Chemical Equations Reactions Materials Conserving Matter Chemical Reaction
















- Slides: 17

Chemical Equations & Reactions Materials: Conserving Matter

Chemical Reaction Process by which one or more substances are changed into one or more different substances

Indications of a Chemical Reaction 1. Evolution of heat & light

Indications of a Chemical Reaction 2. Production of a gas

Indications of a Chemical Reaction 3. Formation of a precipitate

Indications of a chemical reaction 4. Change in color. (sumac leaves change color when chlorophyll is destroyed)

Chemical Reactions l There are many ways where matter seems to disappear: l l l The consumption of fuel by your car. The lunch you ate today. The rusting away of your car. Do they really disappear? NO

Chemical Equation Reactants Products Follows the Law of Conservation of Mass

Chemical Reactions l Law of Conservation of Matter: in a chemical reaction, matter in neither created nor destroyed; the molecules are merely rearranged l l l atoms are forever! you should be able to account for every individual atom in a chemical reaction

Writing Chemical Equations Word Equation Represents facts- only qualitative Example: Methane + oxygen carbon dioxide + water

Writing Chemical Equations Formula Equation Example: CH 4(g) + O 2(g) CO 2(g) + H 2 O(g) (not balanced) Symbols you will see in equations

Writing Chemical Equations Balanced Formula Equation Inserting Coefficients-small whole # that appears in front of a formula Multiplies number of atoms of each element indicated in chemical formula Example: CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g)

Atom Inventory l Name the number and type of atoms in Ag. NO 3 (NH 4)2 CO 3 6 H 2 O

Atom Inventory l In order to follow the law of conservation- l there must be the same number and type of atoms on both sides of the equation.

Atom Inventory l l Is this equation balanced? Take an atom inventory Mg + O 2 Mg. O yes no 2 Na. OH Na 2 O + H 2 O yes no Mg + Cl 2 Mg. Cl 2 yes no

Balancing Chemical Equations Balance the formula equation according to the law of conservation of mass l l l Balance the different types of atoms one at a time Balance polyatomic ions that appear on both sides of the equation as single units Balance H atoms and O atoms after atoms of all other elements have been balanced

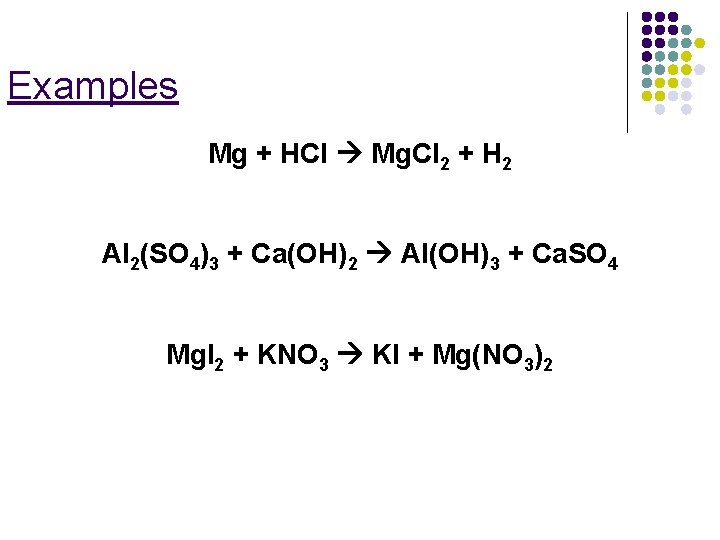
Examples Mg + HCl Mg. Cl 2 + H 2 Al 2(SO 4)3 + Ca(OH)2 Al(OH)3 + Ca. SO 4 Mg. I 2 + KNO 3 KI + Mg(NO 3)2