibrium Constant for reaction: H 2(g) + I 2(g) ⇄ 2 HI(g) p p —————————————— H 2(g) + I 2(g) ⇄ 2 HI(g) p —————————————— p p Initial [ ], M: 0. 200 Change in [ ], M: -0. 158 Equilibrium [ ], M 0. 042 p —————————————— p 0. 200 -0. 158 0. 042 = 57 0. 000 + 0. 316

Another Simple Problem The Kc value for the formation of water from hydrogen and oxygen at 850 C is 4 x 10 -6. If I mix 5. 0 grams of hydrogen and 5. 0 grams of oxygen in a 3 L flask at 850 C, what is the equilibrium concentration of the water?
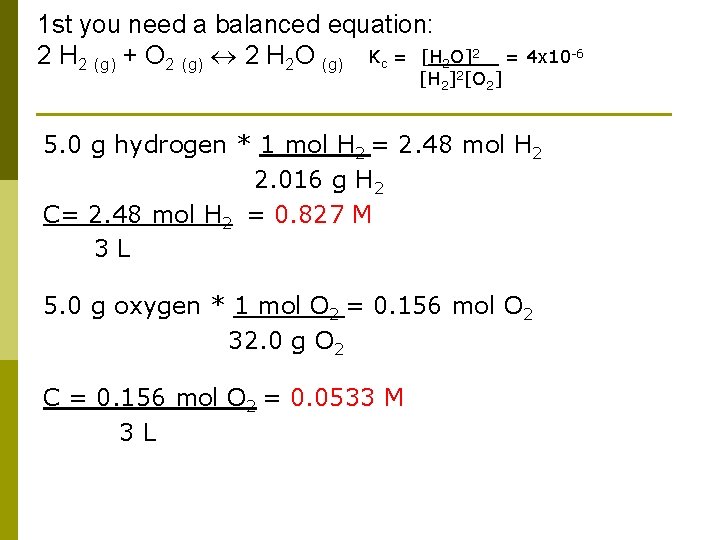
1 st you need a balanced equation: 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Kc = [H 2 O]2 = 4 x 10 -6 [H 2]2[O 2] 5. 0 g hydrogen * 1 mol H 2 = 2. 48 mol H 2 2. 016 g H 2 C= 2. 48 mol H 2 = 0. 827 M 3 L 5. 0 g oxygen * 1 mol O 2 = 0. 156 mol O 2 32. 0 g O 2 C = 0. 156 mol O 2 = 0. 0533 M 3 L
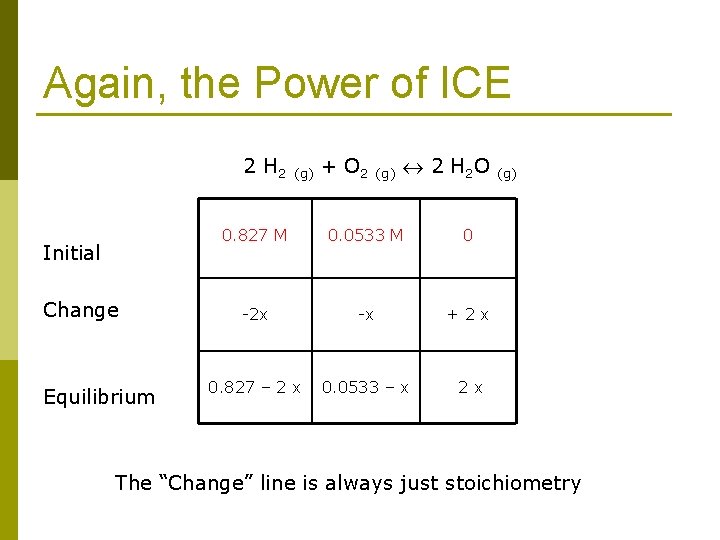
Again, the Power of ICE 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Initial Change Equilibrium 0. 827 M 0. 0533 M 0 -2 x -x + 2 x 0. 827 – 2 x 0. 0533 – x 2 x The “Change” line is always just stoichiometry

We have all of the equilibrium concentrations in terms of x… …we can use Kc to solve for x Kc = [H 2 O]2 = 4 x 10 -6 [H 2]2[O 2] 0. 000004 = (2 x)2 (0. 827 -2 x) 2(0. 0533 -x) If we assume x is small 0. 0533 -x ≈ 0. 0533 0. 827 – 2 x ≈ 0. 827 4 x 10 -6 = (2 x)2 (0. 827 -2 x)2(0. 0533 -x) becomes 4 x 10 -6 = (2 x)2 (0. 827)2(0. 0533) 4 x 10 -6 = 4 x 2 0. 03645 1. 458 x 10 -7 = 4 x 2 3. 645 x 10 -8 = x 2 X = 1. 91 x 10 -4

Was the assumption good? We started by assuming x<<0. 0533 We now “know” that, with this assumption, x is 1. 91 x 10 -4 Is 1. 91 x 10 -4 << 0. 0533? Answer must be 5% of X or 5% of 0. 0533 To make estimation true
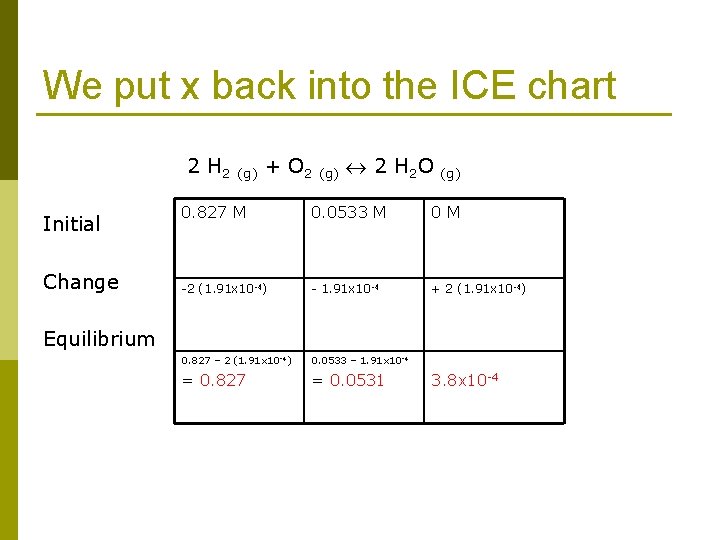
We put x back into the ICE chart 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Initial Change 0. 827 M 0. 0533 M 0 M -2 (1. 91 x 10 -4) - 1. 91 x 10 -4 + 2 (1. 91 x 10 -4) 0. 827 – 2 (1. 91 x 10 -4) 0. 0533 – 1. 91 x 10 -4 = 0. 827 = 0. 0531 3. 8 x 10 -4 Equilibrium

Calculating Equilibrium Constant p Example-2: 0. 500 mole of HI is introduced into a 1. 00 liter sealed flask and heated to a certain temperature. An analysis shows that 0. 105 mole of HI has decomposed as the equilibrium mixture shows as 0. 395 was HI has formed. Calculate the equilibrium concentrations of H 2, I 2 and HI, and the equilibrium constant Kc for the following reaction: H 2(g) + I 2(g) ⇄ 2 HI(g),
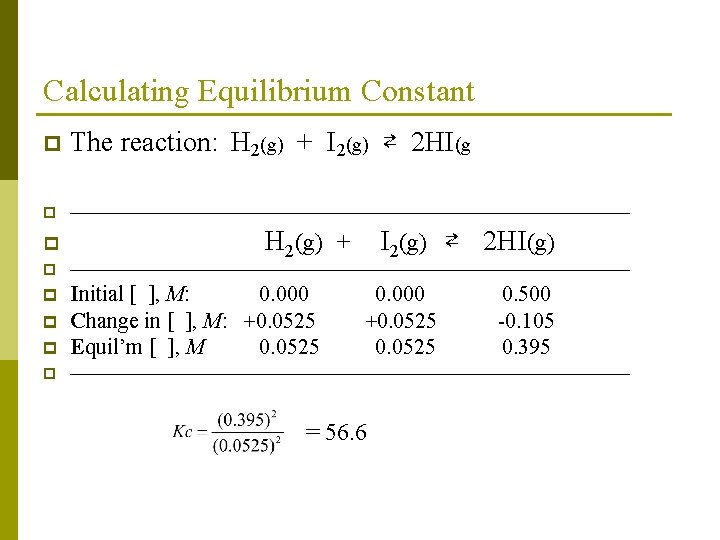
Calculating Equilibrium Constant p The reaction: H 2(g) + I 2(g) ⇄ 2 HI(g p —————————————— p H 2(g) + I 2(g) ⇄ 2 HI(g) p —————————————— p p Initial [ ], M: 0. 000 Change in [ ], M: +0. 0525 Equil’m [ ], M 0. 0525 p —————————————— p 0. 000 +0. 0525 = 56. 6 0. 500 -0. 105 0. 395

Example N 2(g) + 3 H 2(g) 2 NH 3(g)

Example N 2(g) + 3 H 2(g) 2 NH 3(g) Equilibrium pressures at a certain temperature:

Example N 2(g) + 3 H 2(g) 2 NH 3(g)

CALCULATIONS of Kp Example When nitrogen (1 mole) and hydrogen (3 moles) react at constant temperature at a pressure of 8 x 106 Pa, the equilibrium mixture was found to contain 0. 70 moles of ammonia. Calculate Kp. moles (initially) moles (equilibrium) mole @ equil N 2(g) + 3 H 2(g) 1 3 - x(x=0. 35) - 3 x(x=0. 35) 2 NH 3(g) 0 2 x (x=0. 35) b/c 2 x = 0. 70, x=0. 35 0. 65 0. 70 1. 95 at equilibrium we have 0. 7 moles NH 3 and n. Total = (0. 65 +1. 95+ 0. 70) = 3. 30 moles total the total pressure (P) = 8 x 106 Pa. X fraction= Pa/Pt N 2 at equil is = 0. 65/3. 3 = 0. 20 x (8 x 106 Pa) = 1. 60 x 106 Pa and H 2 = 1. 95/3. 3 = 0. 59 x (8 x 106 Pa) = 4. 73 x 106 Pa And NH 3 at 0. 70/3. 3 = 2. 12 x (8 x 106 Pa) = 1. 70 x 106 Pa applying the equilibrium law Kp = (PNH 3)2 with units of Pa-2 (PN 2). (PH 2)3 Substituting in the expression gives Kp = 1. 71 x 10 -14 Pa-2

Relationships between chemical equations and the expressions of equilibrium constants p H 2(g) + I 2(g) ⇄ 2 HI(g); p For the reverse reaction: p 2 HI(g) ⇄ H 2(g) + I 2(g); p And for the reaction: HI(g) ⇄ ½H 2(g) + ½I 2(g) If all doubled…. . p For 2 H 2(g) + 2 I 2(g) ⇄ 4 HI(g); p
- Slides: 39