Chemical Equations Chemical Equations Describe a chemical change

Chemical Equations

Chemical Equations • Describe a chemical change using formulas and symbols • Nickel (II) chloride, dissolved in water, plus sodium hydroxide, dissolved in water, produces solid nickel (II) hydroxide plus sodium chloride, dissolved in water Ni. Cl 2(aq)+2 Na. OH(aq)→Ni(OH)2(s)+2 Na. Cl(aq)

Why use chemical equations?

Prediction Symbol Reactants Products Coefficient Meaning

Prediction Symbol → + Meaning
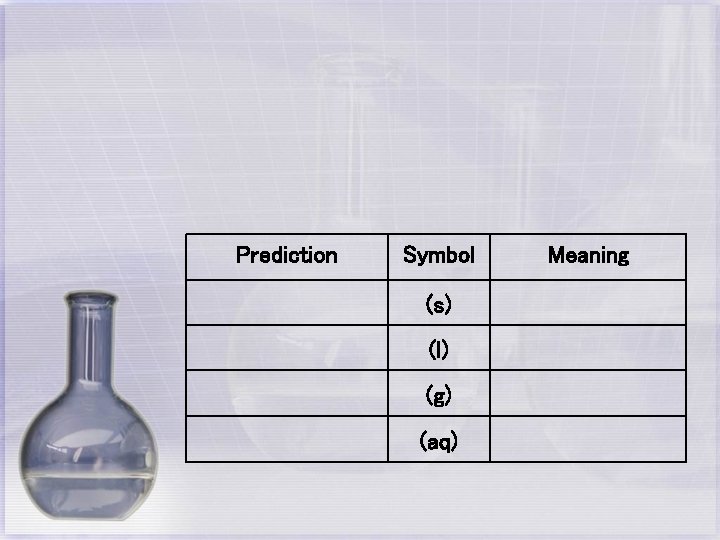
Prediction Symbol (s) (l) (g) (aq) Meaning

Identify parts of this equation Ni. Cl 2(aq)+2 Na. OH(aq)→Ni(OH)2(s)+2 Na. Cl(aq)

Conservation of Mass • Total mass of the products equals the mass of the reactants • To summarize: – Whatever you have in the beginning must be there in the end

Why? • Matter is not created nor destroyed

How can you determine if the Law of Conservation of Mass is being applied to an equation? 1. Calculate the number of atoms of each element on the reactant side 2. Calculate the number of atoms of each element on the product side If they are equal, the law is applied.

Example: Ni. Cl 2(aq)+2 Na. OH(aq)→Ni(OH)2(s)+2 Na. Cl(aq)

Example: Ni. Cl 2(aq)+Na. OH(aq)→Ni(OH)2(s)+Na. Cl(aq) If they are not equal, the law is not applied. You must balance the equation!

Balancing Equations 1. Write the chemical equation. 2. Count the atoms of each element in the reactant side. 3. Count the atoms of each element in the product side. 4. Determine if it is balanced. 5. Choose coefficients and recount the atoms until balanced.

Why do we have balanced equations? To follow the
- Slides: 14