Chemical Equations Chemical equations describe the changes on
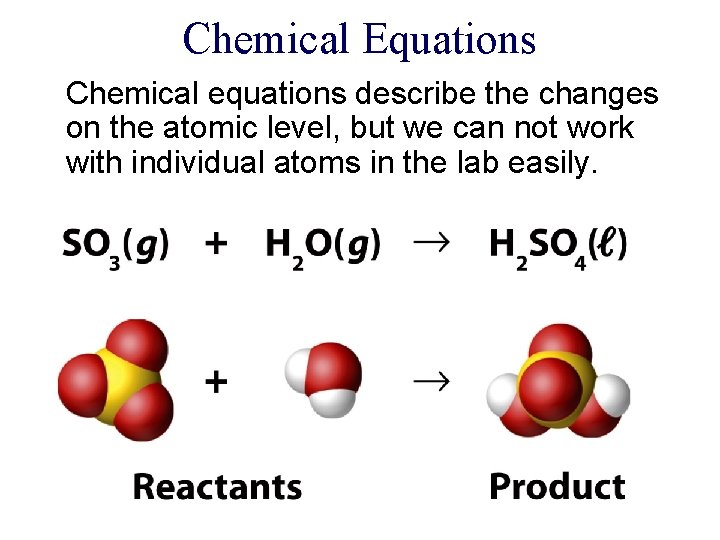
Chemical Equations Chemical equations describe the changes on the atomic level, but we can not work with individual atoms in the lab easily.

Atomic Masses Balanced equations tell us the relative numbers of molecules of reactants and products. C + O 2 CO 2 1 atom of C reacts with 1 molecule of O 2 to make 1 molecule of CO 2 If our sample consists of 2. 50 x 1019 atoms, how many CO 2 molecules can be produced?

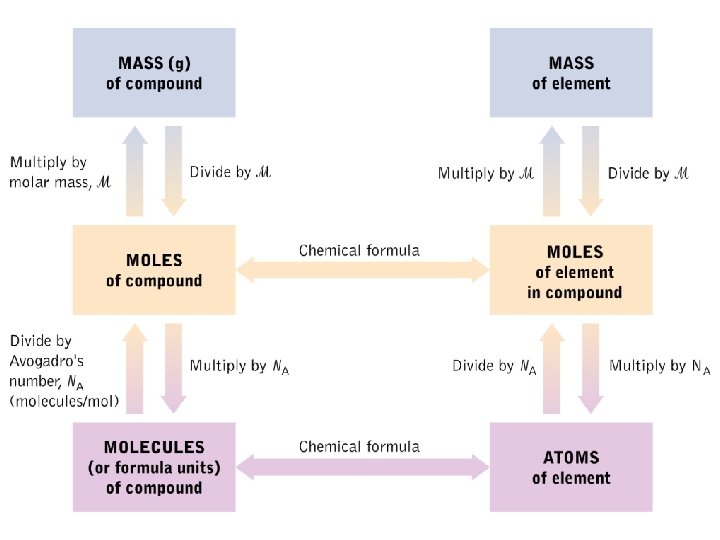
Macroscopic Quantities Counting particles as small as atoms is impractical. The mole concept allows us to relate the mass of a sample of an element to the number of atoms within it. The unit of comparison is called a mole and corresponds to 6. 022 x 1023 particles. A sample of 1 mole each of Cu atoms, and He atoms

Counting units in life & chemistry Common counting units: 1 dozen = 12 1 pair = 2 How would you write each of these as a conversion factor? (#, unit, “tag”) 1 mole = 6. 022 x 1023

One-mole samples of iron (nails), iodine crystals, liquid mercury, and powdered sulfur.

Avogadro’s Number, NA, and the Mole l The number of particles in 1 mole is called Avogadro’s Number l A mole is the number of particles equal to the number of Carbon atoms in 12 g of Carbon-12 1 mole of C atoms weighs 12. 01 g and has 6. 02 x 1023 atoms One mole = 6. 022 x 1023 units

Conversions Converting between a number of particles and an equivalent number of moles (or vice versa) is a matter of dividing (or multiplying) by Avogadro’s number.

All these samples of pure elements contain the same number (a mole) of atoms: 6. 022 x 1023 atoms.

One mole 1 dozen large nails 1 dozen small nails A mole of pure sulfur has more mass and takes up more volume than one mole of pure carbon atoms. C = 12. 01 g/mol S = 32. 07 g/mol

Molar Mass l Molar mass: the mass in grams of one mole of a compound l The average mass of an atom of helium is 4. 003 amu. l The mass of a mole of helium (6. 022 x 1023 atoms of He) is 4. 003 g. l The molar mass (M) of helium is 4. 003 g/mol.

Example Compute the number of moles and the number of atoms in 10. 0 g of Al.

Example (cont. ) Use the periodic table to determine the mass of 1 mole of Al. 1 mole Al = 26. 98 g Al • Use this as a conversion factor for grams-to-moles. •

Mole Calculations l How many moles of Ca atoms are present in 20. 0 g of calcium? l How many Cu atoms are present in 15. 0 g of copper?

Molar Mass l The relative weights of molecules can be calculated from atomic masses: water = H 2 O = 2(1. 008 amu) + 16. 00 amu = 18. 02 amu 1 mole of H 2 O will weigh 18. 02 g, therefore the molar mass of H 2 O is 18. 02 g 1 mole of H 2 O contains: 16. 00 g of oxygen and 2. 02 g of hydrogen
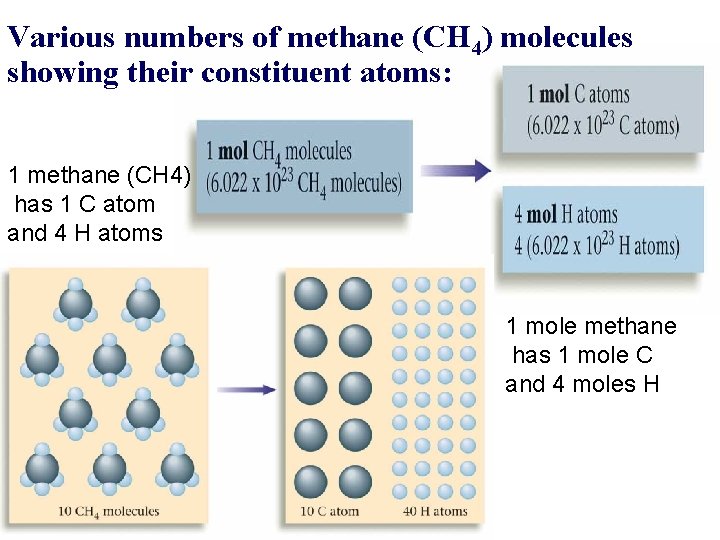
Various numbers of methane (CH 4) molecules showing their constituent atoms: 1 methane (CH 4) has 1 C atom and 4 H atoms 1 mole methane has 1 mole C and 4 moles H

Chemical Formulas as Conversion Factors A chemical formula gives us equivalences between the elements in a compound and the compound itself. l Scientists, manufacturers, farmers, etc. may want to know how much of a particular element is in a certain substance. l n l How much iron is present in a shipment of iron oxide? Many conversion factors can be found from the formula!

Molar Mass Calculate the Molar Mass of Calcium carbonate: Chemical Formula = Ca. CO 3 Ca: 40. 08 g/mol X 1 mole Ca = 40. 08 g Ca C: 12. 01 g/mol X 1 mole C = 12. 01 g C O: 16. 00 g/mol X 3 mole O = 48. 00 g O 40. 08 + 12. 01 + 48. 00 = 100. 09 g/mol Ca. CO 3

Mole Calculations l How many grams are present in 3. 40 moles of nitrogen gas (N 2)? l How many molecules are present in 5. 32 moles of chalk (Ca. CO 3)? A. How many oxygen atoms are present in this sample?

Percent Composition by Mass l Compound formula tells you how many of each type of atom are in one formula unit of a compound l Using formula mass, you can calculate the % by mass of each element in a compound l Example: Calculate the % composition by mass of CO 2 Formula mass of CO 2 = 12. 01 + (2 x 16. 00) = 44. 01 amu %C = (12. 01 amu)/(44. 01 amu) x 100% = 27. 29% %O = (2 x 16. 00 amu)/(44. 01 amu) x 100% = 72. 71%

Juglone, a dye from the husks of black walnuts, and a natural herbicide: C 10 H 6 O 3 Calculate the molar mass of juglone. Find the mass percent of oxygen in this compound.

Mass Percent Calculations l Mass % O in juglone = (? g O per 100 g juglone) l Assume 100 g C 10 H 6 O 3 (find molar mass) l Use the ratio in the compound to convert from juglone to oxygen: Note: this is not molecular n l 1 mole juglone has 3 moles O oxygen, it is the # oxygen atoms in the formula Use molar mass to convert to grams of O

Empirical Formulas l Empirical formulas give the lowest wholenumber ratio of atoms in a compound l Example: for glucose, Gives the formula of an actual compound molecular formula = C 6 H 12 O 6 empirical formula = CH 2 O l Gives the lowest ratio of elements (useful for % composition) Can calculate empirical formula from masses or from % composition by mass Empirical means observed: Combustion analysis gave empirical formulas!

Calculating Empirical Formulas l Example: Calculate empirical formula for compound with 8. 0 g C and 2. 0 g H 8. 0 g C x (1 mol/12. 01 g) = 0. 6661 mol C 2. 0 g H x (1 mol/1. 008 g) = 1. 984 mol H l Divide by the smallest number to obtain a whole # ratio 0. 6661 mol C/0. 6661 mol = 1. 000 C = 1 C 1. 984 mol H/0. 6661 mol = 2. 979 H = 3 H Empirical formula = CH 3 Round to the closest integer

Calculating Molecular Formulas Example: Calculate empirical formula for l compound with 8. 0 g C and 2. 0 g H Empirical formula = CH 3 Molar mass = 30. 0 g/mol. What is the molecular formula? l n Compare the mass of the empirical formula with the mass of the molecular formula
- Slides: 25