Chemical Equations and Reactions 1 Describing Chemical Changes

Chemical Equations and Reactions 1

Describing Chemical Changes or Reactions n Chemical Reaction: process by which one or more substances are changed into one or more different substances. n Original Substances Resulting Substances Reactants Products n 2

Evidence of a Chemical Reaction: n 1. 2. 3. 4. 5. Easily observed changes that indicate a chemical reaction has occurred Evolution of heat and light Production of a gas Formation of a precipitate Color Change Difficult to reverse 3

Word Equations n Write a word equation to describe the reaction of methane (natural gas) as it burns in the air to produce carbon dioxide and water vapor. n Methane + oxygen carbon dioxide + water 4

Representing a Chemical Change n Chemical Equation: represents with symbols and formulas, the identities and relative amounts of the reactants and products in a chemical reaction. 5

Chemical Equation n n n Law of Conservation of Mass must be taken into account. Adjust equation so that the same type of atoms and numbers of atoms are the same on both sides of the equation. CH 4(g) + O 2(g) CO 2(g) + H 2 O(g) CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) Symbols used in chemical equations, see p. 246. (s) solid, (l) liquid, (g) gas, (aq) aqueous, Δ heat Notice (aq) refers to a solution in water and thus to a homogeneous mixture not a pure substance 6

Write a word & chemical equation n n Solid sodium oxide is added to water (at room temp. ) and forms sodium hydroxide which is dissolved in water. Sodium oxide(s) + water(l) sodium hydroxide (aq) Na 2 O(s) + H 2 O(l) Na. OH(aq) Na 2 O(s) + H 2 O(l) 2 Na. OH(aq) 7

Write a word & chemical equation n n Hydrogen peroxide in an aqueous solution decomposes to produce oxygen and water. Hydrogen peroxide oxygen + water H 2 O 2(aq) O 2(g) + H 2 O(l) 2 H 2 O 2(aq) O 2(g) + 2 H 2 O(l) 8

Write a word & chemical equation n Solid copper metal reacts with aqueous silver nitrate to produce solid silver metal and aqueous copper(II) nitrate. n Copper(s) + silver nitrate(aq) silver(s) + copper(II)nitrate(aq) Cu(s) + Ag. NO 3 (aq) Ag (s) + Cu(NO 3)2 (aq) Cu(s) + 2 Ag. NO 3(aq) 2 Ag (s) + Cu(NO 3)2 (aq) n n 9

Write a word & balanced chemical equation n Solid zinc metal reacts with aqueous copper(II) sulfate to produce solid copper metal and aqueous zinc sulfate. Zinc(s) + copper(II)sulfate(aq) copper (s) + zinc sulfate (aq) Zn(s) + Cu. SO 4 (aq) Cu(s) + Zn. SO 4(aq) 10

Guidelines for Balancing Equations 1. 2. 3. Write the chemical equation with the correct formulas for the reactants and products. Count the number and types of atoms on both sides of the equation. Balance the atoms of the elements that appear only once on each side of the equation. 11

Balancing Equations 4. 5. 6. 7. Balance polyatomic ions that appear on each side of the equation. Balance hydrogen and oxygen after all other atoms have been balanced. Check both sides – count each element. Use lowest coefficients. 12

What’s it spell? n n n n Hydrogen Oxygen Fluorine Bromine Iodine Nitrogen Chlorine HOFBr. INCl 13

HOFBr. INCl n n Elements that exist as diatomic molecules. Cl 2 is electrically neutral, whereas in a compound such as Na. Cl, chlorine is the chloride ion with a negative charge. 14

Write a Balanced Chemical Equation: n Zinc(s) + hydrochloric acid(aq) zinc chloride (aq) and hydrogen gas. n Zn(s) + HCl (aq) Zn. Cl 2(aq) + H 2(g) n Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) 15
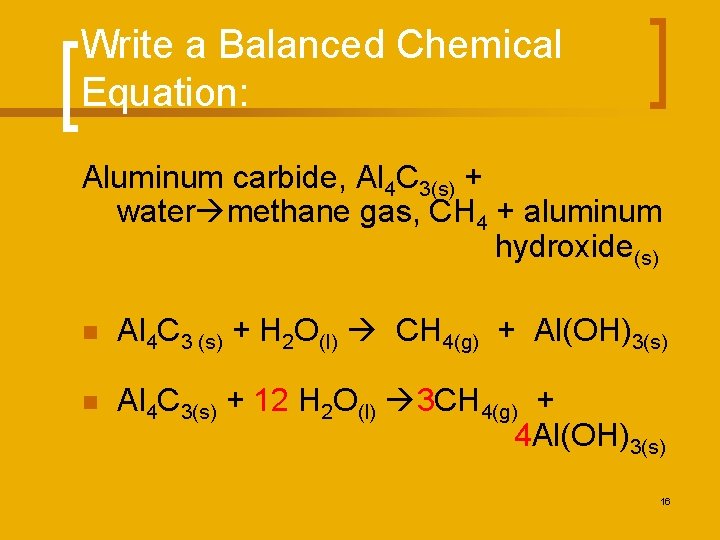
Write a Balanced Chemical Equation: Aluminum carbide, Al 4 C 3(s) + water methane gas, CH 4 + aluminum hydroxide(s) n Al 4 C 3 (s) + H 2 O(l) CH 4(g) + Al(OH)3(s) n Al 4 C 3(s) + 12 H 2 O(l) 3 CH 4(g) + 4 Al(OH)3(s) 16

Write a Balanced Chemical Equation: Aluminum sulfate + calcium hydroxide aluminum hydroxide + calcium sulfate Al 2(SO 4)3 + Ca(OH)2 Al(OH)3 + Ca. SO 4 n Al 2(SO 4)3 +3 Ca(OH)2 2 Al(OH)3 + 3 Ca. SO 4 17

CHO method: Combustion Reactions n n Combustion Reaction contains a hydrocarbon + oxygen to yield carbon dioxide and water. Balance carbon first, then hydrogen and finally oxygen. C 2 H 6 + O 2 CO 2 + H 2 O 2 C 2 H 6 +7 O 2 4 CO 2 + 6 H 2 O 18

Types of Chemical Reactions Five Basic Types of Chemical Reactions: 1. Synthesis 2. Decomposition 3. Single-displacement 4. Double-displacement 5. Combustion 19

Synthesis n Two or more substances combine to form a new compound. n A + B AB (A & B may be compounds or elements) 20

Synthesis Reactions Here is an example 21

Synthesis Examples n Mg(s) + O 2(g) Mg. O(s) 2 Mg(s) + O 2(g) 2 Mg. O(s) n Ba (s) + S 8 (s) Ba. S(s) 8 Ba (s) + S 8 (s) 8 Ba. S(s) Na(s) + Cl 2(g) Na. Cl 2 Na(s) + Cl 2 (g) 2 Na. Cl n n 22

Basic Oxides n Metal oxides react with water to produce Bases e. g. Na 2 O + H 2 O → 2 Na. OH K 2 O +H 2 O Ca. O +H 2 O 2 KOH Ca(OH)2 23
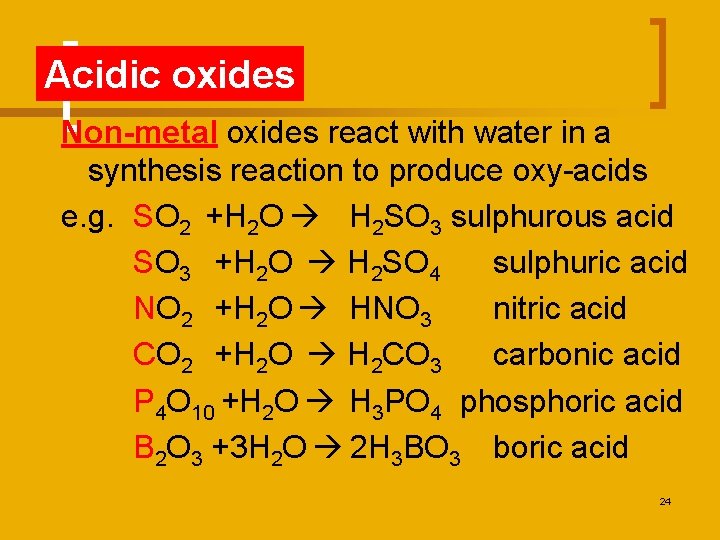
Acidic oxides Non-metal oxides react with water in a synthesis reaction to produce oxy-acids e. g. SO 2 +H 2 O H 2 SO 3 sulphurous acid SO 3 +H 2 O H 2 SO 4 sulphuric acid NO 2 +H 2 O HNO 3 nitric acid CO 2 +H 2 O H 2 CO 3 carbonic acid P 4 O 10 +H 2 O H 3 PO 4 phosphoric acid B 2 O 3 +3 H 2 O 2 H 3 BO 3 boric acid 24

Acidic oxide and basic oxide together! n n n When an acidic oxide (ie nonmetal oxide) reacts with a basic oxide (ie metal oxide) the result is a salt with a polyatomic ion Li 2 O + CO 2 → lithium carbonate Li 2 O + CO 2 → Li 2 CO 3 25

Synthesis reactions- overview n A- elmt +elmt = Cmpd ¡ n B- water + NMO(acidic oxide) =Acid ¡ n H 2 O + CO 2= H 2 CO 3 C- water + MO (basic oxide) = Base ¡ n 2 Na + Cl 2= 2 Na. Cl H 2 O + Li 2 O= 2 Li. OH D- MO + NMO= Salt with polyatomic ¡ Li 2 O + CO 2= Li 2 CO 3 26

Decompositon n A single compound undergoes a reaction that produces 2 or more simpler substances. AB A + B (A & B can be elements or compounds) n 27

Decomposition Reactions • Another view of a decomposition reaction: 28

Decomposition Examples n n H 20 (l) H 2 (g) + O 2 (g) 2 H 20 (l) 2 H 2 (g) + O 2 (g) n Hg. O (s) Hg (g) + O 2 (g) 2 Hg. O (s) 2 Hg (g) + O 2 (g) n Ca. CO 3 (s) Ca. O (s) + CO 2 (g) n 29

Decomposition Exceptions • Carbonates and bicarbonates are special case decomposition reactions that do not go to the elements. • Carbonates (CO 32 -) decompose to carbon dioxide and a metal oxide • • Example: Ca. CO 3 CO 2 + Ca. O Hydrogen Carbonates (HCO 3 -) decompose to carbon dioxide, water and a metal oxide • Example: Ca(HCO 3)2 2 CO 2 + H 2 O + Ca. O 30

Decomposition reactionsopposites of synthesis n A- Cmpd = elmt +elmt ¡ n B- Acid = water + NMO(acidic oxide) ¡ n H 2 CO 3 = H 2 O + CO 2 C- Base = water + MO (basic oxide) ¡ n 2 Na. Cl = 2 Na + Cl 2 2 Li. OH = H 2 O + Li 2 O D- Salt with polyatomic = MO + NMO ¡ Li 2 CO 3 = Li 2 O + CO 2 31

Single Displacement Reactions n One element replaces a similar element in a compound. n A + BC AC + B where A and B are metals and C is a non-metal or A + BC BA + C where A and C are non- metals and B is a metal n 32
Single Displacement Reactions • Another view: 33

Single Displacement Examples 2 Al(s) + 3 Pb(NO 3)2 (aq) 3 Pb(s) + 2 Al(NO 3)3 (aq) n Al replaces Pb (2 metal ions change position) n Cl 2(g) + 2 KBr(aq) 2 KCl(aq) + Br 2(g) n Cl replaces Br (2 non-metal ions change position) 34

Activity Series n The ability of an element to react is called the elements “activity. ” n The more readily an element reacts – the greater “activity” it has. 35

: Activity Series n n A list organized by the ease at which certain elements will undergo a chemical reaction. (p. 89) Li K Ba Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Hg Ag Au n n The H near the end represents H of acid and NOT H of water Notice all the metals used in jewelry are on the far right side n The order is determined by single-displacement reactions. n The list on p. 165, the first 5 react with water (HOH) and the ones up to H react with acid (first 12) 36

Activity Series n Most Active Most active Least active n Least Active n The most active element is placed at the top (or on the left) and can replace each of the elements below it. (or to the right) 37

n An element further down the list can only replace an element below it –not above it. n The Activity Series will predict whether or not a reaction will take place. 38

increasing reactivity lithium potassium barium calcium sodium magnesium aluminum zinc iron nickel Tin Lead copper mercury silver gold Activity series of metals React vigorously with cold water React slowly with cold water React very slowly with steam but are quite reactive in acid React moderately with acid Unreactive in acid 39

increasing reactivity lithium potassium barium calcium sodium magnesium aluminum zinc iron Activity series of metals used to predict reactions 1. Will potassium react with sodium iodide? Yes/No? ? ? 2. Will calcium react with copper selenide? Yes/No? ? ? 3. Will sodium iodide react with potassium? Yes/No? ? ? Should be same answer as 1 nickel Tin Lead copper mercury silver gold 4. Will zinc chloride react with iron? Yes/No? ? ? 5. Will zinc chloride react with water? Yes/No? ? ? 6. Will zinc chloride react with an acid? Yes/No? ? ? 40

Halogen activity series n n For the halogens in a single displacement reaction the order is fluorine (on top) most reactive followed by Cl, Br, I, At (least reactive) just as the order in column 17 of the periodic table Thus, F can replace I but not the other way around 41
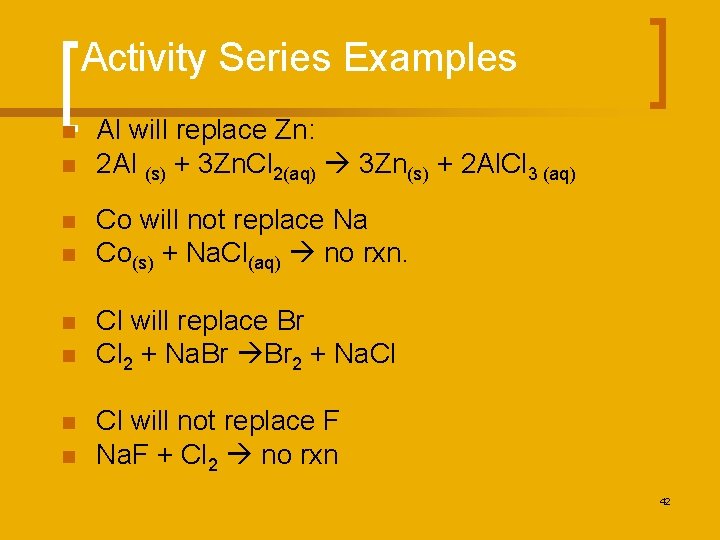
Activity Series Examples n n n n Al will replace Zn: 2 Al (s) + 3 Zn. Cl 2(aq) 3 Zn(s) + 2 Al. Cl 3 (aq) Co will not replace Na Co(s) + Na. Cl(aq) no rxn. Cl will replace Br Cl 2 + Na. Br Br 2 + Na. Cl Cl will not replace F Na. F + Cl 2 no rxn 42

Double Displacement Reactions n n n The ions of two compounds exchange places in an aqueous solution to form two new compounds. A double displacement reaction is likely to occur if on the product side there is the formation of a liquid (as in water), a gas (CO 2, SH 2, NH 3), or a solid (a precipitate insoluble i. e (s) for solid…see charts) AB + CD AD + CB 43

Double Displacement Examples 2 KI(aq) + Pb(NO 3)2(aq) 2 KNO 3(aq) + Pb. I 2 (s) n n K cation replaces Pb cation I anion replaces nitrate anion Fe. S(s) + 2 HCl(aq) H 2 S(g) + Fe. Cl 2(aq) n n Fe cation replaces H cation S anion replaces Cl anion 44

…but there is more!! • • • Double displacement reactions only occur if at least one product is a solid, liquid or a gas! Like: CO 2(g), H 2 O (l), NH 3(g), SO 2(g), Pb. SO 4(s), H 2 S (g) Not like ______ (aq) + _____ (aq) If both products are aqueous, then no reaction occurs!! Check your solubility rules! Write in the state of your previous products If the products H 2 CO 3, H 2 SO 3, NH 4 OH are produced in a double displacement reaction, they further break down and form a gas and water. 45

n n n H 2 CO 3 breaks down into CO 2(g) and H 2 O(l) H 2 SO 3 breaks down into SO 2(g) and H 2 O(l) NH 4 OH breaks down into NH 3(g) and H 2 O(l) Example: Na 2 SO 3(aq) + 2 HCl(aq) 2 Na. Cl(aq) + H 2 SO 3(aq) Na 2 SO 3(aq)+2 HCl(aq) 2 Na. Cl(aq) +H 2 O(l) + SO 2(g) 46

Some simple solubility rules n n n All acids and alkali metal salts and NH 4+ salts are soluble All nitrate and acetate salts are soluble. All chlorides are soluble except Ag. Cl and Hg 2 Cl 2 Pb. Cl 2 All sulfates are soluble except Pb. SO 4, Hg 2 SO 4, Sr. SO 4, Ba. SO 4. Ag 2 SO 4 and Ca. SO 4. All sulfides are insoluble except those of the Group IA (1), IIA (2) and ammonium sulfide. All hydroxides are insoluble except those of the group IA(1) and Ba(OH)2. Sr(OH)2 and Ca(OH)2 are slightly soluble. 47

Reading the solubility chart n n If a compound is soluble…it dissolves in water then you can write (aq) after the formula if water is present. If a compound is insoluble (not soluble) then you can write (s) after the formula since it does not dissolve. 48

Mixed Practice • 1. 2. 3. 4. 5. State the type, predict the products, and balance the following reactions: Ba. Cl 2 + H 2 SO 4 Zn + Cu. SO 4 Cs + Br 2 Fe. CO 3 C 3 H 8 + O 2 49

Mixed Practice • 1. 2. 3. 4. 5. State the type, predict the products, and balance the following reactions: Ba. Cl 2 + H 2 SO 4 2 HCl +Ba. SO 4 Zn + Cu. SO 4 Cu + Zn. SO 4 2 Cs + Br 2 2 Cs. Br CS 2 C + 2 S C 3 H 8 + 5 O 2 3 CO 2 + 4 H 20 50

Practice makes perfect n n n Try we, ce, bce p. 155 Try synthesis/decomposition reactions p. 161 Try single/double displacement reactions p. 169 p. 177 51
- Slides: 51