Chemical Engineering Equilibrium Separations Lectures 14 17 Oct

“Chemical Engineering Equilibrium Separations” Lectures 14 17 Oct 2012 1

Overview • Aspen. Plus: o Shortcut methods: DSTWU o Rigorous method: RADFRAC • Efficiencies • Introduction to multicomponent distillation (Chapter 9) 2

Multicomponent Distillation (Introduction) • In binary distillation we could specify x. D and x. B …. • Now: define “Key Components” Decreasing Relative Volatility 1 2 3 4 Keys Light Heavy Most of LK obtained in distillate product Most of HK obtained in bottoms product Today, most multicomponent systems are solved rigorous simulation. But need to do shortcut methods to get good starting point (FUG-Kirkbride). 3

Multicomponent Shortcut Methods Minimum number of stages: Fenske Equation 4

Multicomponent Shortcut Methods Minimum reflux ratio: Underwood Equation Case A: NKs don’t distribute Case B & C: NKs distribute, or there is a “sandwich” NK: see Wankat; numerical iterative procedures can be involved. R = factor * Rmin Approximate number of equilibrium stages (N): Gilliland correlation 5
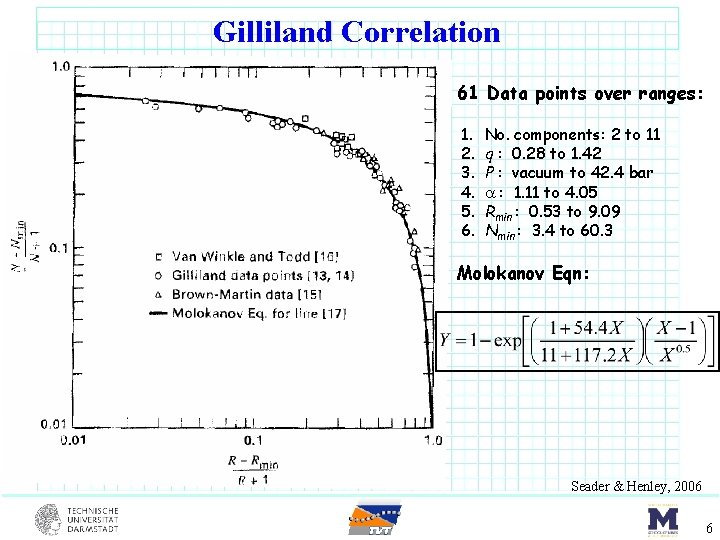
Gilliland Correlation 61 Data points over ranges: 1. 2. 3. 4. 5. 6. No. components: 2 to 11 q : 0. 28 to 1. 42 P : vacuum to 42. 4 bar : 1. 11 to 4. 05 Rmin : 0. 53 to 9. 09 Nmin : 3. 4 to 60. 3 Molokanov Eqn: Seader & Henley, 2006 6

Multicomponent Shortcut Methods Optimum feed stage location (NF): Kirkbride Equation DSTWU (Aspen. Plus) • Uses Winn, Underwood, and Gilliland methods to find Nmin, Rmin, & N. • Specify LK and HK recoveries in the distillate product stream • If input -1. 2 for reflux ratio; it finds N at R = 1. 2 * Rmin. • N given by DSTWU is number of equilibrium stages (includes partial condensers and/or partial reboilers) 7

In-Class Problem benzene (17 mol%) toluene (66 mol%) m-xylene (17 mol%) DSTWU F = 100 kmol/s sat’d liquid 1 atm Component Benzene Toluene m-Xylene 1. 0135 bar Tbp o. C 80. 1 110. 7 139. 1 Pi star [bar] 80. 1 o. C 123 o. C 1. 0135 3. 217 0. 387 1. 42 0. 151 0. 645 8
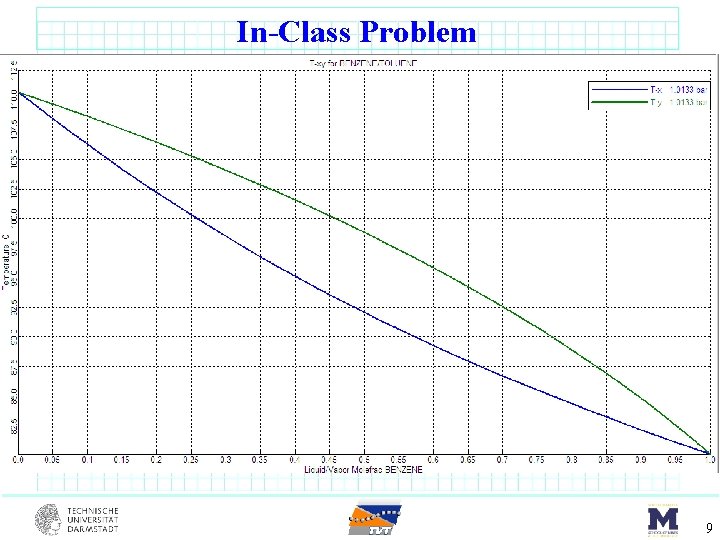
In-Class Problem 9
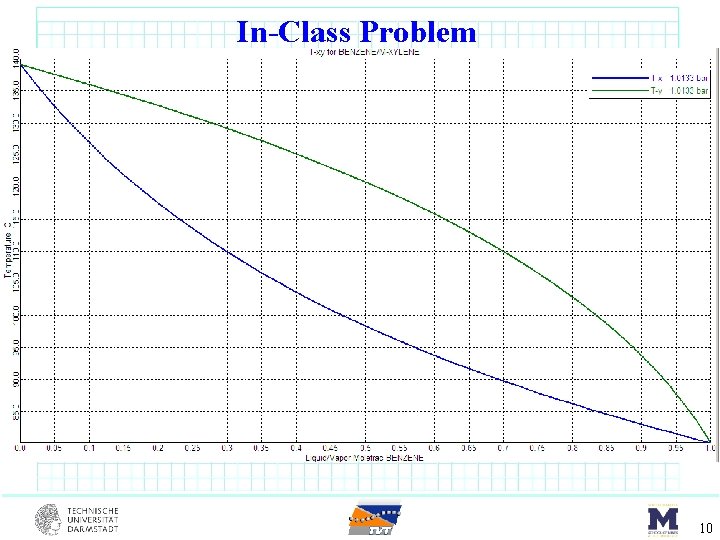
In-Class Problem 10
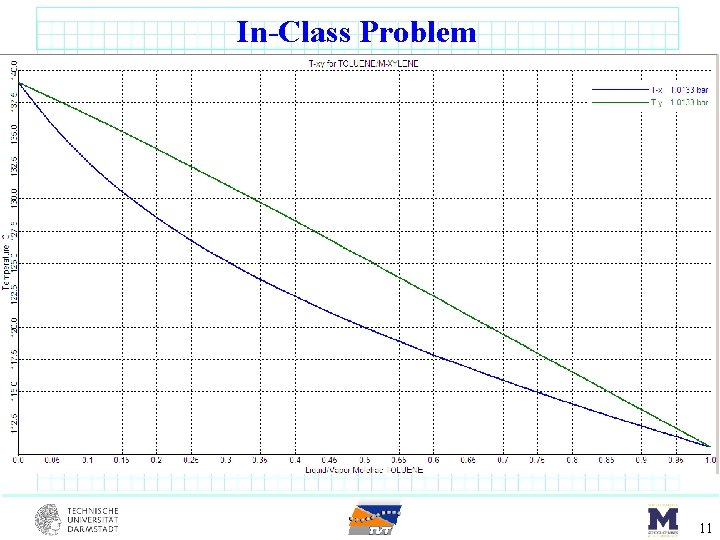
In-Class Problem 11

In-Class Problem By hand calculations first. Then use to verify Aspen. Plus results… 12
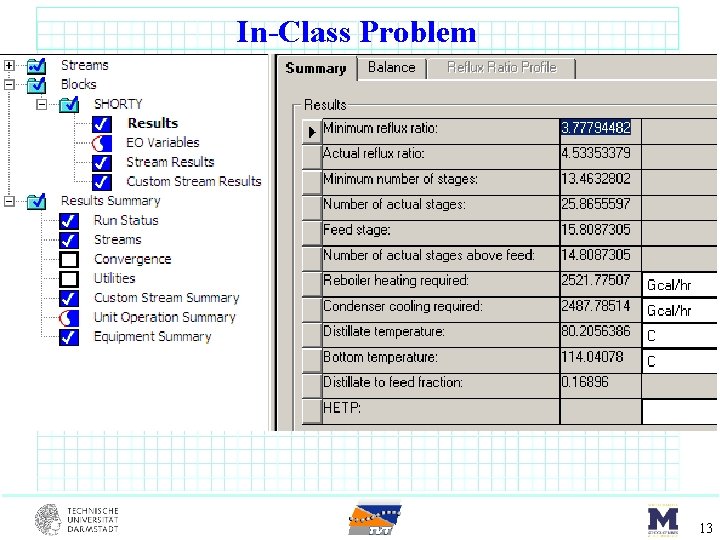
In-Class Problem 13
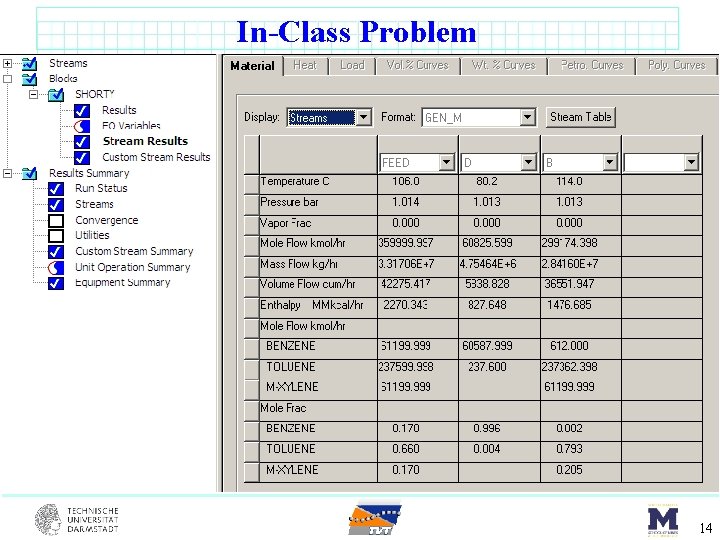
In-Class Problem 14

Homework Problem benzene (17 mol%) toluene (66 mol%) m-xylene (17 mol%) Xbz = 99 mol% DSTWU F = 100 kmol/s sat’d liquid 1 atm Component Benzene Toluene m-Xylene Xbz = 0. 1 mol% 1. 0135 bar Tbp o. C 80. 1 110. 7 139. 1 Pi star [bar] 80. 1 o. C 123 o. C 1. 0135 3. 217 0. 387 1. 42 0. 151 0. 645 15

Overview • Aspen. Plus: o Shortcut methods: DSTWU o Rigorous method: RADFRAC • Efficiencies • Introduction to multicomponent distillation (Chapter 9) 16
- Slides: 16