Chemical Engineering Thermodynamics And Reaction Engineering System A





























- Slides: 45

Chemical Engineering Thermodynamics And Reaction Engineering

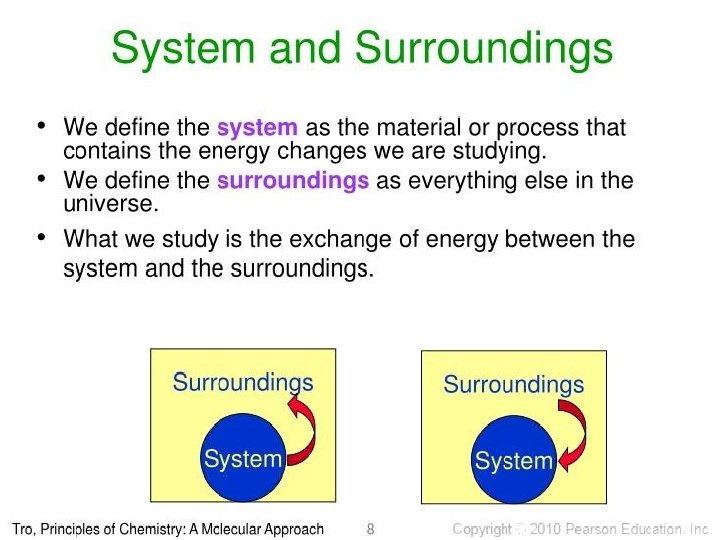
System: A thermodynamic system is a quantity of matter of fixed identity, around which we can draw a boundary. The boundaries may be fixed or moveable. Work or heat can be transferred across the system boundary. Surrounding: Everything outside the boundary is the surroundings


Homogenous System: A homogeneous thermodynamic system is defined as the one whose chemical composition and physical properties are the same in all parts of the system, or change continuously from one point to another.

Heterogeneous System: A heterogeneous system is denned as one consisting of two or more homogeneous bodies. The homogeneous bodies of a heterogeneous system are referred to as phases. Each phase is separated from other phases by interfaces, or boundaries. An example of a heterogeneous system is water with ice floating in it. This system has two homogeneous bodies, water and ice.

State Function: It is a thermodynamic term that is used to name a property whose value does not depend on the path taken to reach that specific value. State functions are also known as point functions. A state function only depends on the current state of thermodynamic system and its initial state

Path Function: It is a thermodynamic term that is used to name a property whose value depends on the path taken to reach that specific value. In other words, a path function depends on the path taken to reach a final state from an initial state. Path function is also called a process function.

Extensive properties • Extensive properties are additive. Thus, if the system is divided into a number of subsystems, the value of the property for the whole system is equal to the sum of the values for the parts. Volume is an extensive property. . • Specific properties are extensive properties per unit mass and are denoted by lower case letters.

Intensive properties • Intensive properties do not depend on the quantity of matter present. Temperature and pressure are intensive properties. • Specific properties are intensive because they do not depend on the mass of the system.

Internal Energy : • In thermodynamics, the internal energy of a system is the total energy contained within the system. • It keeps account of the gains and losses of energy of the system that are due to changes in its internal state. • The internal energy U of a given state of the system is determined relative to that of a standard state of the system, by adding up the macroscopic transfers of energy

Enthalpy : • Enthalpy , a property of a thermodynamic system, is equal to the system's internal energy plus the product of its pressure and volume. • In a system enclosed so as to prevent matter transfer, for processes at constant pressure, the heat absorbed or released equals the change in enthalpy. • The unit of measurement for enthalpy in the SI is the joule.


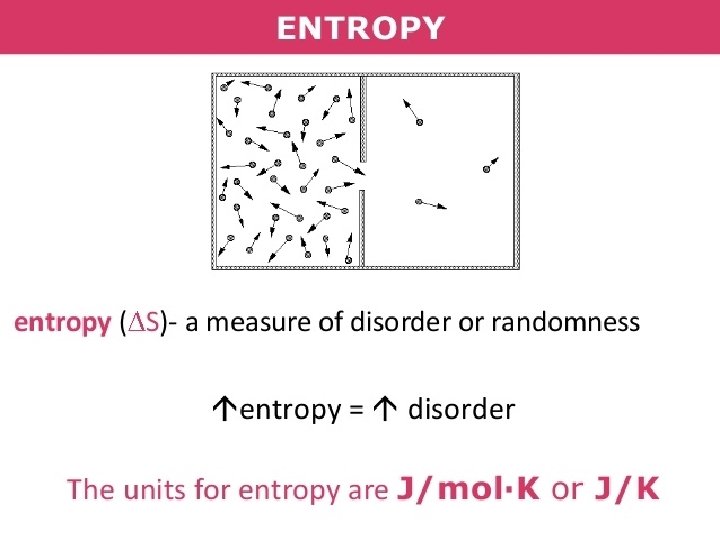
Entropy : • Entropy, the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. • Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.





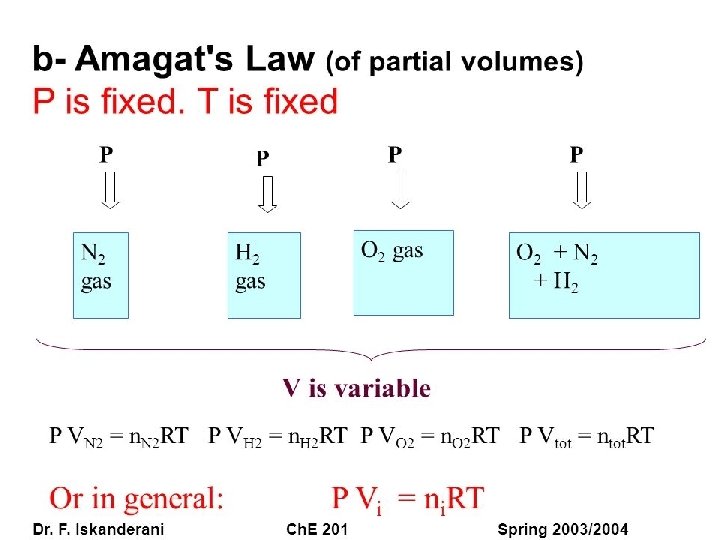
Amagat's law : • According to Amagat's law of partial volume, the total volume of a non-reacting mixture of gases at constant temperature and pressure should be equal to the sum of the individual partial volumes of the constituent gases.







UNIT : 2














UNIT : 3





