Chemical Equations Conservation of Matter 2017 EQ Why









- Slides: 12

Chemical Equations & Conservation of Matter 2017

EQ: Why are coefficients important? (TOP RIGHT) Title your page: Chemical Reactions

(Yields/Produce) DRAW and LABEL this! Chemical Reactions Chemical Reaction – a reaction in which one or more substances are changed to new substances. Reactants – substances that are about to react (LEFT side of equation) Products – new substances produced (RIGHT side of equation) Law of Conservation of Mass – total starting mass of all reactants MUST equal the total final mass of all products (cannot create or destroy mass!)

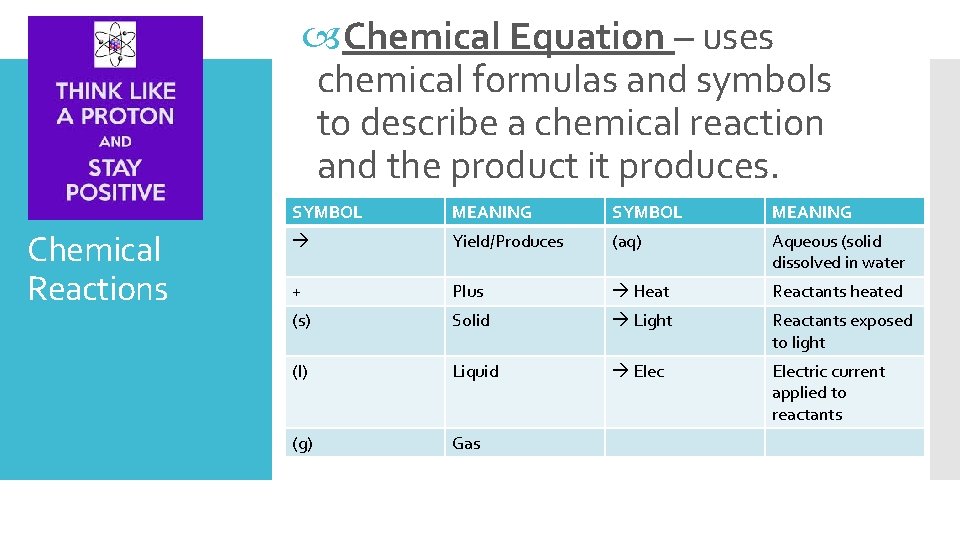
Chemical Equation – uses chemical formulas and symbols to describe a chemical reaction and the product it produces. Chemical Reactions SYMBOL MEANING Yield/Produces (aq) Aqueous (solid dissolved in water + Plus Heat Reactants heated (s) Solid Light Reactants exposed to light (l) Liquid Electric current applied to reactants (g) Gas

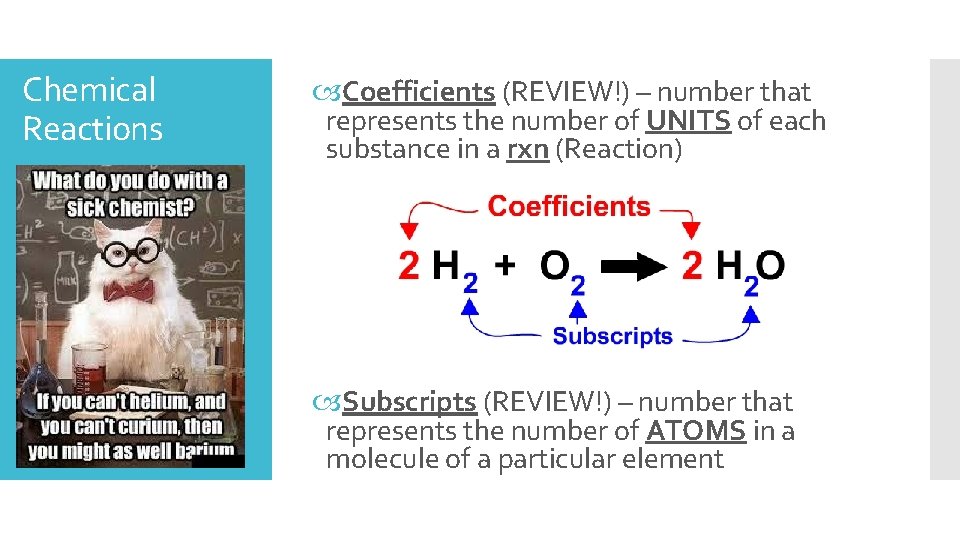
Chemical Reactions Coefficients (REVIEW!) – number that represents the number of UNITS of each substance in a rxn (Reaction) Subscripts (REVIEW!) – number that represents the number of ATOMS in a molecule of a particular element

Answer EQ: Why are coefficients important? (TOP LEFT) Videos to Review: http: //usatestprep. co m/movies/29/189/116 4 - Chemical Reactions http: //usatestprep. c om/movies/29/189/27 6 - Conservation of Matter http: //usatestprep. c om/movies/29/189/11 58 - Reactants

EQ: Why do chemical equations need to be balanced? (TOP RIGHT) Title your page: Balancing Chemical Equations

Balancing Chemical Equations Balanced chemical equations – have the same number of atoms of each element on BOTH sides of equation When balancing chemical equations NEVER change the SUBSCRIPTS (NO TOUCHY!), instead change the COEFFICIENT in FRONT of the compounds needing balance. YES!!! NO!!!

Balancing Chemical Equations Steps to Balancing a Chemical Equation STEP #1 – Write a chemical equation for the reaction using formulas and symbols – make sure REACTANTS are on the LEFT and PRODUCTS are on the RIGHT! (This will ALMOST ALWAYS be DONE for YOU!!) STEP #2 – COUNT the ATOMS in reactants and products. STEP #3 – Choose COEFFICIENTS that balance the equation. HINT! Generally, if there is an EVEN number of an element on one side and an ODD on the other side, place a 2 in front of compound containing ODD number of atoms – then COUNT AGAIN! STEP #4 – RECHECK the numbers of each ATOM on BOTH sides of equation & ADJUST coefficients if necessary REMINDER – NEVER change SUBSCRIPTS!!

Balancing Chemical Equations Let’s Work the Sample! Follow the STEPS! ___Na. Br + ___ Cl 2 ___Na. Cl + ___Br 2 List the atoms… Count the atoms… Choose coefficients – remember the HINT! Count & Recheck!

_2_Na. Br + ___ Cl 2 Balancing Chemical Equations _2__Na. Cl + ___Br 2 Reactants Products Na = 1 need 2 Br = 2 ok Cl = 2 Cl = 1 need 2 *Put a 2 in front of the ODD! Recheck! Na = 2 on BOTH sides Br = 2 on BOTH sides Cl = 2 on BOTH sides

Answer EQ: Why do chemical equations need to be balanced? (TOP LEFT) Videos to Review: http: //usatestprep. com/movies/ 29/189/122 - Balancing Chemical EQ 1 http: //usatestprep. com/movies/ 29/189/123 - Balancing Chemical EQ 2 http: //usatestprep. com/movies/ 29/189/2283 - Balancing Chemical EQ 3 http: //usatestprep. com/movies/ 29/189/124 - Balancing Chemical EQ 4 https: //phet. colorado. edu/en/si mulation/balancing-chemicalequations - Balancing Equations SIMULATOR!!