Chemical Equations Reactions Describing a Chemical Reaction Indications

Chemical Equations & Reactions

Describing a Chemical Reaction Indications of a Chemical Reaction – Evolution of heat, light, and/or sound – Production of a gas – Formation of a precipitate – Color change

Chemical Equations Depict the kind of reactants and products and their relative amounts in a reaction. 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) The numbers in the front are called stoichiometric coefficients The letters (s), (g), and (l) are the physical states of compounds.

Chemical Equations 4 Al(s) + 3 O 2(g) 2 Al 2 O 3(s) This equation means: 4 Al atoms + 3 O 2 molecules yield 2 molecules of Al 2 O 3 or 4 Al moles + 3 O 2 moles yield 2 moles of Al 2 O 3

Chemical Equations Because the same atoms are present in a reaction at the beginning (reactants) and at the end (reactants), the amount of matter in a system does not change. The Law of Conservation of Matter

Chemical Equations Because of the principle of the conservation of matter, matter An equation must be balanced It must have the same number of atoms of the same kind on both sides. Lavoisier, 1788

Signs of Chemical Reactions There are five main signs that indicate a chemical reaction has taken place: input release change in color change in odor production of new gases or vapor input or release of energy difficult to reverse

Characteristics of Chemical Equations • The equation must represent known facts. • The equation must contain the correct formulas for the reactants and products. • The law of conservation of mass must be satisfied. • Reactants→Products

Word Equations • A WORD EQUATION describes chemical change using the names of the reactants and products. • Write the word equation for the reaction of methane gas with oxygen gas to form carbon dioxide and water. methane + oxygen Reactant carbon dioxide + Product water
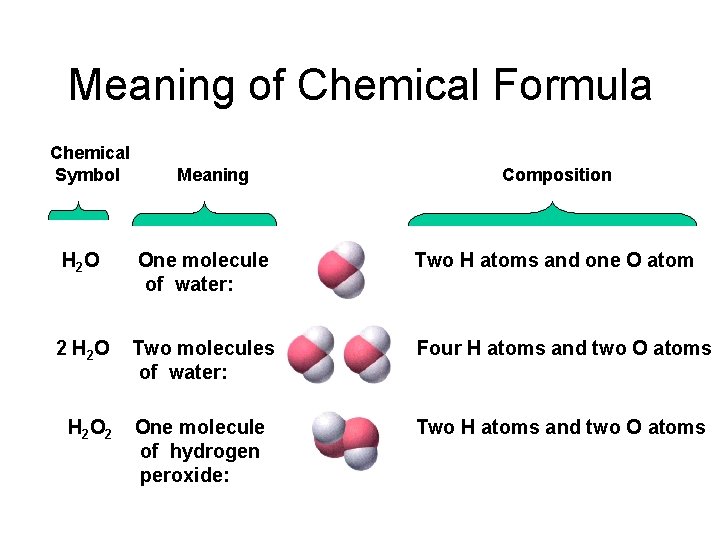
Meaning of Chemical Formula Chemical Symbol Meaning Composition H 2 O One molecule of water: Two H atoms and one O atom 2 H 2 O Two molecules of water: Four H atoms and two O atoms One molecule of hydrogen peroxide: Two H atoms and two O atoms H 2 O 2

Symbols Used in Chemical Equations “Yields”; indicates result of reaction Used to indicate a reversible reaction (s) A reactant or product in the solid state; also used to indicate a precipitate Alternative to (s), but used only to indicate a precipitate (l) A reactant or product in the liquid state (aq) A reactant or product in an aqueous solution (dissolved in water) (g) A reactant or product in the gaseous state

Additional Symbols Used in Chemical Equations Alternative to (g), but used only to indicate a gaseous product D 2 atm pressure Reactants are heated Pressure at which reaction is carried out, in this case 2 atm Pressure at which reaction is carried out exceeds normal atmospheric pressure 0 o. C Temperature at which reaction is carried out, in this case 0 o. C Mn. O 2 Formula of catalyst, in this case manganese (IV) oxide, used to alter the rate of the reaction

Showing Phases in Chemical Equations H 2 O(s) H 2 O(l) H 2 O(g) • Solid Phase – the substance is relatively rigid and has a definite volume and shape. Na. Cl(s) • • Liquid Phase – the substance has a definite volume, but is able to change shape by flowing. H 2 O(l) • • Gaseous Phase – the substance has no definite volume or shape, and it shows little response to gravity. Cl 2(g)

Types of Chemical Reactions Synthesis (Combination) reaction Decomposition reaction ASingle-replacement reaction BDouble-replacement reaction Acid base reaction Combustion reaction (of a hydrocarbon) Ause activity series to predict Bdriving force…water, gas, or precipitate A + B AB AB A + BC AC + B AB + CD AD + CB HX + BOH BX + HOH CH + O 2 CO 2 + H 2 O

Synthesis Reaction Direct combination reaction (Synthesis) 2 Na + Cl 2 Na. Cl Cl Na+ Cl - Cl Cl - Na+ Na General form: A element or compound + B element or compound AB compound

Decomposition Reaction Decomposition reaction 2 H 2 O 2 H 2 + O 2 H O H + H O H General form: AB compound A + B two or more elements or compounds

Single and Double Replacement Reactions Single-replacement reaction Mg + Cu. SO 4 General form: A + BC Mg. SO 4 AC + + Cu B Double-replacement reaction Ca. CO 3 + General form: AB + 2 HCl Ca. Cl 2 + H 2 CO 3 CD AD + CB

Combustion Reactions A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. Reactive elements combine with oxygen P 4(s) + 5 O 2(g) P 4 O 10(s) (This is also a synthesis reaction) The burning of natural gas, wood, gasoline C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(g)
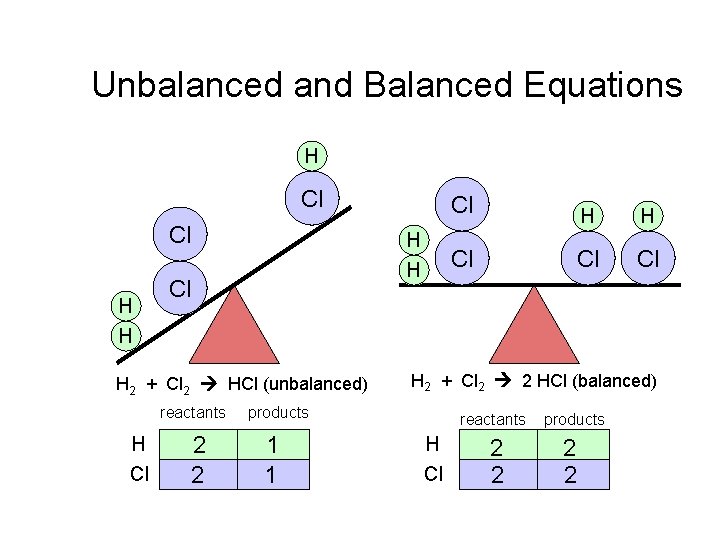
Unbalanced and Balanced Equations H Cl Cl H H Cl H 2 + Cl 2 HCl (unbalanced) reactants H Cl 2 2 H H Cl Cl Cl H 2 + Cl 2 2 HCl (balanced) products 1 1 Cl H Cl reactants products 2 2
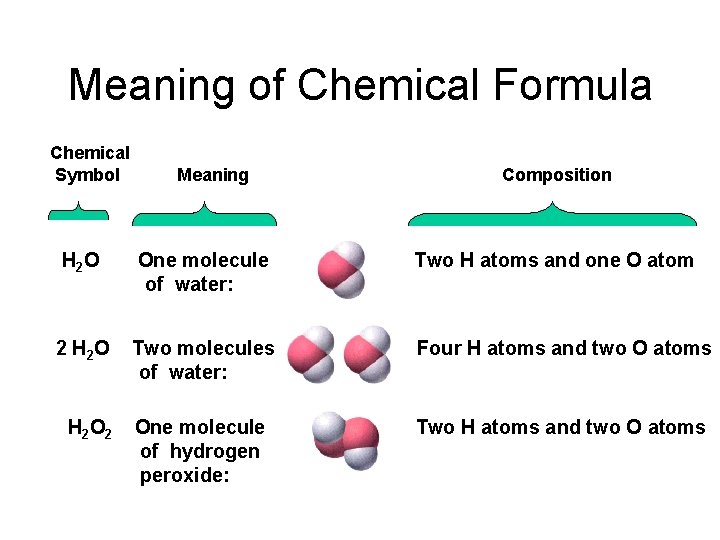
Meaning of Chemical Formula Chemical Symbol Meaning Composition H 2 O One molecule of water: Two H atoms and one O atom 2 H 2 O Two molecules of water: Four H atoms and two O atoms One molecule of hydrogen peroxide: Two H atoms and two O atoms H 2 O 2

Balancing Chemical Equations Balanced Equation – one in which the number of atoms of each element as a reactant is equal to the number of atoms of that element as a product What is the relationship between conservation of mass and the fact that a balanced equation will always have the same number of atoms of each element on both sides of an equation? Determine whether the following equation is balanced. 2 Na + H 2 O 2 Na. OH + H 2 2 Na + 2 H 2 O 2 Na. OH + H 2

Balancing Chemical Equations • Write a word equation for the reaction. • Write the correct formulas for all reactants and products. • Determine the coefficients that make the equation balance.

Write a balanced equation for the reaction between chlorine and sodium bromide to produce bromine and sodium chloride. 1) Write a word equation for the reaction. chlorine + sodium bromide bromine + sodium chloride 2) Write the correct formulas for all reactants and products. Cl 2 + Na. Br Br 2 + Na. Cl 3) Determine the coefficients that make the equation balance. Cl 2 + 2 Na. Br Br 2 + 2 Na. Cl

Write the balanced equation for the reaction between aluminum sulfate and calcium chloride to form a white precipitate of calcium sulfate. 1) Write a word equation for the reaction. aluminum sulfate + calcium chloride calcium sulfate + aluminum chloride 2) Write the correct formulas for all reactants and products. Al 2(SO 4)3 + Ca. Cl 2 Ca. SO 4 + Al. Cl 3 3) Determine the coefficients that make the equation balance. Al 2(SO 4)3 + 3 Ca. Cl 2 3 Ca. SO 4 + 2 Al. Cl 3
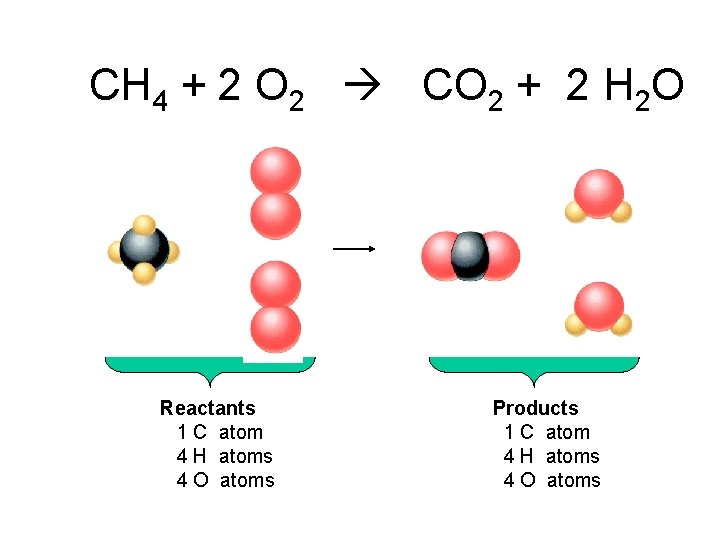
CH 4 + 2 O 2 CO 2 + 2 H 2 O Reactants 1 C atom 4 H atoms 4 O atoms Products 1 C atom 4 H atoms 4 O atoms
- Slides: 25