CHEMICAL EQUATIONS REACTION STOICHIOMETRY Chemical Equations symbolic representation

CHEMICAL EQUATIONS & REACTION STOICHIOMETRY
Chemical Equations � symbolic representation of a chemical reaction that shows: ◦ reactants on left side of reaction ◦ products on right side of equation ◦ relative amounts of each with stoichiometric coefficients ◦ attempt to show on paper what is happening at the molecular level

Law of Conservation of Matter � Matter is not created or destroyed, there is no detectable change in quantity of matter in an ordinary chemical reaction � discovered by Lavoisier � balance chemical reactions using coefficients � balancing equations is a skill acquired only with lots of practice � propane, C 3 H 8, burns in oxygen to give carbon dioxide and water C 3 H 8 + 5 O 2 ® 3 CO 2 + 4 H 2 O there are equal numbers of atoms of each element on both sides of equation

Calculations Based on Chemical Eqns Ex. 1) How many CO molecules are required to react with 27 formula units of Fe 2 O 3 to make iron and carbon dioxide
Ex. 2) What mass of CO is required to react with 140. 0 g of iron (III) oxide?
Ex. 3) How many pounds of carbon monoxide would react with 128 pounds of iron (III) oxide?

Limiting Reactant (Reagent) � Most experiments do not have the exact amounts needed to completely react. LR helps us decide which reactant is in excess (the one we have plenty of) and which reactant is limiting (the one that will determine the outcome of the reaction) � ~ If a box has 87 bolts, 113 washers and 99 nuts, how many sets consisting of one bolt, two washers and one nut can you make?
Ex. 4) What is the maximum mass of sulfur dioxide that can be produced by the reaction of 95. 6 g of carbon disulfide with 111 g of oxygen? (combustion rxn) How much of the excess chemical remains?

Percent Yields from Reactions � Most rxns in real life do not go to completion. A few reasons why ◦ Reactants did not react 100% to give the products ◦ Side reactions can occur to form undesirable products as well as the wanted products � Percent yield: The ratio of actual to theoretical

Percent Yields from Reactions � theoretical yield is what we have been calculating on paper - The amount of product formed when a limiting reactant is completely consumed. This assumes perfect conditions and gives a maximum amount. Not likely! � actual yield is what you have made in the lab, what really happens.

Ex. 5) A 10. 0 g sample of ethanol, C 2 H 5 OH, was boiled with excess acetic acid, CH 3 COOH, to produce 15. 0 g of ethyl acetate, CH 3 COOC 2 H 5. What is the percent yield? (The other product is H 2 O)

Ex. 6) Using the percent yield from Ex. 5, how much C 2 H 5 OH, is needed to give a yield of 45. 0 g of CH 3 COOC 2 H 5?

Sequential Reactions � Sequential ~ multiple steps are needed for a rxn to go to completion HNO 3 Sn C 6 H 6 C 6 H 5 NO 2 C 6 H 5 NH 2 H 2 SO 4 Conc HCl

Ex. 7) Starting with 100. g of benzene (C 6 H 6), calculate theoretical yield of nitrobenzene (C 6 H 5 NO 2) and of aniline (C 6 H 5 NH 2). (1 to 1 mole ratios)

Ex. 8) If 46. 7 g of aniline is prepared from 100. g of benzene, what is the percentage yield?

The Atomic Weight Scale & Atomic Weights define mass of 12 C as 12 amu exactly 1 amu = (1/12) mass of 12 C mass of one 24 Mg atom = 24. 3050 amu experimentally determined 1 mol of 24 Mg atoms = 24. 3050 g

Fundamental Particles The following three fundamental particles make up atoms. The table below lists these particles together with their masses and their charges.

� atomic weight - weighted average of the masses of its constituent isotopes. ◦ Atoms have masses of whole numbers, HOWEVER samples of quadrillions of atoms have a few that are heavier or lighter isotopes due to different numbers of neutrons present

Ex. 9) Naturally occurring chromium consists of four isotopes. It is 4. 31% 50 Cr, mass = 49. 946 amu, 83. 76% 52 Cr, mass = 51. 941 amu, 9. 55% 53 Cr, mass = 52. 941 amu, and 2. 38% 54 Cr, mass = 53. 939 amu. Calculate the atomic weight of chromium.

Ex. 10) Naturally occurring Lithium exists as two isotopes, 6 Li (mass = 6. 015 amu) and 7 Li (mass = 7. 016 amu). The atomic weight is 6. 941 amu. Which isotope is more abundant? Why?

� Mass Spectrometry & Isotopic Abundances spectrometry - is an analytical technique that measures the mass-to-charge ratio of charged particles. ◦ It is normally used to find the composition of a sample by generating a mass spectrum showing the masses of each type of component in the sample. This is how we find the relative abundances of elements ◦ Can also be used for identification of chemical structures, determination of mixtures, and quantitative elemental analysis. http: //www. youtube. com/watch? v=J-wao 0 O 0_q. M
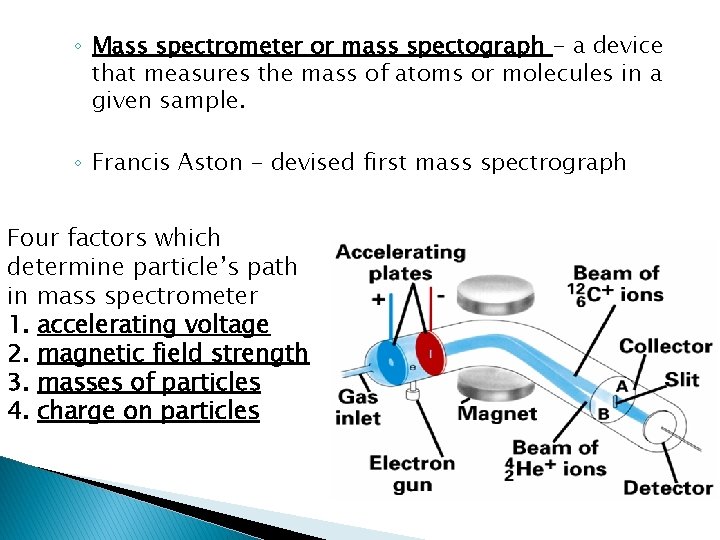
◦ Mass spectrometer or mass spectograph - a device that measures the mass of atoms or molecules in a given sample. ◦ Francis Aston - devised first mass spectrograph Four factors which determine particle’s path in mass spectrometer 1. accelerating voltage 2. magnetic field strength 3. masses of particles 4. charge on particles

How does a mass spectrometer work? � load in a pure sample of a substance � Atoms or molecules are pushed through a beam of high -speed electrons, which knocks electrons OFF the atoms or molecules turning them into cations � An electric field is applied which speeds up the cations (b/c they are repelled from the + (positive) pole and attracted to the – (negative) pole) � The accelerated cations are sent through a magnetic field and deflected in proportion to their mass. � Heavy cations deflect less than lighter cations and the ions hit a detector where the measurements are obtained from

This technique is widely used in: � identifying unknown compounds by the mass of the compound molecules or their fragments � determining compound the isotopic composition of elements in a � determining the structure of a compound by observing its fragmentation � quantifying the amount of a compound in a sample using carefully designed methods � studying the fundamentals of gas phase ion chemistry (the chemistry of ions and neutrals in vacuum)
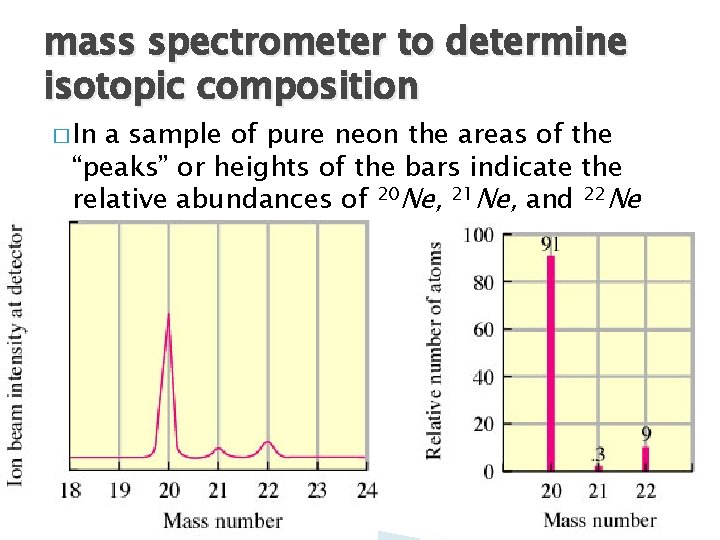
mass spectrometer to determine isotopic composition � In a sample of pure neon the areas of the “peaks” or heights of the bars indicate the relative abundances of 20 Ne, 21 Ne, and 22 Ne
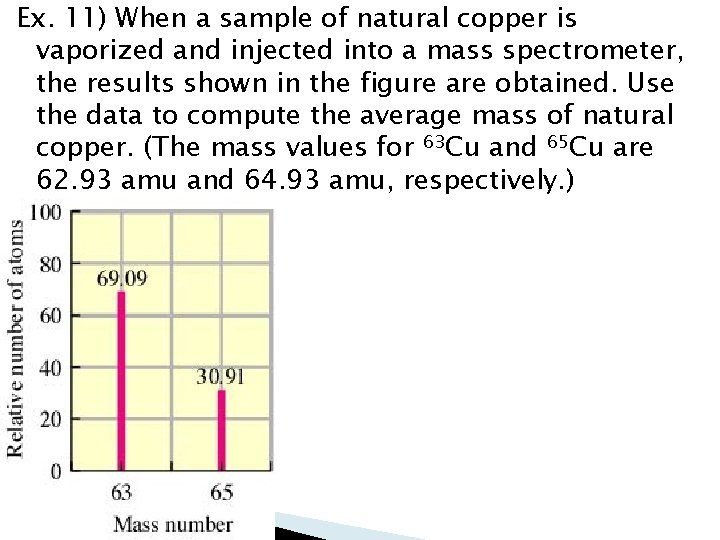
Ex. 11) When a sample of natural copper is vaporized and injected into a mass spectrometer, the results shown in the figure are obtained. Use the data to compute the average mass of natural copper. (The mass values for 63 Cu and 65 Cu are 62. 93 amu and 64. 93 amu, respectively. )

Ex. 12) The average atomic mass of boron is 10. 811 amu. The masses of the two naturally occurring isotopes 510 B and 511 B, are 10. 013 and 11. 009 amu, respectively. Which isotope is most common? Calculate the fraction and percentage of each isotope. ◦ requires a little algebra ◦ remember X + (1 -X) = 1

Ex. 13) Nickel has five isotopes that occur in the following percentages and isotopic masses. What is the isotopic mass of 60 Ni? Isotope 58 Ni 60 Ni 61 Ni 62 Ni 64 Ni Mass (amu) 57. 935 ? 60. 931 61. 928 63. 928 % 68. 27 26. 10 1. 13 3. 59 0. 91

Concentration of Solutions solution – a homogeneous mixture of 2 or more substances. Usually one substance is dissolved in another � solute – what gets dissolved � solvent – what does the dissolving (water is the universal solvent) � concentration - amount of solute dissolved in a solvent � � � Mass of solution = mass of solute + mass of solvent % by mass of solute = mass of solute x 100% mass of solution

Ex. 14) Calculate the mass of potassium nitrate required to make 277 g of solution that is 20. 0% KNO 3 by mass. What is the mass of water in the solution?

Ex. 15) What volume of 24. 5% lead(II) nitrate solutions contains 40. 0 g of lead(II) nitrate? The density of the solution is 1. 25 g/m. L

Molarity (Molar Concentration) � molarity = mol solute/L of solution = M Ex. 16) Calculate the molarity of a solution that contains 12. 8 g of sulfuric acid in 1. 70 L of solution.

Ex. 17) Determine the mass of calcium nitrate required to prepare 3. 55 L of 0. 822 M solution.

Ex. 18) The specific gravity of concentrated HCl is 1. 185 and it is 36. 31% w/w HCl. What is its molarity? (What is the p. H of the soln? )

Dilution of Solutions number of moles of solute remains constant � M 1 V 1 = M 2 V 2 works because # of moles is constant � If 10. 0 m. L of 12 M HCl is added to enough water to give 100. m. L of solution, what is concentration of the new solution? �

Ex. 19) What volume of 18. 0 M sulfuric acid is required to make 2. 50 L of a 2. 44 M sulfuric acid solution? M 1 V 1 = M 2 V 2 n 1 n 2

Solution Stoichiometry � Combine the ideas of molarity and stoichiometry Ex. 20) What volume of 0. 500 M Ba. Cl 2 is required to completely react with 4. 32 g of Na 2 SO 4?

Ex. 21) a) What volume of 0. 220 M Na. OH will react with 50. 0 m. L 0 f 0. 220 M aluminum nitrate? b) What mass of aluminum hydroxide precipitates?

Titrations � method of determining the concentration of solutions once the concentration of a standard solution is known � requires special glassware � buret, pipet, flasks, and an indicator

Ex. 22) What is the molarity of a barium hydroxide solution if 44. 4 m. L of 0. 103 M HCl is required to react with 38. 8 m. L of the barium hydroxide solution?

� In the lab, acid-base rxns are carried out as a titration: ◦ Standard solution: a soln of known concentration ◦ Standardization: process by which the conc. of a standard soln is determined. ◦ Equivalence point: point in a titration at which chemically equivalent amounts of reactants have reacted ◦ Endpoint: point in a titration where the indicator changes color and the titration is stopped.

� http: //apchemistrynmsi. wikispaces. com/

Atomic level practice Each diagram (A-P) shows a sample of a substance as viewed at the atomic level. Characterize the contents of each container in terms of the following categories: I. Homogeneous mixture, heterogeneous mixture or pure substance II. Element(s), compound(s) or both III. Solid, liquid, gas or combination of phases http: //intro. chem. okstate. edu/1314 F 00/Lectur e/Chapter 1/Atomic. html
- Slides: 43