Chemical Compositional Analysis of Lignocellulosic Biomass 1 What

Chemical Compositional Analysis of Lignocellulosic Biomass 1
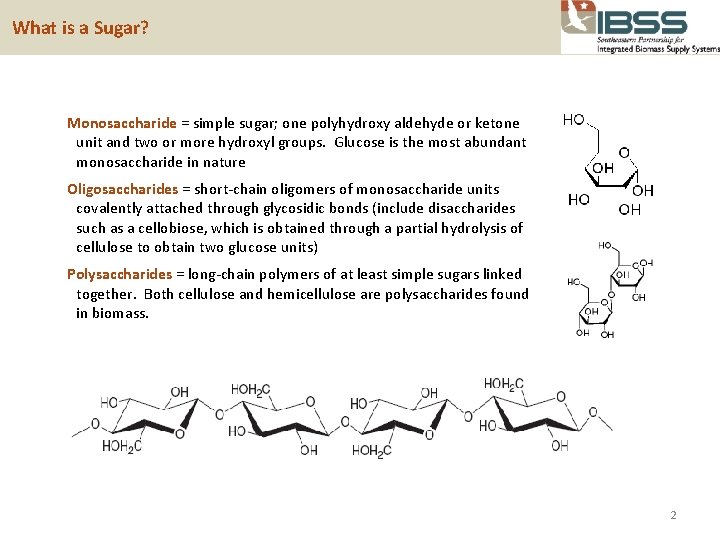
What is a Sugar? Monosaccharide = simple sugar; one polyhydroxy aldehyde or ketone unit and two or more hydroxyl groups. Glucose is the most abundant monosaccharide in nature Oligosaccharides = short-chain oligomers of monosaccharide units covalently attached through glycosidic bonds (include disaccharides such as a cellobiose, which is obtained through a partial hydrolysis of cellulose to obtain two glucose units) Polysaccharides = long-chain polymers of at least simple sugars linked together. Both cellulose and hemicellulose are polysaccharides found in biomass. 2

What is Lignin? • Lignin further acts as structural and protective element in the cell wall around the carbohydrate components, and is to some extent covalently bonded to cell wall hemicelluloses. • It’s a complex aromatic polymer consisting of phenylpropane units bonded together by ether and carbon-carbon linkages. • Little is known of the natural structure of lignin and its complex network of bonds • Difficult to measure quantitatively as the structure differs among plant species 3
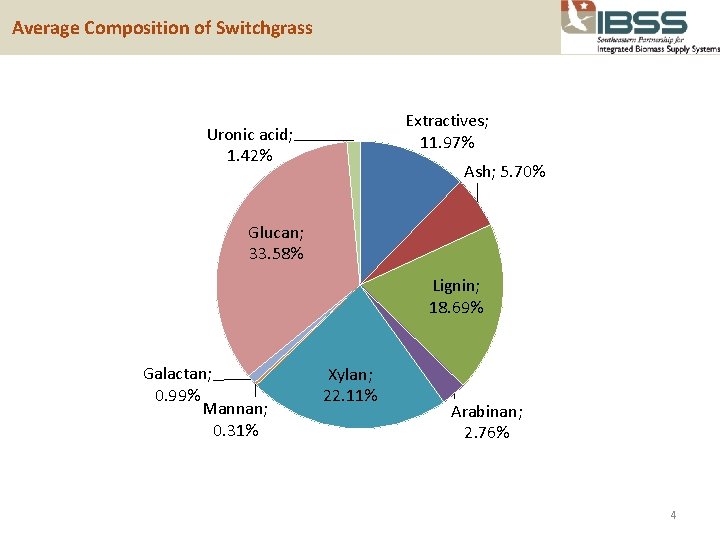
Average Composition of Switchgrass Extractives; 11. 97% Uronic acid; 1. 42% Ash; 5. 70% Glucan; 33. 58% Lignin; 18. 69% Galactan; 0. 99% Mannan; 0. 31% Xylan; 22. 11% Arabinan; 2. 76% 4
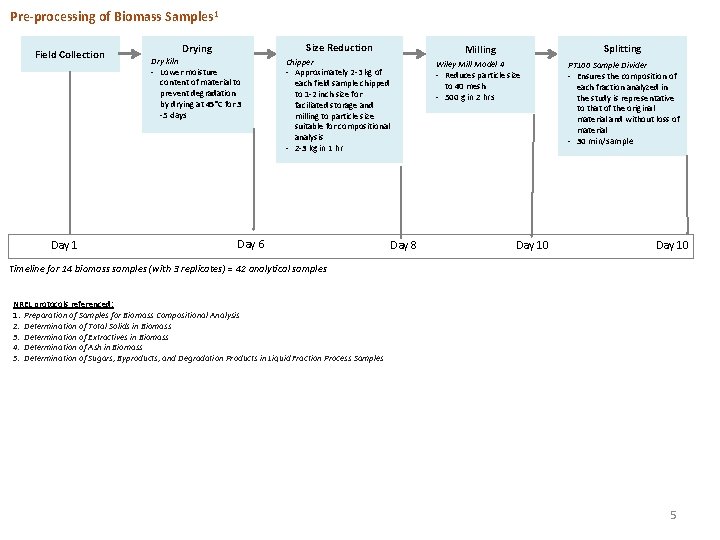
Pre-processing of Biomass Samples 1 Field Collection Day 1 Drying Size Reduction Dry kiln - Lower moisture content of material to prevent degradation by drying at 45°C for 3 -5 days Milling Splitting Chipper - Approximately 2 -3 kg of each field sample chipped to 1 -2 inch size for faciliated storage and milling to particle size suitable for compositional analysis - 2 -3 kg in 1 hr Wiley Mill Model 4 - Reduces particle size to 40 mesh - 500 g in 2 hrs PT 100 Sample Divider - Ensures the composition of each fraction analyzed in the study is representative to that of the original material and without loss of material - 30 min/sample Day 6 Day 8 Day 10 Timeline for 14 biomass samples (with 3 replicates) = 42 analytical samples NREL protocols referenced: 1. Preparation of Samples for Biomass Compositional Analysis 2. Determination of Total Solids in Biomass 3. Determination of Extractives in Biomass 4. Determination of Ash in Biomass 5. Determination of Sugars, Byproducts, and Degradation Products in Liquid Fraction Process Samples 5
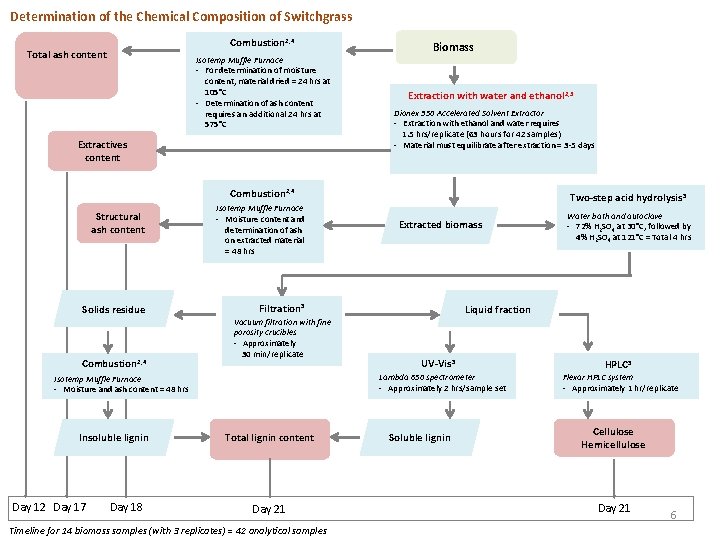
Determination of the Chemical Composition of Switchgrass Combustion 2, 4 Total ash content Isotemp Muffle Furnace - For determination of moisture content, material dried = 24 hrs at 105°C - Determination of ash content requires an additional 24 hrs at 575°C Extractives content Biomass Extraction with water and ethanol 2, 3 Dionex 350 Accelerated Solvent Extractor - Extraction with ethanol and water requires 1. 5 hrs/replicate (63 hours for 42 samples) - Material must equilibrate after extraction = 3 -5 days Combustion 2, 4 Structural ash content Solids residue Combustion 2, 4 Isotemp Muffle Furnace - Moisture content and determination of ash on extracted material = 48 hrs Day 12 Day 17 Day 18 Extracted biomass Filtration 5 Vacuum filtration with fine porosity crucibles - Approximately 30 min/replicate Total lignin content Day 21 Timeline for 14 biomass samples (with 3 replicates) = 42 analytical samples Water bath and autoclave - 72% H 2 SO 4 at 30°C, followed by 4% H 2 SO 4 at 121°C = Total 4 hrs Liquid fraction UV-Vis 5 Lambda 650 spectrometer - Approximately 2 hrs/sample set Isotemp Muffle Furnace - Moisture and ash content = 48 hrs Insoluble lignin Two-step acid hydrolysis 5 Soluble lignin HPLC 5 Flexar HPLC system - Approximately 1 hr/replicate Cellulose Hemicellulose Day 21 6
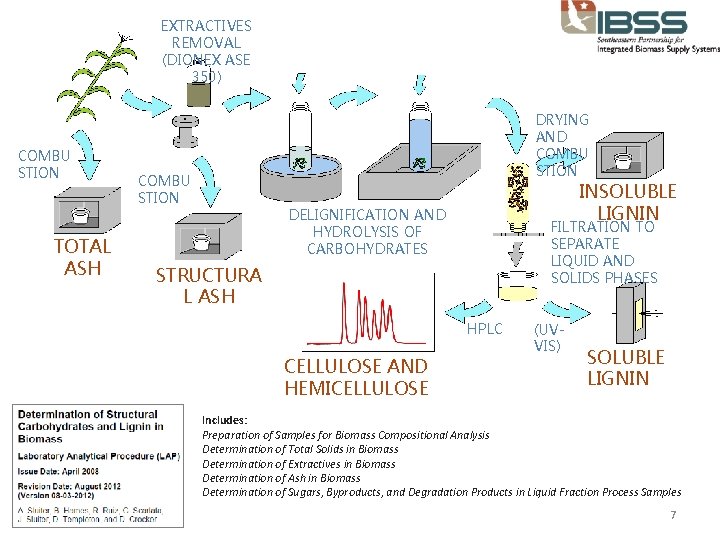
EXTRACTIVES REMOVAL (DIONEX ASE 350) COMBU STION TOTAL ASH DRYING AND COMBU STION INSOLUBLE LIGNIN DELIGNIFICATION AND HYDROLYSIS OF CARBOHYDRATES FILTRATION TO SEPARATE LIQUID AND SOLIDS PHASES STRUCTURA L ASH HPLC CELLULOSE AND HEMICELLULOSE (UVVIS) SOLUBLE LIGNIN Includes: Preparation of Samples for Biomass Compositional Analysis Determination of Total Solids in Biomass Determination of Extractives in Biomass Determination of Ash in Biomass Determination of Sugars, Byproducts, and Degradation Products in Liquid Fraction Process Samples 7

Preparation of Samples for Biomass Compositional Analysis: Physical processing/size reduction of biomass material (grinding): • Convert biomass to uniform material suitable for compositional analysis (similar to ASTM E 1757 -01) • NREL LAPs have been optimized using samples with a specific particle size range and moisture content • Too small = low bias for sugars (high lignin) due to excessive carbohydrate degradation • Too large = low bias for sugars (high lignin) due to incomplete hydrolysis of polymeric sugars to monomeric sugars • NREL LAPs optimized for ~10% moisture content • Too high = alters acid concentration in the hydrolysis steps, leading to incomplete hydrolysis of sugars. Oligomeric sugars not soluble in 4% acid and incorrectly counted as acid insoluble residue (high lignin bias) • Sieving the sample is optional “If the entire biomass sample needs to be analyzed, sieving can frequently cause fractionation and should not be performed” 8

Preparation of Samples for Biomass Compositional Analysis: Physical processing/size reduction of biomass material (grinding): 1) Lower moisture content of material through either air-drying, convection oven drying, or lyophilization • Air-drying best for large-scale sampling (>20 g) of field samples • Start with 2 x 2 inch (5 x 5 cm) pieces • Spread out on suitable surface, no deeper than 15 cm • Turn at least once a day to ensure even drying and prevent mold • Material is considered dry when MCw< 10%, and the change in weight is <1% in 24 hours • Convection oven drying best for very wet samples at risk for microbial growth • Place biomass in container at no higher than 45 ± 3°C • Looking for MCw< 10% and the change in weight is <1% in 24 hours • Higher temperatures may lead to cell wall collapse 2) Mill the dried biomass for material -20/+80 mesh • Monitor temperature to prevent damage to biomass, rest if necessary • Sample should be stored in sealable polyethylene bag and kept at -20°C 9

Determination of Total Solids (Moisture Content): Total solids = Amount of solids remaining after heating the sample at 105°C to constant weight Moisture content = Amount of water (and other components volatilized at 105°C) present in biomass sample 1) Dry crucible at 105°C for a minimum of 4 hours, place in desiccator and cool to room temperature 2) Using gloves, weigh pan 3) Weigh 0. 5 -2 g (more is better) of biomass material to nearest 0. 1 mg 4) Dry at 105 ± 3°C for a minimum of 4 hours, cool in desiccator 5) Dry to constant weight if needed (<1% change in %TS after drying for 1 hour) 10

Determination of Total Solids (Moisture Content): Two measurements for moisture content in biomass: 1) Moisture Content on Oven-dry Basis (MCd) • More commonly used in wood industry, relates fiber saturation point, shrinkage, etc. • MCd can exceed 100% 2) Moisture Content on Wet or Original Basis (MCw) • More commonly used in pulp and paper, wood energy industries, NREL protocols • MCw can never reach 100% Convert between the two: All values in the compositional analysis are performed on a dry-basis (% of the dried sample), so you must correct for it prior to each analytical step Oven Dry Weight (ODW) = the weight of biomass mathematically corrected for the amount of moisture present in the sample at the time of weighing. Example: For hydrolysis , a 0. 3 g switchgrass sample with MCw=7% would have an ODW = 0. 279 g 11

Determination of Ash: 1) 2) 3) 4) 5) 6) 7) 8) Clean crucibles at 575°C for a minimum of 4 hours, cool for specific period of time (1 hr recommended) Dry to constant weight is < 0. 3 mg change after 1 hour of reheating sample Use 0. 5 -2 g (more is better) Perform “Determination of Total Solids in Biomass” at the same time Pre-ignition, followed by combustion at 575 ± 25°C for 24 ± 6 hrs Remove sample directly to desiccator and cool for time equal to initial cool time of the crucibles. *Sample will easily blow away with drafts. Ash to constant weight Analyze in duplicate, at minimum 12
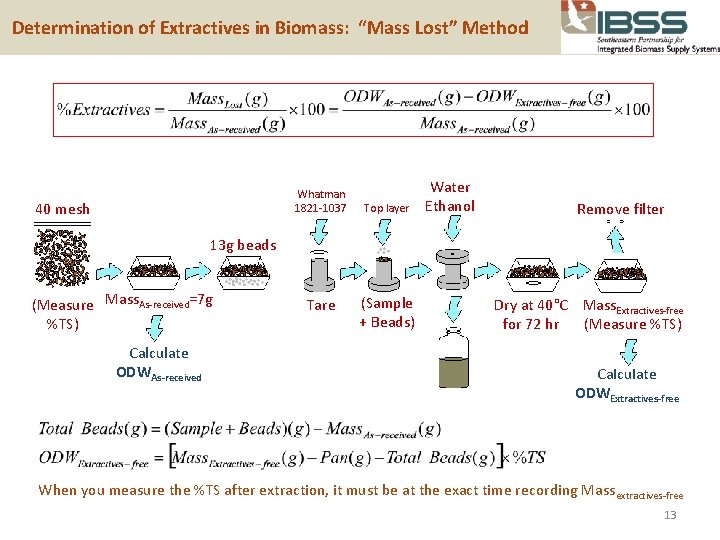
Determination of Extractives in Biomass: “Mass Lost” Method Whatman 1821 -1037 40 mesh Top layer Water Ethanol Remove filter 13 g beads (Measure Mass. As-received=7 g %TS) Calculate ODWAs-received Tare (Sample + Beads) Dry at 40°C Mass. Extractives-free for 72 hr (Measure %TS) Calculate ODWExtractives-free When you measure the %TS after extraction, it must be at the exact time recording Mass extractives-free 13
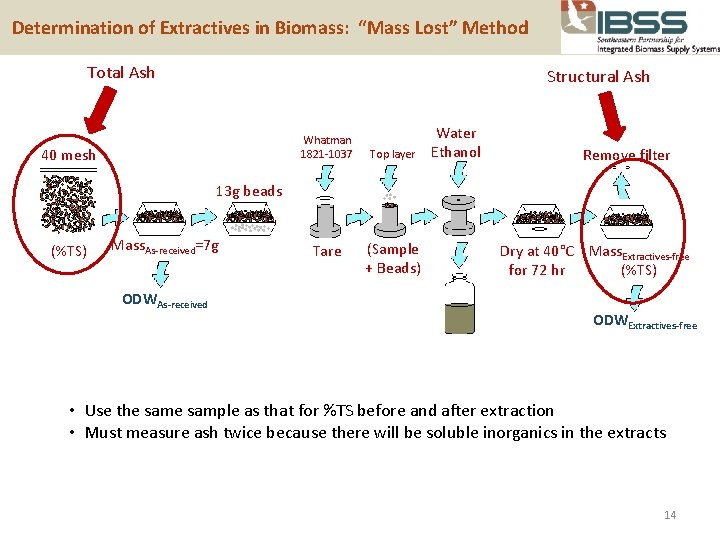
Determination of Extractives in Biomass: “Mass Lost” Method Total Ash Structural Ash Whatman 1821 -1037 40 mesh Top layer Water Ethanol Remove filter 13 g beads Mass =7 g (%TS) Total Ash As-received Structural Ash ODWAs-received Tare (Sample + Beads) Dry at 40°C Mass. Extractives-free (%TS) for 72 hr ODWExtractives-free • Use the sample as that for %TS before and after extraction • Must measure ash twice because there will be soluble inorganics in the extracts 14
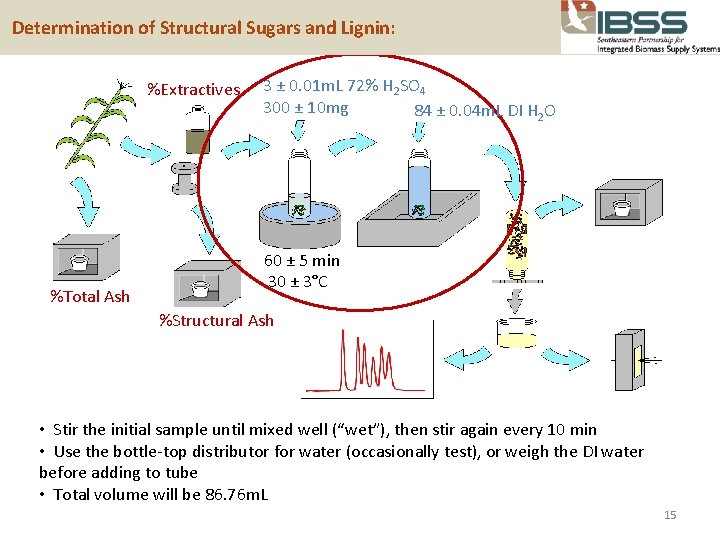
Determination of Structural Sugars and Lignin: %Extractives %Total Ash 3 ± 0. 01 m. L 72% H 2 SO 4 300 ± 10 mg 84 ± 0. 04 m. L DI H 2 O 60 ± 5 min 30 ± 3°C %Structural Ash • Stir the initial sample until mixed well (“wet”), then stir again every 10 min • Use the bottle-top distributor for water (occasionally test), or weigh the DI water before adding to tube • Total volume will be 86. 76 m. L 15

Determination of Structural Sugars and Lignin: Two-step hydrolysis • First step (high acid, low temp) breaks down hemicellulose and starts removal of lignin from the carbohydrates • Second step (low acid, high temp) finishes breaking down all the oligomers to monosugars • Two checks needed for the efficiency of the hydrolysis: Presence of cellobiose: • If cellobiose is present in the hydrolyzate, the conditions were “too little” and there was an incomplete hydrolysis of cellulose (look for [cellobiose]>3 mg/m. L) Sugar Recovery Standard (SRS): • Quality check that it is treated to the same conditions as the real samples • Known amount of monosugars added to 4% acid • Test for the %recovery of these monosugars • If hydrolysis is “too much”, SRS correction factor accounts for losses due to deconstruction of sugars during the dilute acid hydrolysis 16
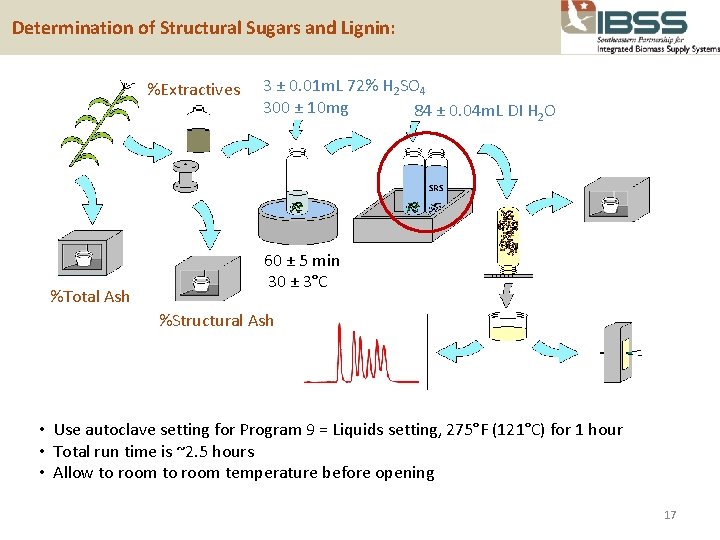
Determination of Structural Sugars and Lignin: %Extractives 3 ± 0. 01 m. L 72% H 2 SO 4 300 ± 10 mg 84 ± 0. 04 m. L DI H 2 O SRS %Total Ash 60 ± 5 min 30 ± 3°C %Structural Ash • Use autoclave setting for Program 9 = Liquids setting, 275°F (121°C) for 1 hour • Total run time is ~2. 5 hours • Allow to room temperature before opening 17
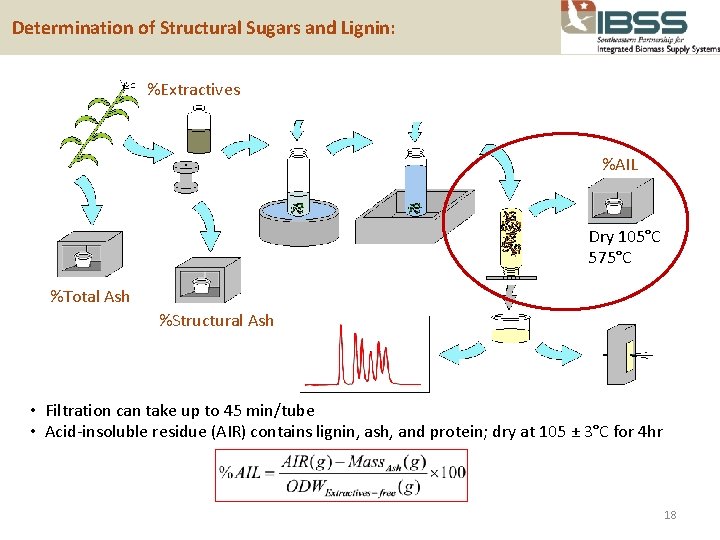
Determination of Structural Sugars and Lignin: %Extractives %AIL Dry 105°C 575°C %Total Ash %Structural Ash • Filtration can take up to 45 min/tube • Acid-insoluble residue (AIR) contains lignin, ash, and protein; dry at 105 ± 3°C for 4 hr 18
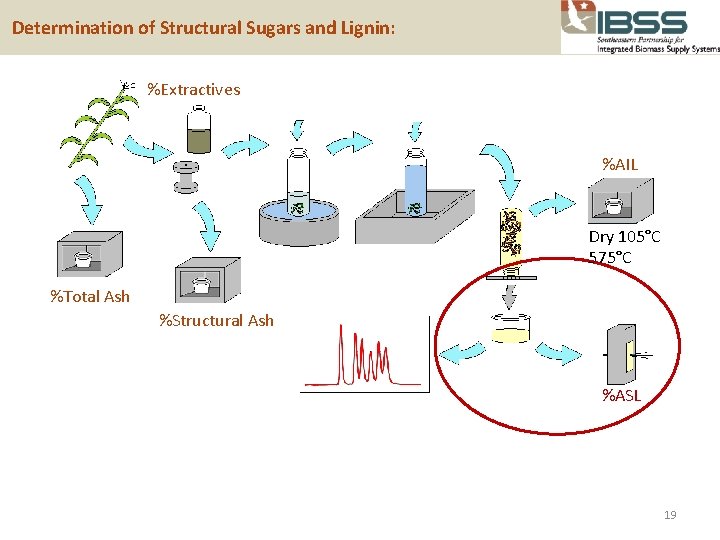
Determination of Structural Sugars and Lignin: %Extractives %AIL Dry 105°C 575°C %Total Ash %Structural Ash %ASL 19

Determination of Structural Sugars and Lignin: Beer’s Law: Abs Sample Concentration Incident Light, Io [X] Transmission light, I E(L/g-cm) = slope [X] (g/L) Optical pathlength, l = 1 cm Y=mx+b Abs = E x [X] x l Transmittance, T = I/Io %T = I/Io x 100 Abs = log(1/T) = log(Io/I) 20
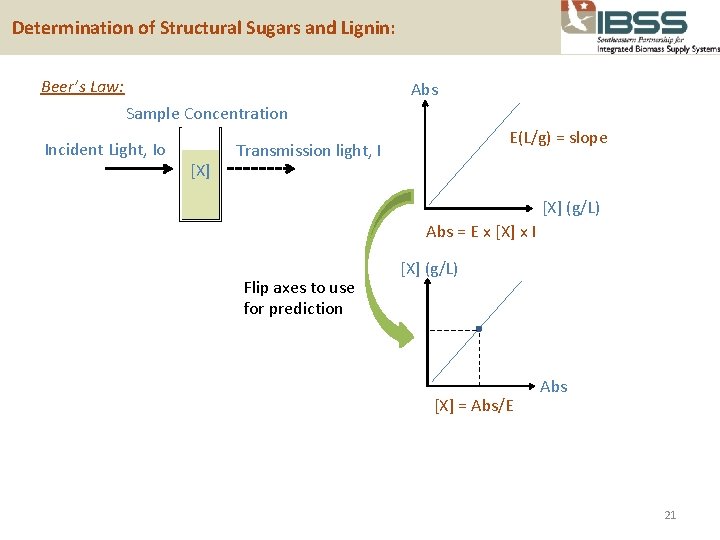
Determination of Structural Sugars and Lignin: Beer’s Law: Abs Sample Concentration Incident Light, Io [X] E(L/g) = slope Transmission light, I [X] (g/L) Abs = E x [X] x l Flip axes to use for prediction [X] (g/L) [X] = Abs/E Abs 21
![Determination of Structural Sugars and Lignin: [X] (g/L) = Abs/E Abs *Dilution of Determination of Structural Sugars and Lignin: [X] (g/L) = Abs/E Abs *Dilution of](http://slidetodoc.com/presentation_image/326731f05f3f3d2da362339fa9976538/image-22.jpg)
Determination of Structural Sugars and Lignin: [X] (g/L) = Abs/E Abs *Dilution of 50μL in 700μL good place to start 22
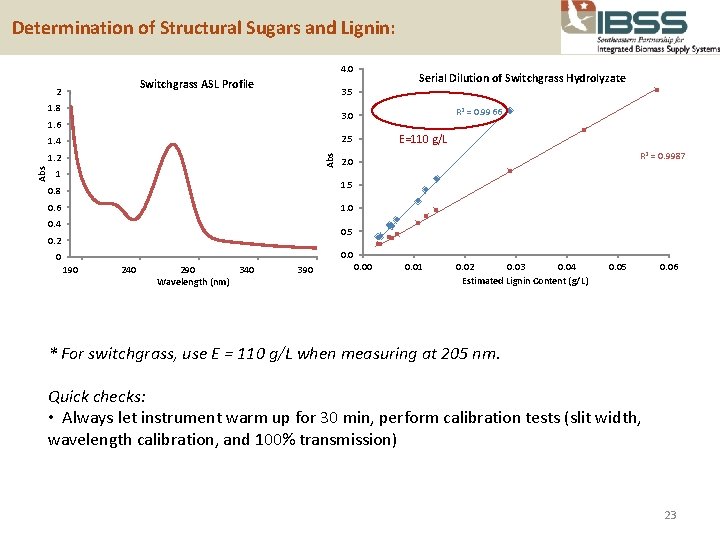
Determination of Structural Sugars and Lignin: 4. 0 Switchgrass ASL Profile 2 1. 8 R 2 = 0. 9966 3. 0 1. 6 E=110 g/L 2. 5 1. 4 Abs 1. 2 Abs Serial Dilution of Switchgrass Hydrolyzate 3. 5 1 R 2 = 0. 9987 2. 0 1. 5 0. 8 0. 6 1. 0 0. 4 0. 5 0. 2 0. 0 0 190 240 290 340 Wavelength (nm) 390 0. 01 0. 02 0. 03 0. 04 Estimated Lignin Content (g/L) 0. 05 0. 06 * For switchgrass, use E = 110 g/L when measuring at 205 nm. Quick checks: • Always let instrument warm up for 30 min, perform calibration tests (slit width, wavelength calibration, and 100% transmission) 23
Determination of Structural Sugars and Lignin: %Extractives %AIL %Total Ash %Structural Ash %Sugars %ASL 24
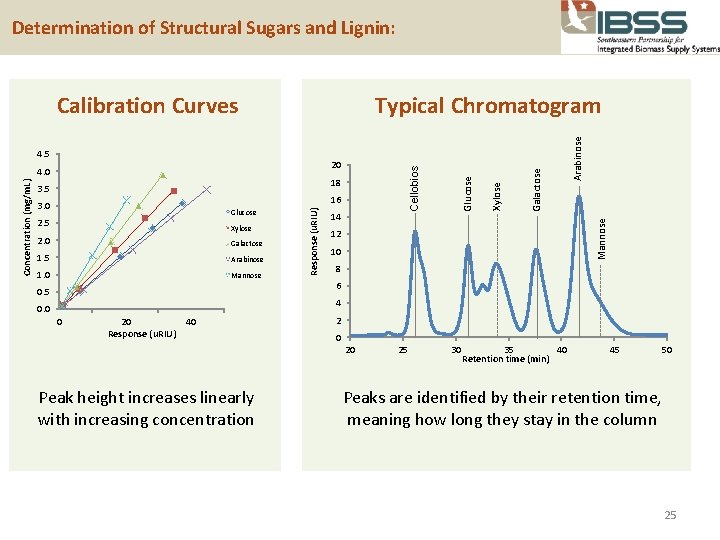
Determination of Structural Sugars and Lignin: 16 Glucose 2. 5 Xylose 2. 0 Galactose 1. 5 Arabinose 1. 0 Mannose 0. 5 14 Mannose 3. 0 Arabinose 18 3. 5 Response (u. RIU) Concentration (mg/m. L) Cellobios 20 4. 0 Galactose 4. 5 Xylose Typical Chromatogram Glucose Calibration Curves 12 10 8 6 4 0. 0 0 20 Response (u. RIU) 40 2 0 20 Peak height increases linearly with increasing concentration 25 30 35 40 Retention time (min) 45 50 Peaks are identified by their retention time, meaning how long they stay in the column 25

Determination of Structural Sugars and Lignin: 3) Anhydro correction – necessary to correction for the addition of the water molecule during the hydrolysis of cellulose and hemicellulose to monosugars Known molar mass: Cellulose = 162 g/mol Glucose = 180 g/mol Water = 18 g/mol 1 mol Cellulose = 162 g Hydrolysis Addition of water molecule (18 g/mol) 1 mol Glucose = 180 g Anhydro correction = 162/180 = 0. 9 Correction factors: • Xylose, arabinose = 0. 88 • Glucose, galactose, mannose = 0. 9 26

Determination of Structural Sugars and Lignin: Think of your biomass and its components as pieces of a pie. You start with biomass with an ODW = 5 g. You present the data on a %dry basis, so calculations are completed with the ODW (oven dry weight) of the material at 105°C. Compositional analysis is completed on an extractives-free basis, so this part is missing from the pie. Extractives = 0. 5 g Ash Add the extractives back into the pie to calculate the total composition on an as-received basis: Ash Extractives Hemicellulose Lignin = 1 g Cellulose Total mass = 5 g. You remove the dried extracts, so the extractives-free biomass = 4. 5 g 22. 2% Now the ODW of the biomass is 5 g. 20. 0% You can calculate the composition on an as-received basis if you know the extractives content with the following calculation: 27
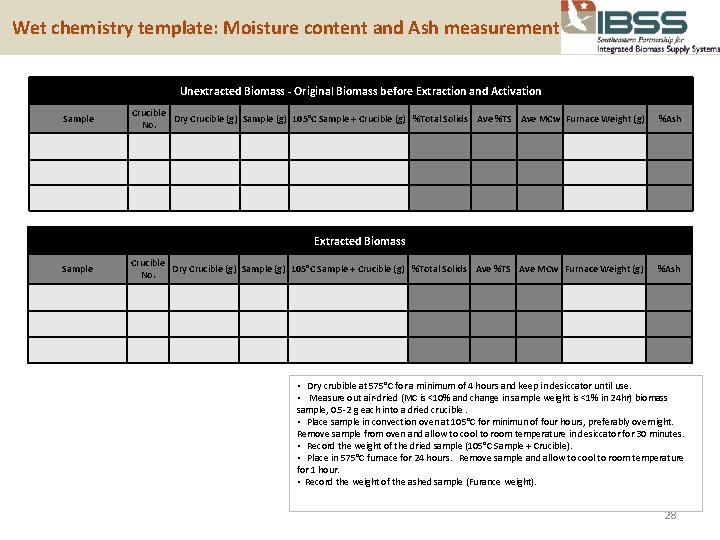
Wet chemistry template: Moisture content and Ash measurement Unextracted Biomass - Original Biomass before Extraction and Activation Sample Crucible Dry Crucible (g) Sample (g) 105°C Sample + Crucible (g) %Total Solids Ave %TS Ave MCw Furnace Weight (g) No. %Ash Extracted Biomass Sample Crucible Dry Crucible (g) Sample (g) 105°C Sample + Crucible (g) %Total Solids Ave %TS Ave MCw Furnace Weight (g) No. %Ash • Dry crubible at 575°C for a minimum of 4 hours and keep in desiccator until use. • Measure out air-dried (MC is <10% and change in sample weight is <1% in 24 hr) biomass sample, 0. 5 -2 g each into a dried crucible. • Place sample in convection oven at 105°C for minimun of four hours, preferably overnight. Remove sample from oven and allow to cool to room temperature in desiccator for 30 minutes. • Record the weight of the dried sample (105°C Sample + Crucible). • Place in 575°C furnace for 24 hours. Remove sample and allow to cool to room temperature for 1 hour. • Record the weight of the ashed sample (Furance weight). 28
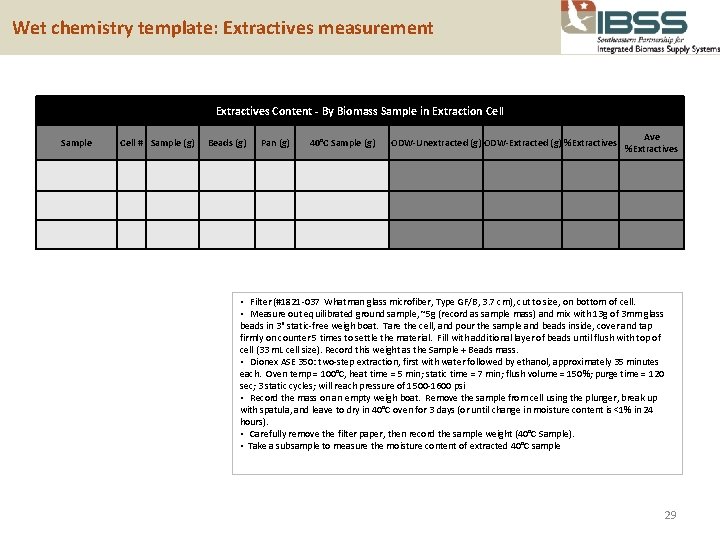
Wet chemistry template: Extractives measurement Extractives Content - By Biomass Sample in Extraction Cell Sample Cell # Sample (g) Beads (g) Pan (g) 40°C Sample (g) ODW-Unextracted (g) ODW-Extracted (g) %Extractives Ave %Extractives • Filter (#1821 -037 Whatman glass microfiber, Type GF/B, 3. 7 cm), cut to size, on bottom of cell. • Measure out equilibrated ground sample, ~5 g (record as sample mass) and mix with 13 g of 3 mm glass beads in 3" static-free weigh boat. Tare the cell, and pour the sample and beads inside, cover and tap firmly on counter 5 times to settle the material. Fill with additional layer of beads until flush with top of cell (33 m. L cell size). Record this weight as the Sample + Beads mass. • Dionex ASE 350: two-step extraction, first with water followed by ethanol, approximately 35 minutes each. Oven temp = 100°C, heat time = 5 min; static time = 7 min; flush volume = 150%; purge time = 120 sec; 3 static cycles; will reach pressure of 1500 -1600 psi • Record the mass on an empty weigh boat. Remove the sample from cell using the plunger, break up with spatula, and leave to dry in 40°C oven for 3 days (or until change in moisture content is <1% in 24 hours). • Carefully remove the filter paper, then record the sample weight (40°C Sample). • Take a subsample to measure the moisture content of extracted 40°C sample 29
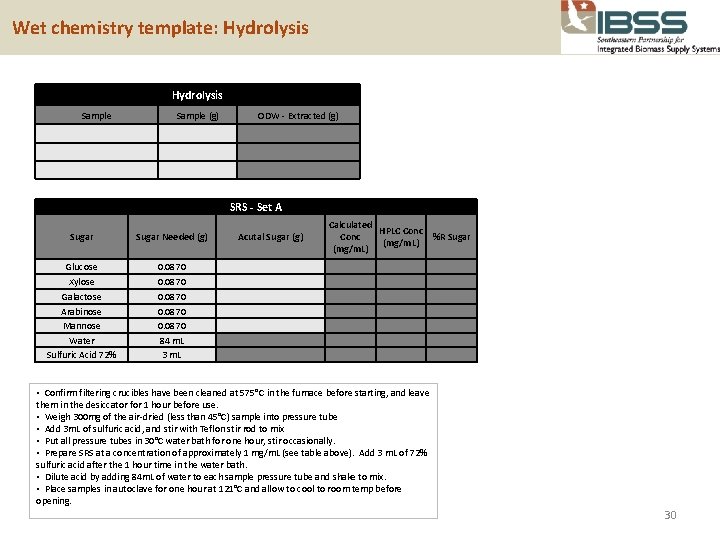
Wet chemistry template: Hydrolysis Sample (g) ODW - Extracted (g) SRS - Set A Sugar Needed (g) Acutal Sugar (g) Glucose 0. 0870 Xylose Galactose Arabinose Mannose Water Sulfuric Acid 72% 0. 0870 84 m. L 3 m. L Calculated HPLC Conc %R Sugar (mg/m. L) • Confirm filtering crucibles have been cleaned at 575°C in the furnace before starting, and leave them in the desiccator for 1 hour before use. • Weigh 300 mg of the air-dried (less than 45°C) sample into pressure tube • Add 3 m. L of sulfuric acid, and stir with Teflon stir rod to mix • Put all pressure tubes in 30°C water bath for one hour, stir occasionally. • Prepare SRS at a concentration of approximately 1 mg/m. L (see table above). Add 3 m. L of 72% sulfuric acid after the 1 hour time in the water bath. • Dilute acid by adding 84 m. L of water to each sample pressure tube and shake to mix. • Place samples in autoclave for one hour at 121°C and allow to cool to room temp before opening. 30
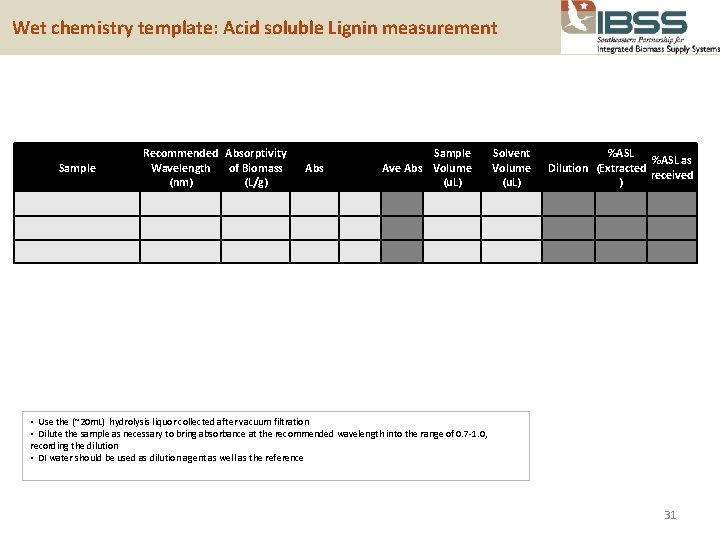
Wet chemistry template: Acid soluble Lignin measurement Sample Recommended Absorptivity Wavelength of Biomass (nm) (L/g) Abs Sample Ave Abs Volume (u. L) Solvent Volume (u. L) %ASL as Dilution (Extracted received ) • Use the (~20 m. L) hydrolysis liquor collected after vacuum filtration • Dilute the sample as necessary to bring absorbance at the recommended wavelength into the range of 0. 7 -1. 0, recording the dilution • DI water should be used as dilution agent as well as the reference 31
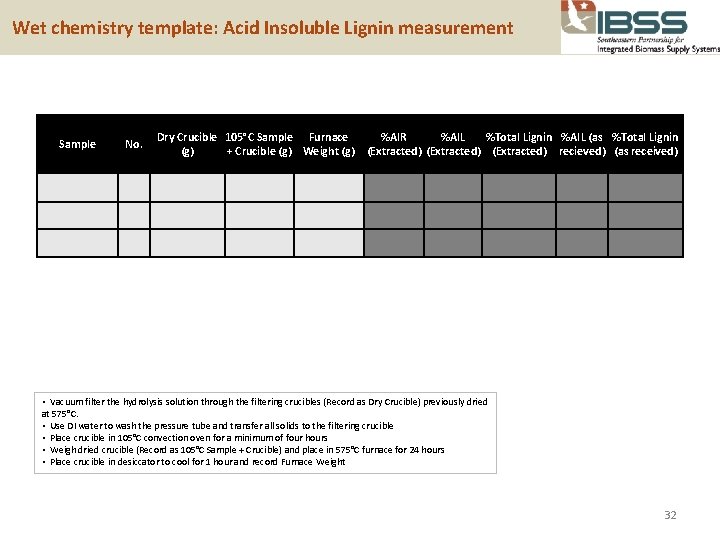
Wet chemistry template: Acid Insoluble Lignin measurement Sample No. Dry Crucible 105°C Sample Furnace (g) + Crucible (g) Weight (g) %AIR %AIL %Total Lignin %AIL (as %Total Lignin (Extracted) recieved) (as received) • Vacuum filter the hydrolysis solution through the filtering crucibles (Record as Dry Crucible) previously dried at 575°C. • Use DI water to wash the pressure tube and transfer all solids to the filtering crucible • Place crucible in 105°C convection oven for a minimum of four hours • Weigh dried crucible (Record as 105°C Sample + Crucible) and place in 575°C furnace for 24 hours • Place crucible in desiccator to cool for 1 hour and record Furnace Weight 32
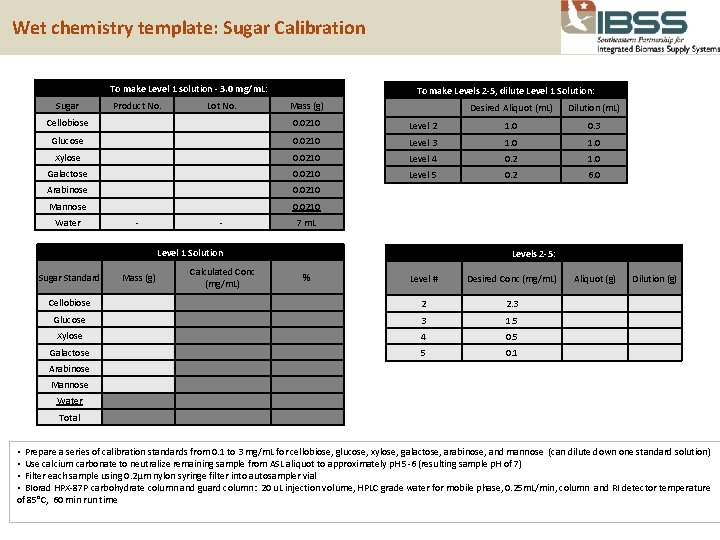
Wet chemistry template: Sugar Calibration To make Level 1 solution - 3. 0 mg/m. L: To make Levels 2 -5, dilute Level 1 Solution: Sugar Product No. Lot No. Mass (g) Desired Aliquot (m. L) Dilution (m. L) Cellobiose 0. 0210 Level 2 1. 0 0. 3 Glucose 0. 0210 Level 3 1. 0 Xylose 0. 0210 Level 4 0. 2 1. 0 Galactose 0. 0210 Level 5 0. 2 6. 0 Arabinose 0. 0210 Mannose 0. 0210 Water - - 7 m. L Level 1 Solution Sugar Standard Mass (g) Cellobiose Calculated Conc (mg/m. L) Levels 2 -5: % Level # Desired Conc (mg/m. L) Aliquot (g) Dilution (g) 2 2. 3 Glucose 3 1. 5 Xylose 4 0. 5 Galactose 5 0. 1 Arabinose Mannose Water Total • Prepare a series of calibration standards from 0. 1 to 3 mg/m. L for cellobiose, glucose, xylose, galactose, arabinose, and mannose (can dilute down one standard solution) • Use calcium carbonate to neutralize remaining sample from ASL aliquot to approximately p. H 5 -6 (resulting sample p. H of 7) • Filter each sample using 0. 2μm nylon syringe filter into autosampler vial • BIorad HPX-87 P carbohydrate column and guard column: 20 u. L injection volume, HPLC grade water for mobile phase, 0. 25 m. L/min, column and RI detector temperature of 85°C, 60 min run time 33
- Slides: 33