Chemical Equations and Reactions Describing Chemical Reactions A

Chemical Equations and Reactions

Describing Chemical Reactions • A chemical reaction is the process by which one or more substances are changed into one or more different substances. • Reactants are the original substances • Products are the resulting or new substances • Law of Conservation of mass states, the total mass of reactants must equal the total mass of products for any given chemical reaction. • Chemical equations represent, with symbols and formulas, the identities and relative molecular or molar amounts of the reactants and products in a chemical reaction
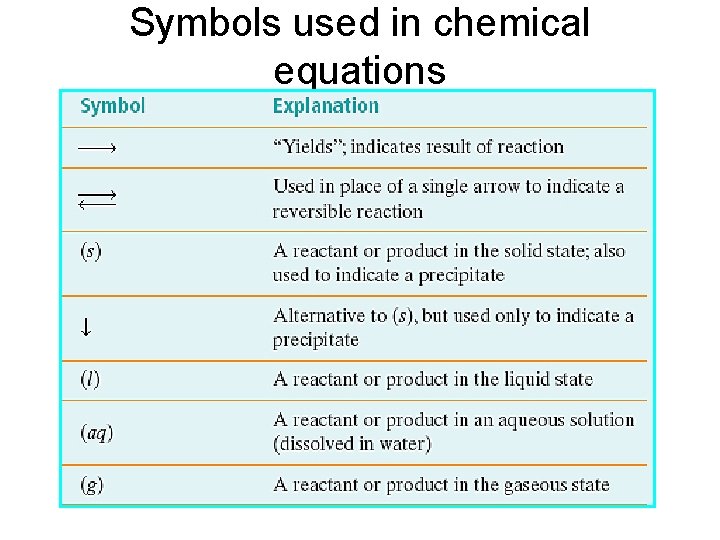
Symbols used in chemical equations

Symbols used in chemical equations (cont. )

Symbols used in chemical equations (cont. )

Indications of a Chemical Reaction • Evolution of energy as heat and light • Production of a gas (bubbling) • Formation of a precipitate – A solid that is produced as a result of a chemical reaction in solution and that separates from the solution. • Color change

Characteristics of Chemical Equations All properly written chemical equations will share these characteristics • The equation must represent known facts. – All reactants and products must be identified • The equation must contain the correct formulas for the reactants and products. – Watch out for HOFBr. INCl’s • The law of conservation of mass must be satisfied. – Atoms are never created or destroyed in a chemical reaction. – Atoms on the reactant side must equal atoms on the product side. – Coefficients are used to “balance” all the atoms in a chemical equation. They are inserted in front of a chemical formula and act as a multiplier.

Types of Chemical Reactions • Categorizing chemical reactions into different types helps us to predict the products which will occur during reaction • There are 5 basic types of Reactions we will discuss: – – – Synthesis Decomposition Single displacement (replacement) Double displacment (replacement) Combustion

Synthesis Reaction • Also known as a composition reaction • Two or more substances react to form a new compound A + B AB 2 Mg(s) + O 2(g) 2 Mg. O(s)

Decomposition Reactions • A single compound undergoes a reaction that produces two or more simplier substances. AB A + B 2 H 2 O 2 H 2 + O 2 Ca. CO 3 Ca. O + CO 2

Combustion Reactions • A substance combines with oxygen and produces a combination of water and carbon dioxide. Usually involves hydrocarbons Cx. Hy + O 2 CO 2 + H 2 O C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O

Single Displacement Reactions • One element in a compound replaces another element in a different compound A + BC AC + B 2 Al + 3 Pb(NO 3)2 3 Pb + 2 Al(NO 3)3

Single Displacement Reactions (cont. ) • Not all single displacement reaction will take place. • Must refer to the activity series of metals to predict whether the metal is “strong” enough to displace the other metal.
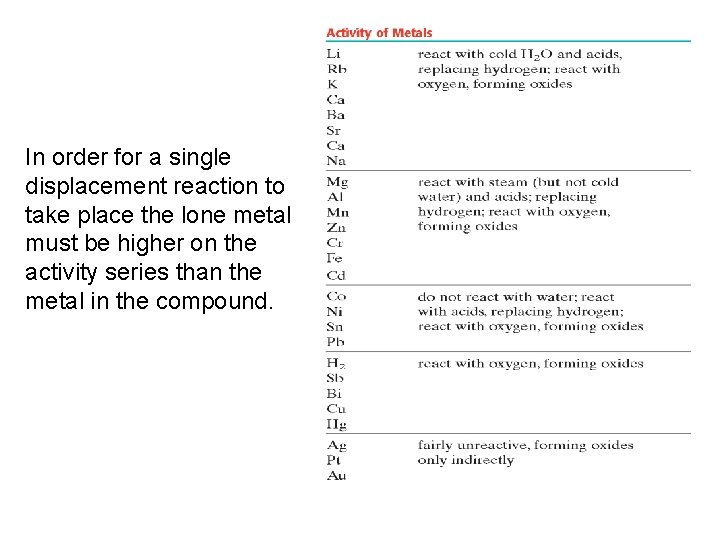
In order for a single displacement reaction to take place the lone metal must be higher on the activity series than the metal in the compound.

Double Displacement Reactions • The ions of two compounds exchange places to form two new compounds AB + CD AD + CB 2 KI + Pb(NO 3)2 Pb. I 2 + 2 KNO 3
- Slides: 15