Chemical Change Chapter 2 Dr Suzan A Khayyat






- Slides: 50

Chemical Change Chapter 2 Dr. Suzan A. Khayyat 1

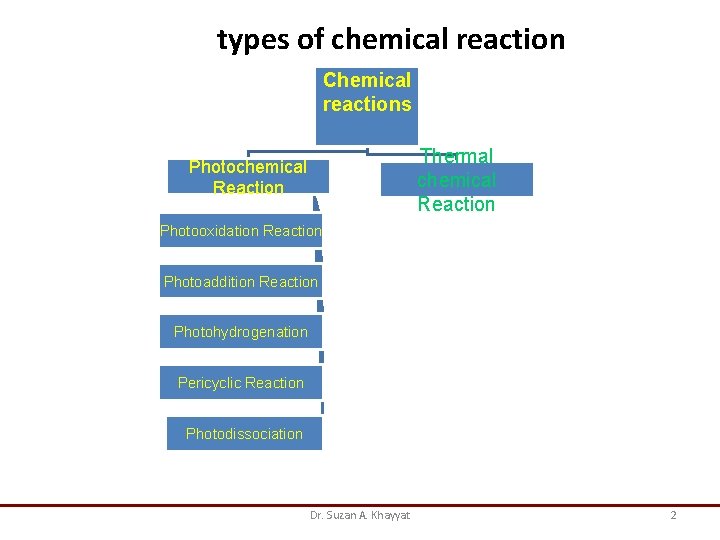
types of chemical reaction Chemical reactions Thermal chemical Reaction Photooxidation Reaction Photoaddition Reaction Photohydrogenation Pericyclic Reaction Photodissociation Dr. Suzan A. Khayyat 2

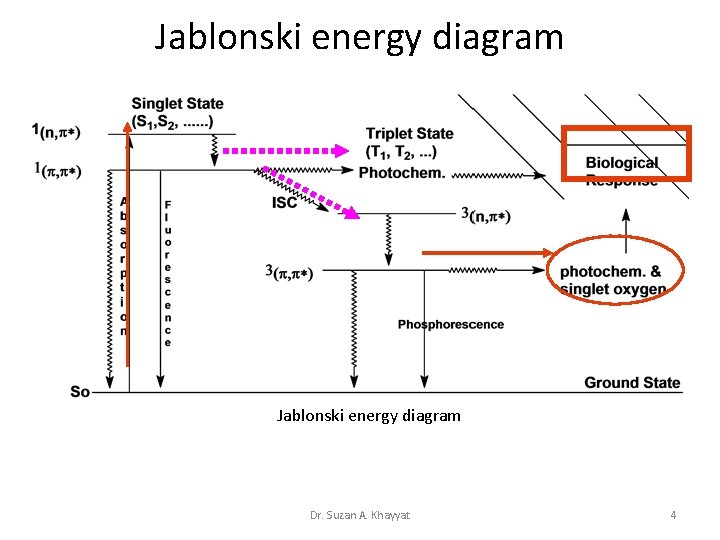
• The Jablonski Diagram • The energy gained by a molecule when it absorbs a photon causes an electron to be promoted to a higher electronic energy level. Figure 3 illustrates the principal photophysical radiative and non-radiative processes displayed by organic molecules in solution. The symbols So, S 1, T 2, etc. , refer to the ground electronic state (So), first excited singlet state (S 1), second excited triplet state (T 2), and so on. The horizontal lines represent the vibrational levels of each electronic state. Straight arrows indicate radiative transitions, and curly arrows indicate nonradiative transitions. The boxes detail the electronic spins in each orbital, with electrons shown as up and down arrows, to distinguish their spin. • Note that all transitions from one electronic state to another originate from the lowest vibrational level of the initial electronic state. For example, fluorescence occurs only from S 1, because the higher singlet states (S 2, etc. ) decay so rapidly by internal conversion that fluorescence from these states cannot compete. Dr. Suzan A. Khayyat 3

Jablonski energy diagram Dr. Suzan A. Khayyat 4

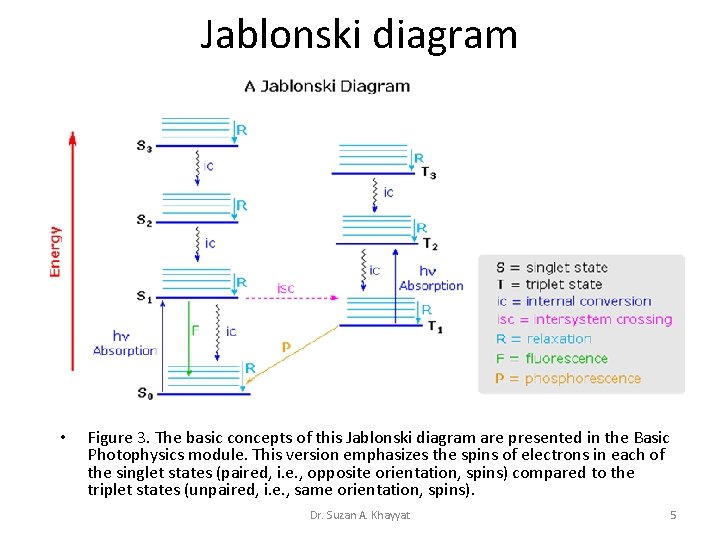
Jablonski diagram • Figure 3. The basic concepts of this Jablonski diagram are presented in the Basic Photophysics module. This version emphasizes the spins of electrons in each of the singlet states (paired, i. e. , opposite orientation, spins) compared to the triplet states (unpaired, i. e. , same orientation, spins). Dr. Suzan A. Khayyat 5

Photooxygenation Reaction Dr. Suzan A. Khayyat 6

(1 O 2 ) Dr. Suzan A. Khayyat 7

Dr. Suzan A. Khayyat 8

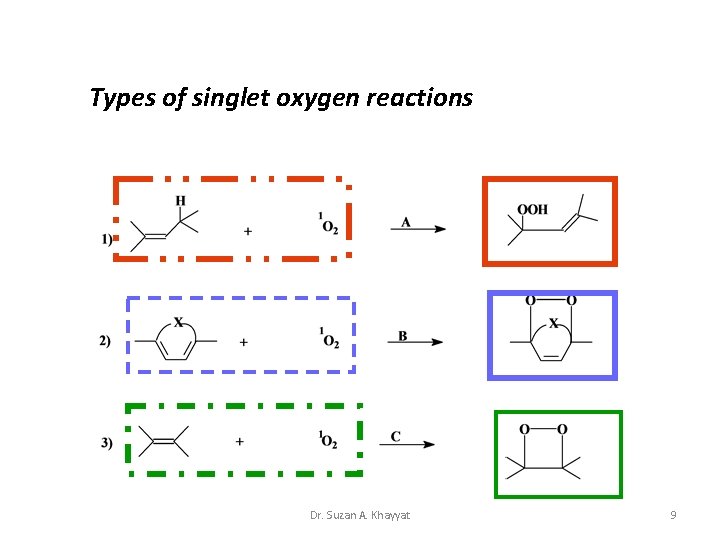
Types of singlet oxygen reactions Dr. Suzan A. Khayyat 9

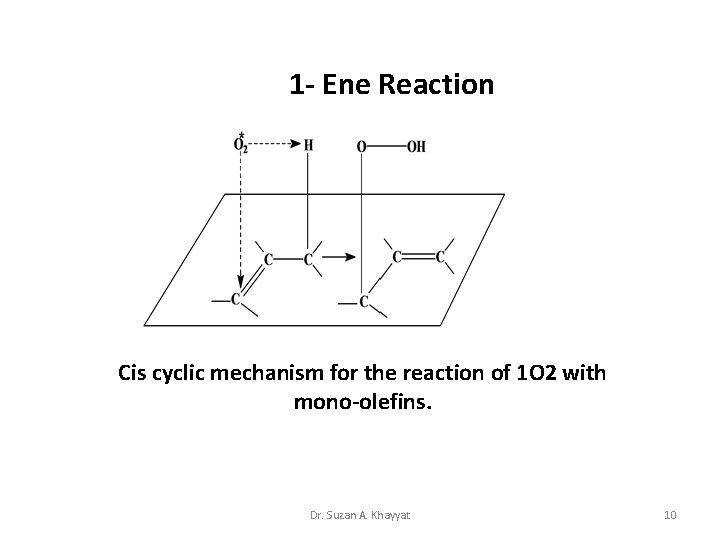
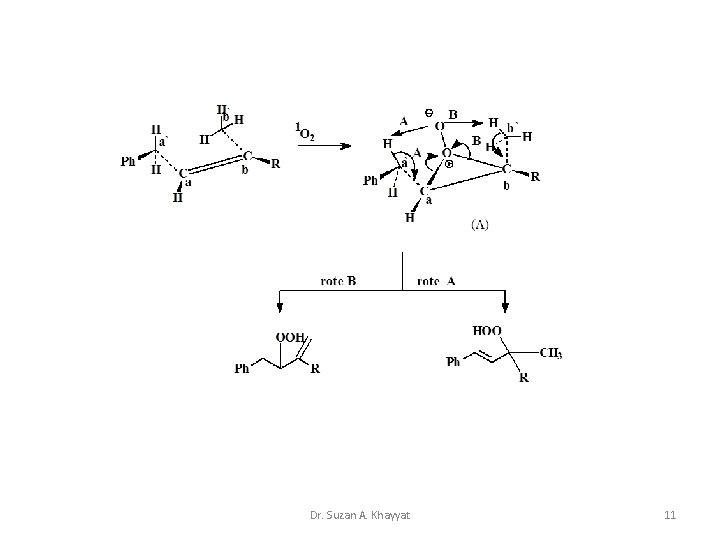
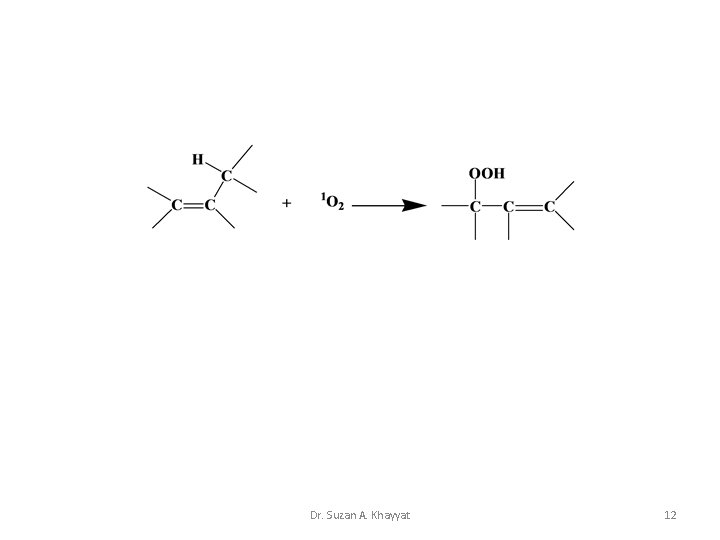
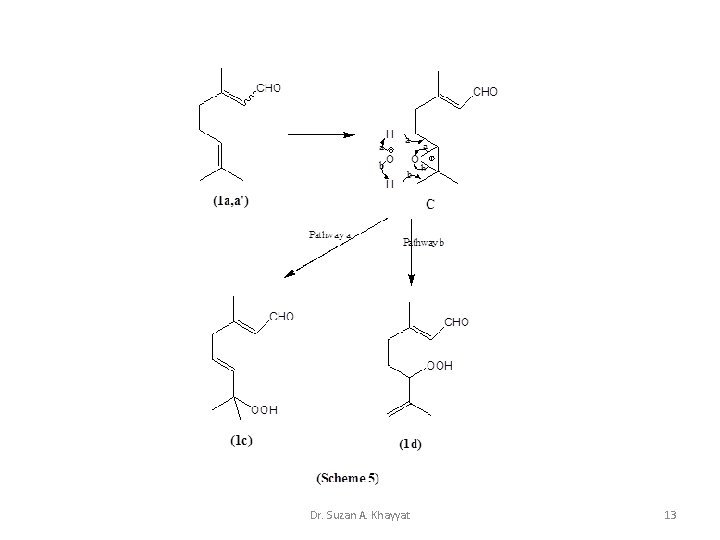
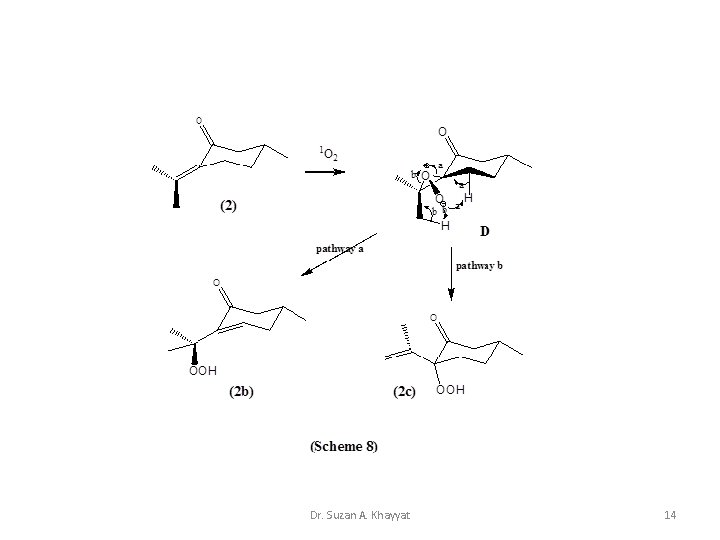
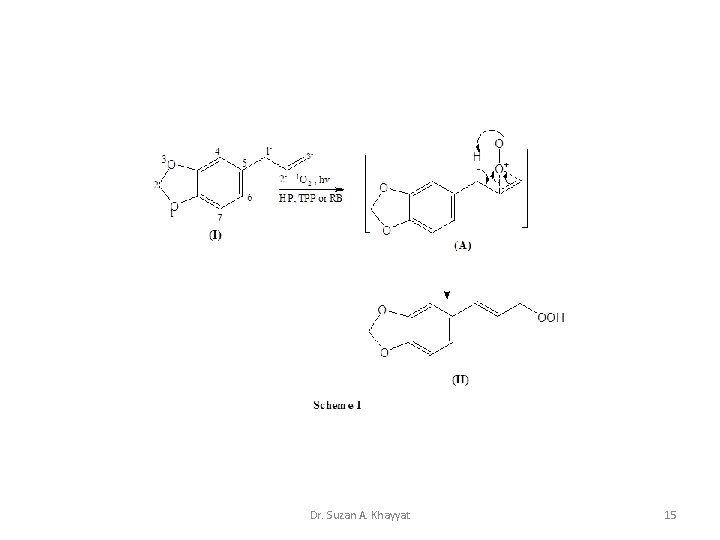
1 - Ene Reaction Cis cyclic mechanism for the reaction of 1 O 2 with mono-olefins. Dr. Suzan A. Khayyat 10

Dr. Suzan A. Khayyat 11

Dr. Suzan A. Khayyat 12

Dr. Suzan A. Khayyat 13

Dr. Suzan A. Khayyat 14

Dr. Suzan A. Khayyat 15

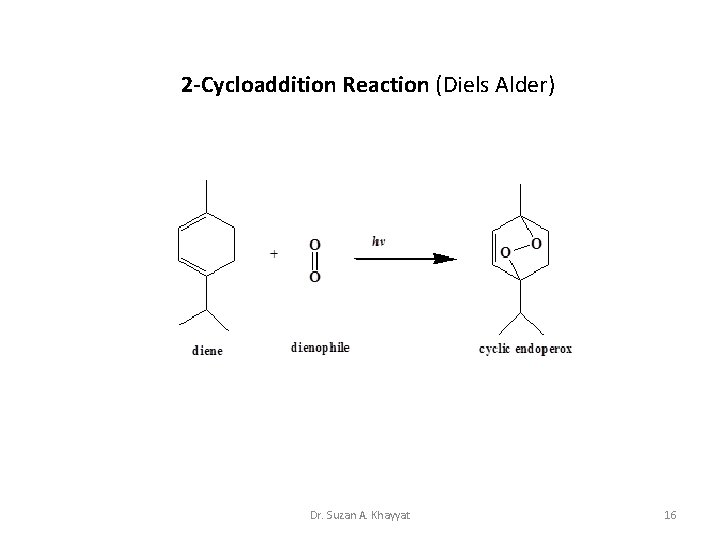
2 -Cycloaddition Reaction (Diels Alder) Dr. Suzan A. Khayyat 16

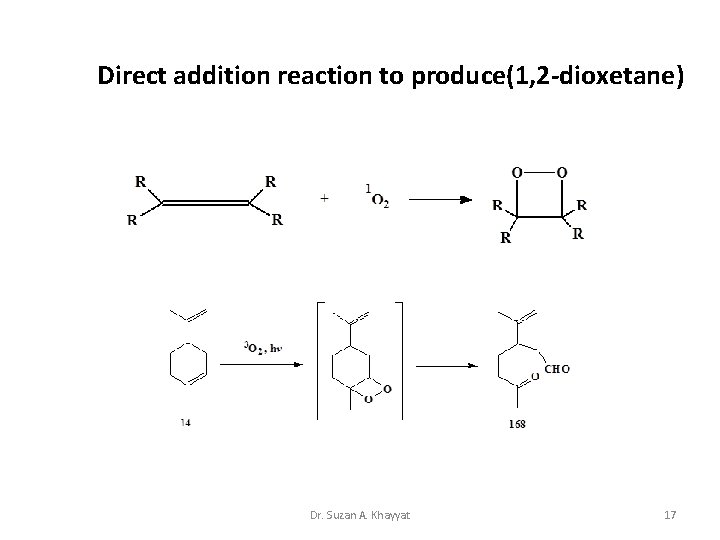
Direct addition reaction to produce(1, 2 -dioxetane) Dr. Suzan A. Khayyat 17

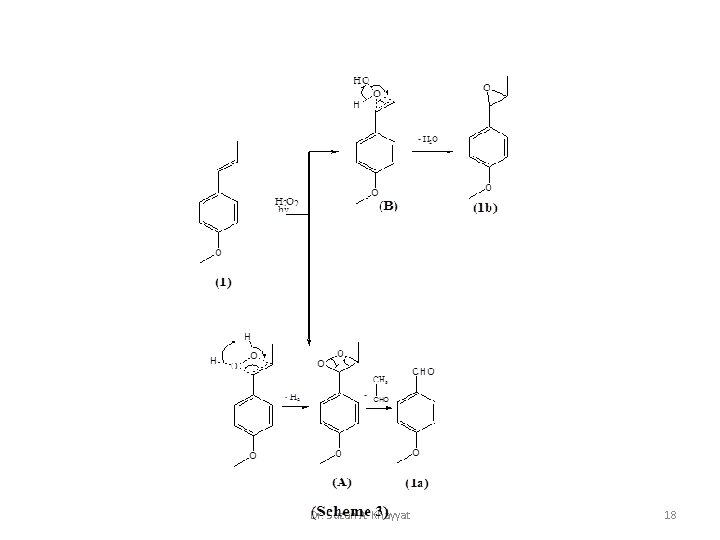
Dr. Suzan A. Khayyat 18

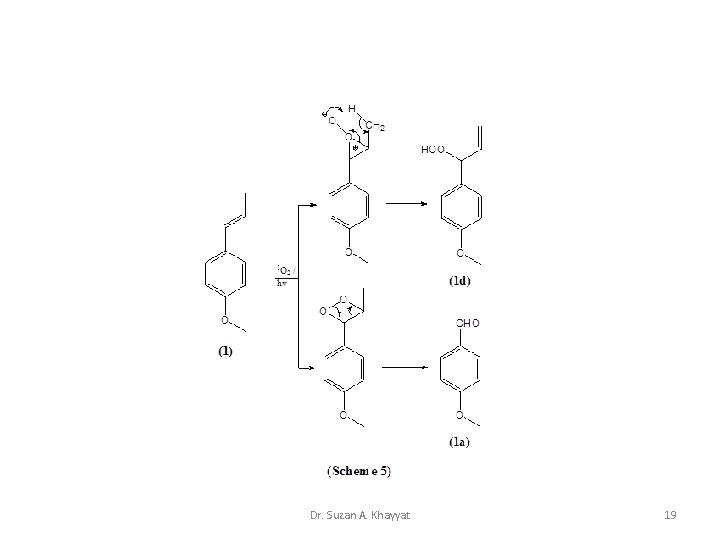
Dr. Suzan A. Khayyat 19

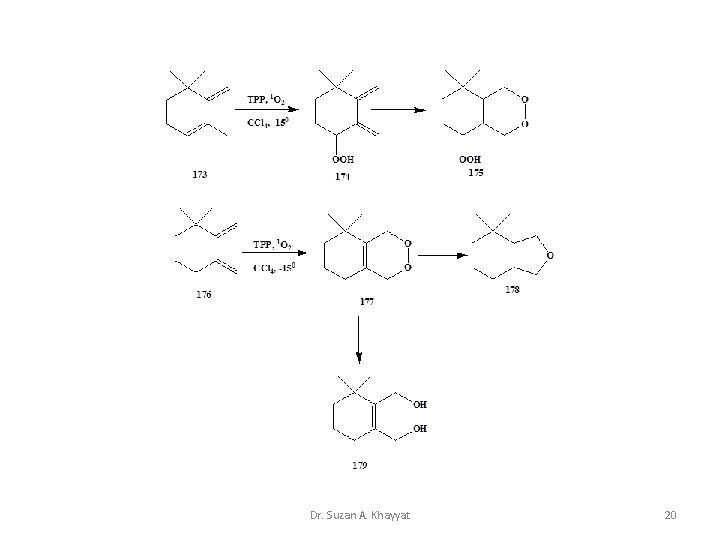
Dr. Suzan A. Khayyat 20

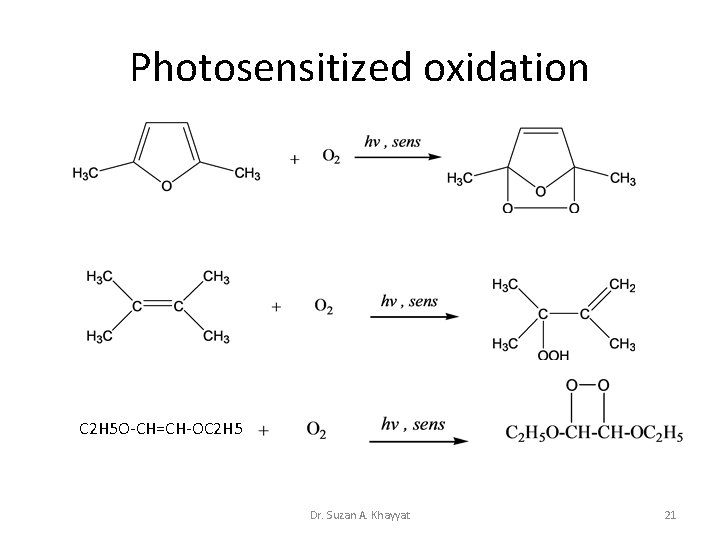
Photosensitized oxidation C 2 H 5 O-CH=CH-OC 2 H 5 Dr. Suzan A. Khayyat 21

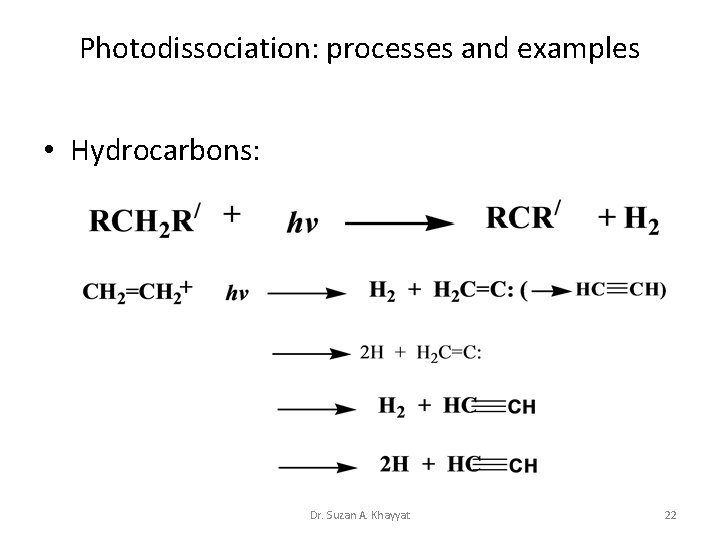
Photodissociation: processes and examples • Hydrocarbons: Dr. Suzan A. Khayyat 22

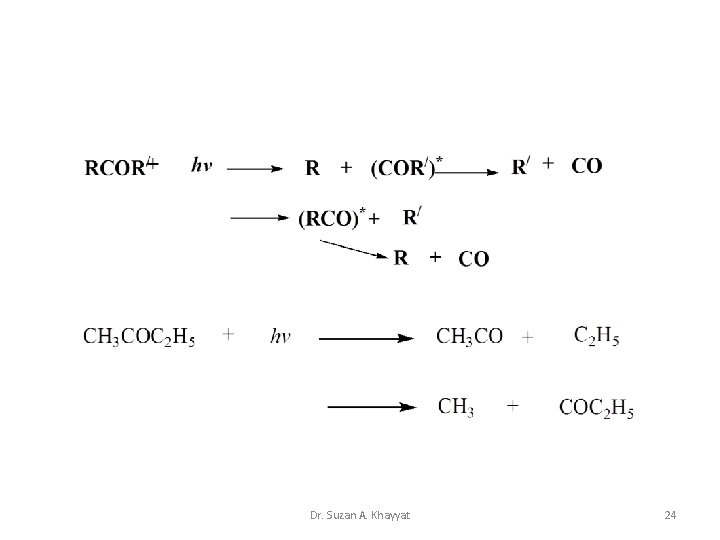
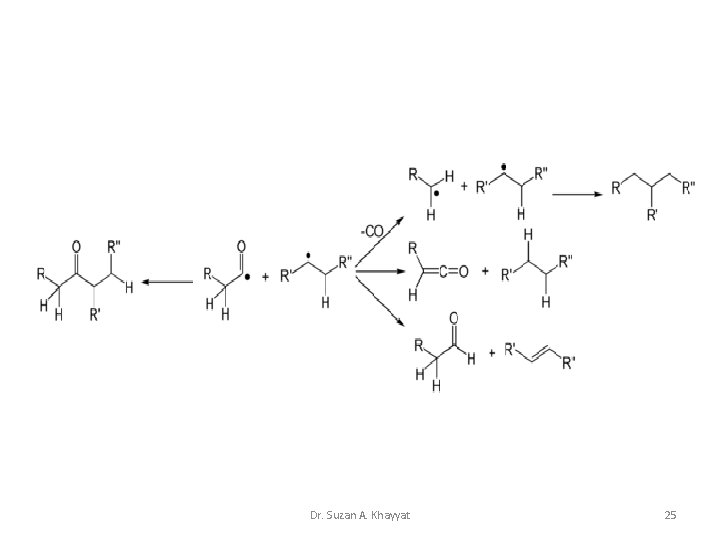
Carbonyl Compounds 1 - Keetones: • Norrish Type I: The Norrish type I reaction is the photochemical cleavage or homolysis of aldehydes and ketones into two free radical intermediates. The carbonyl group accepts a photon and is excited to a photochemical singlet state. Through intersystem crossing the triplet state can be obtained. On cleavage of the α-carbon bond from either state, two radical fragments are obtained. Dr. Suzan A. Khayyat 23

Dr. Suzan A. Khayyat 24

Dr. Suzan A. Khayyat 25

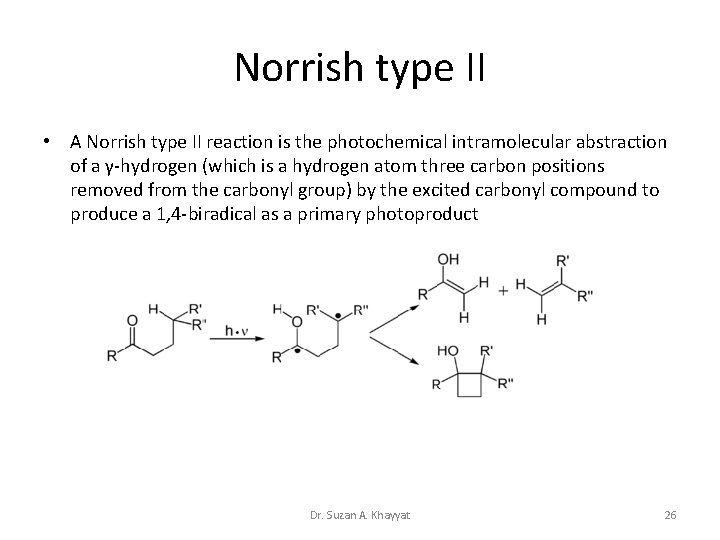
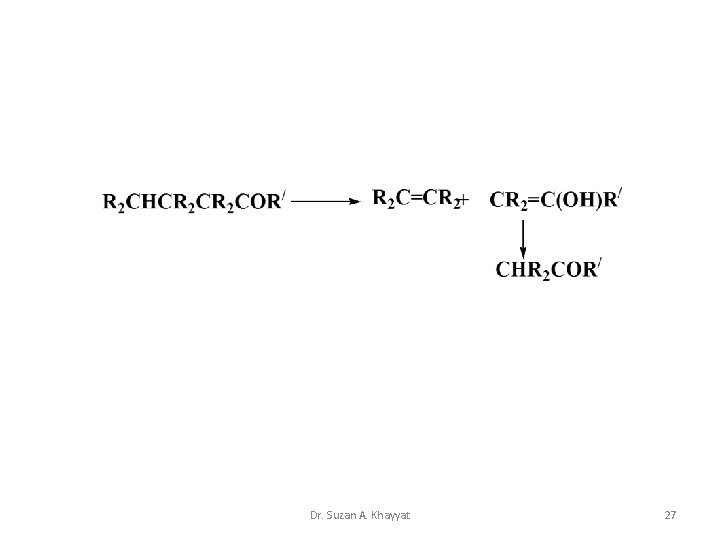
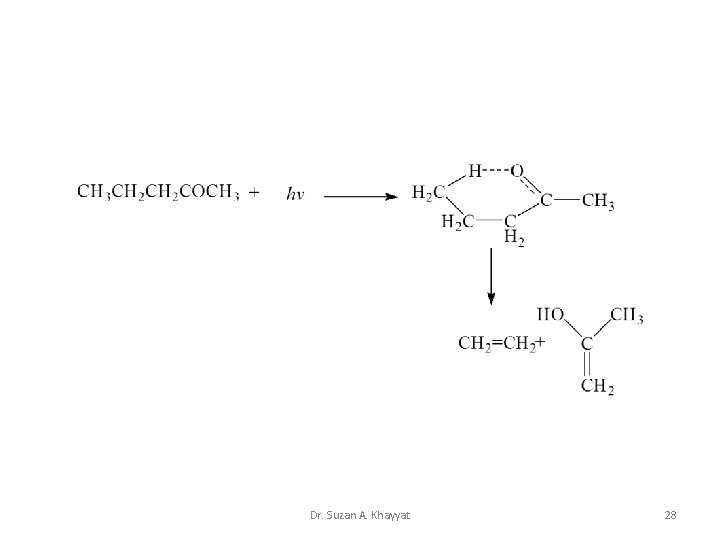
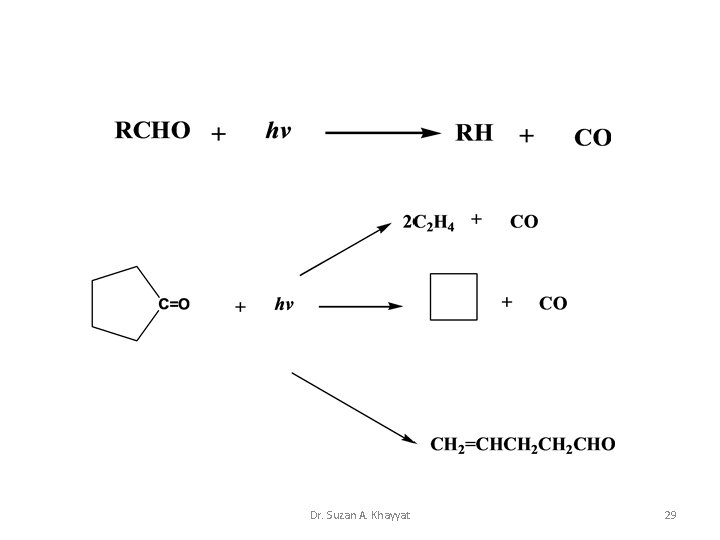
Norrish type II • A Norrish type II reaction is the photochemical intramolecular abstraction of a γ-hydrogen (which is a hydrogen atom three carbon positions removed from the carbonyl group) by the excited carbonyl compound to produce a 1, 4 -biradical as a primary photoproduct Dr. Suzan A. Khayyat 26

Dr. Suzan A. Khayyat 27

Dr. Suzan A. Khayyat 28

Dr. Suzan A. Khayyat 29

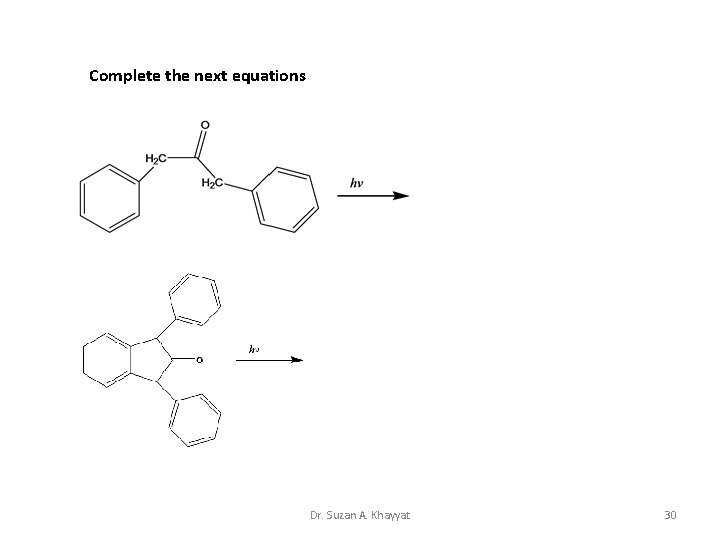
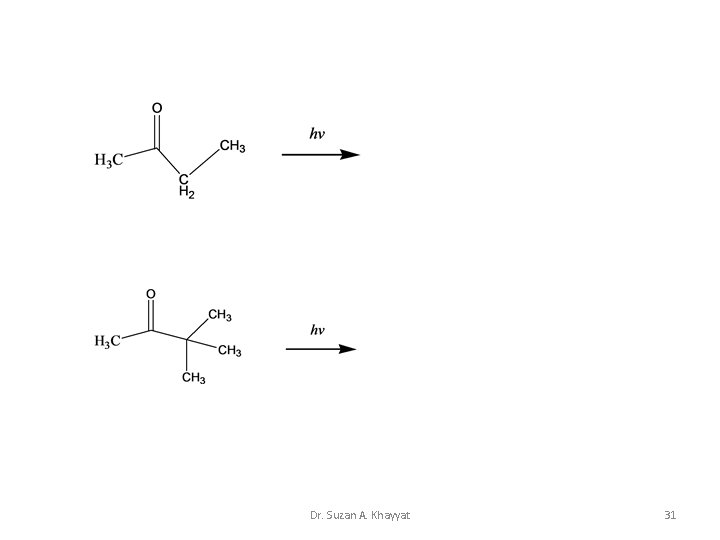
Complete the next equations Dr. Suzan A. Khayyat 30

Dr. Suzan A. Khayyat 31

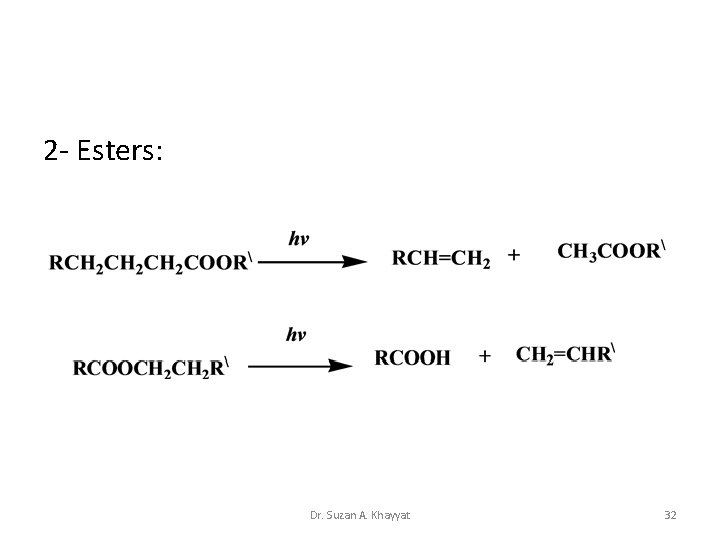
2 - Esters: Dr. Suzan A. Khayyat 32

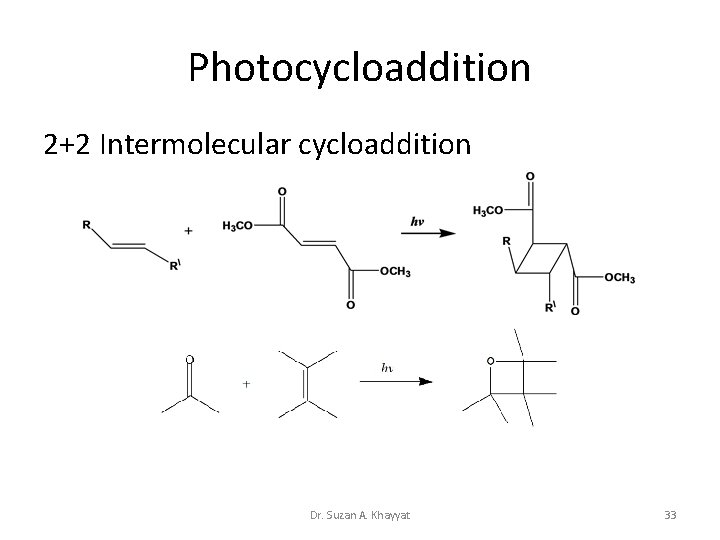
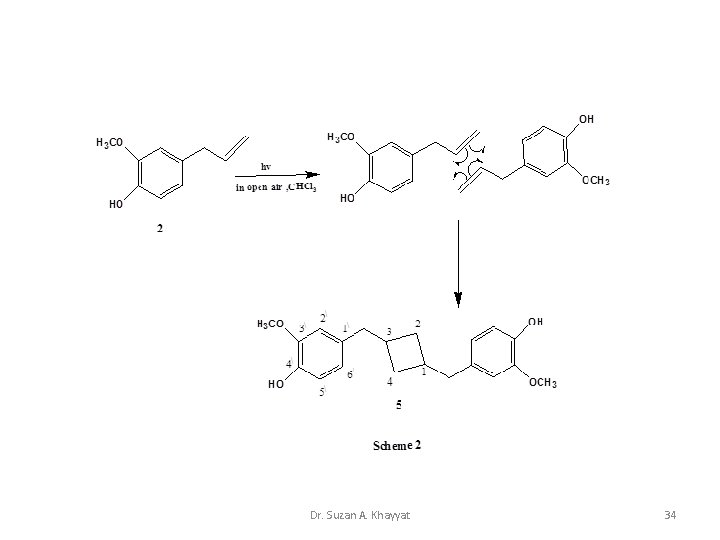
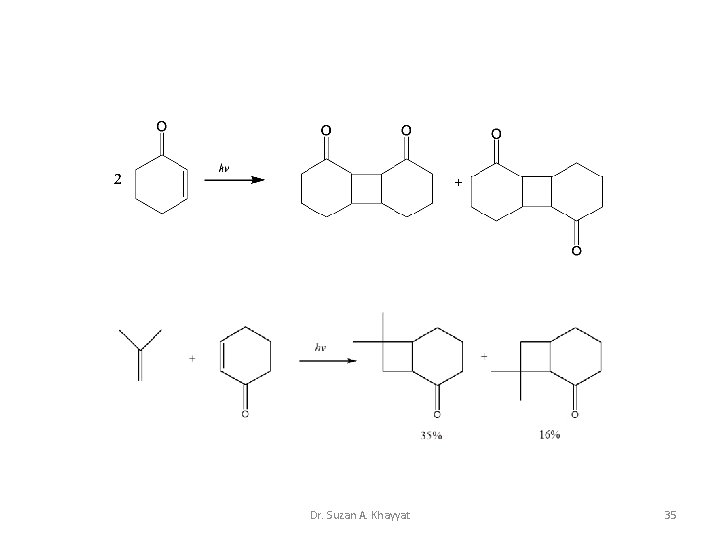
Photocycloaddition 2+2 Intermolecular cycloaddition Dr. Suzan A. Khayyat 33

Dr. Suzan A. Khayyat 34

Dr. Suzan A. Khayyat 35

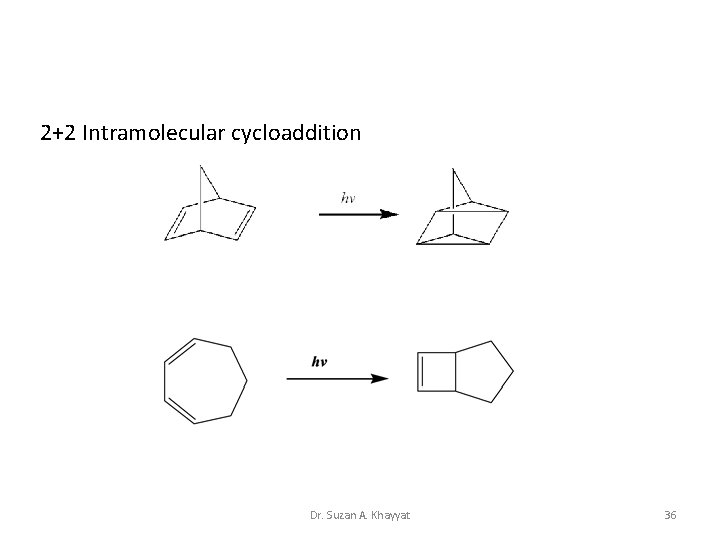
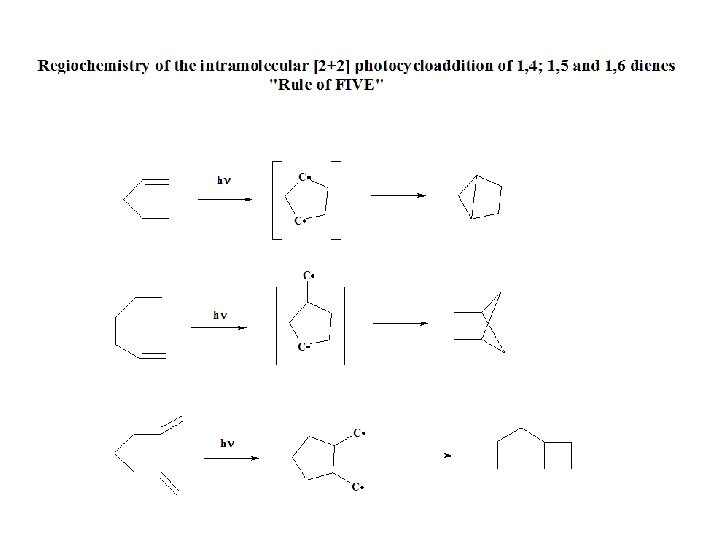
2+2 Intramolecular cycloaddition Dr. Suzan A. Khayyat 36

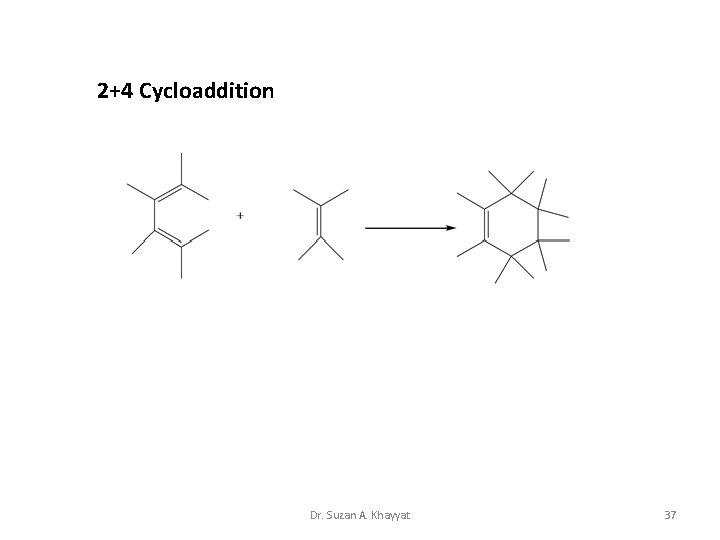
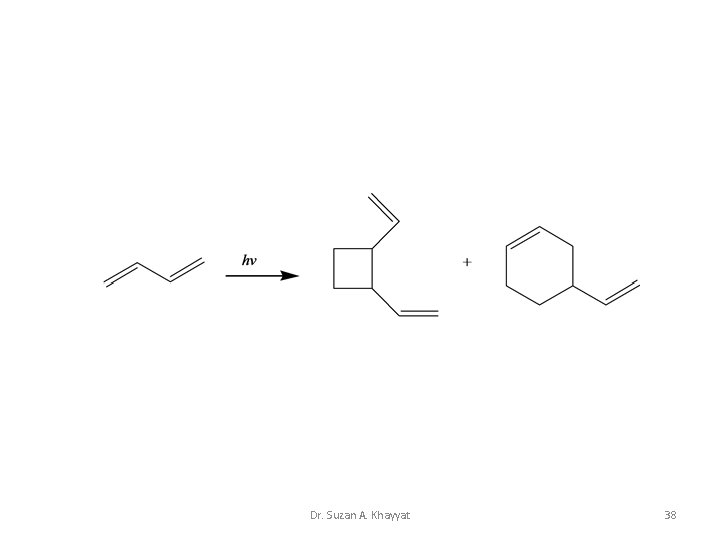
2+4 Cycloaddition Dr. Suzan A. Khayyat 37

Dr. Suzan A. Khayyat 38

Dr. Suzan A. Khayyat 39

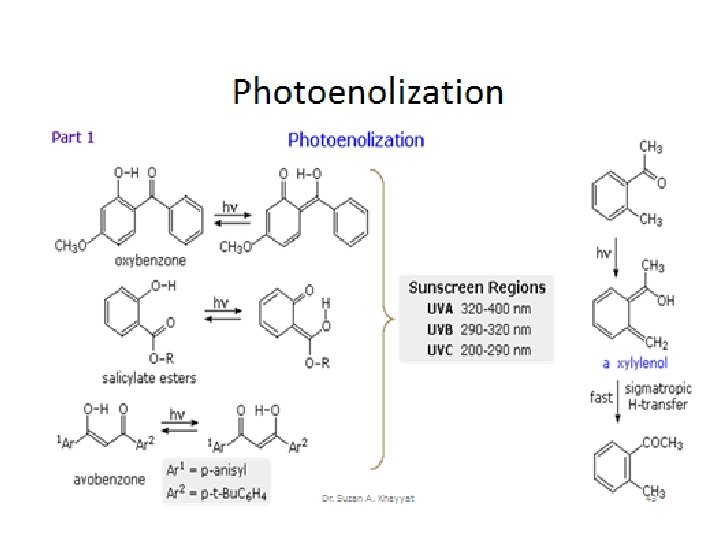
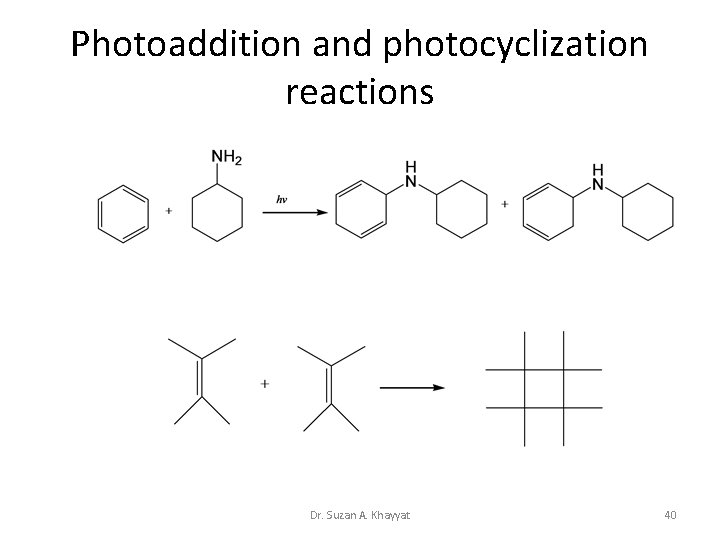
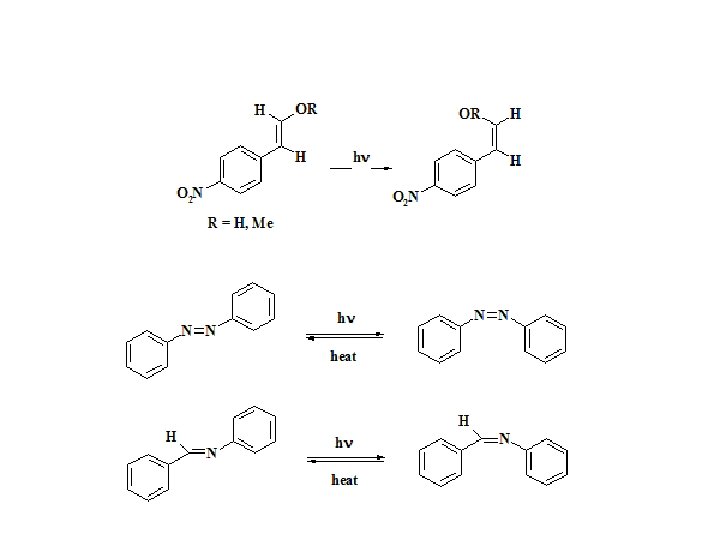
Photoaddition and photocyclization reactions Dr. Suzan A. Khayyat 40

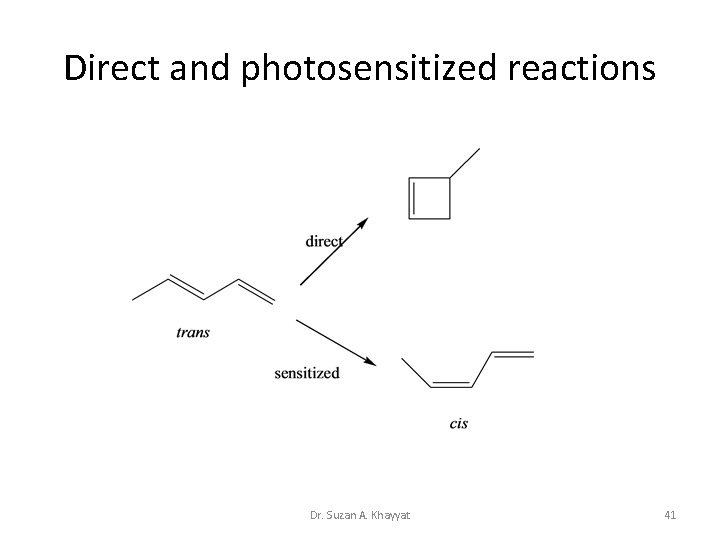
Direct and photosensitized reactions Dr. Suzan A. Khayyat 41

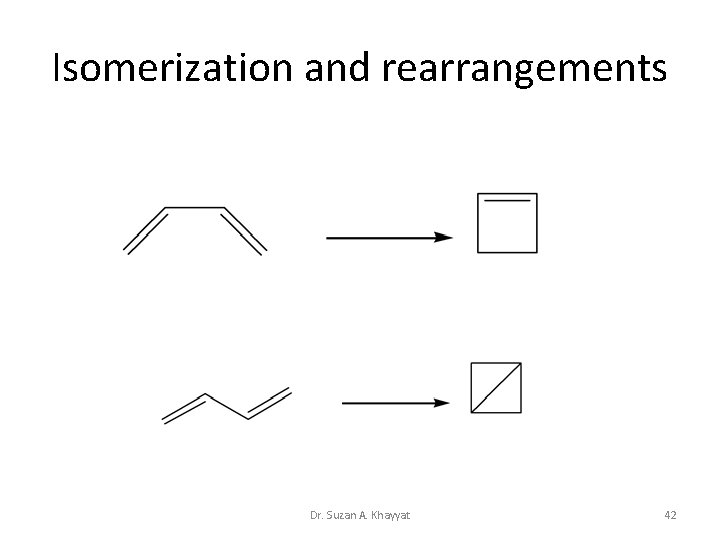
Isomerization and rearrangements Dr. Suzan A. Khayyat 42



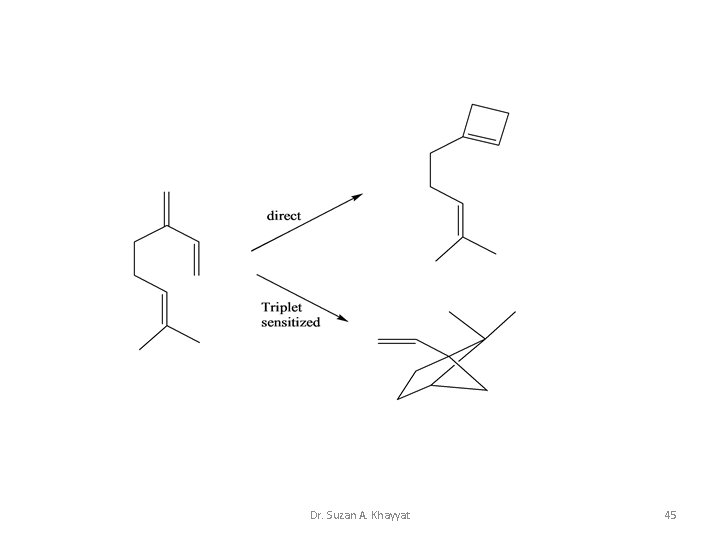
Dr. Suzan A. Khayyat 45

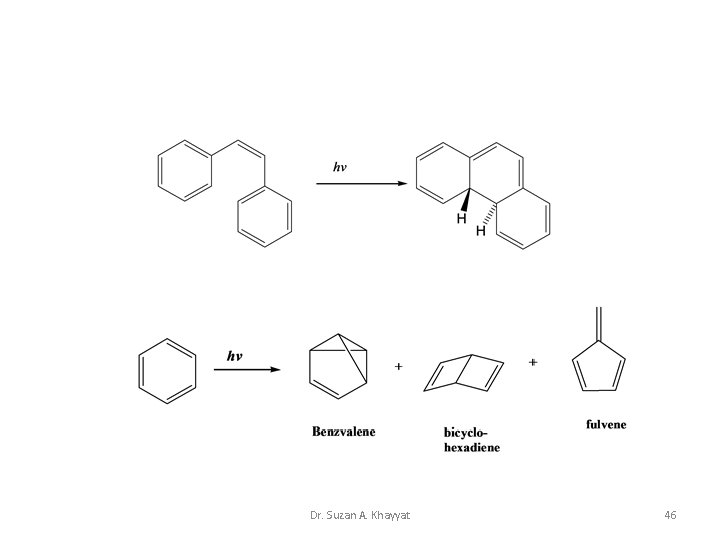
Dr. Suzan A. Khayyat 46

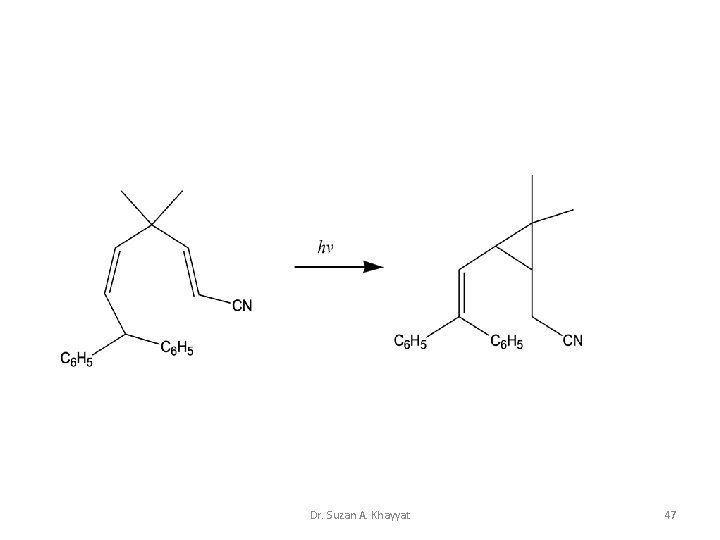
Dr. Suzan A. Khayyat 47

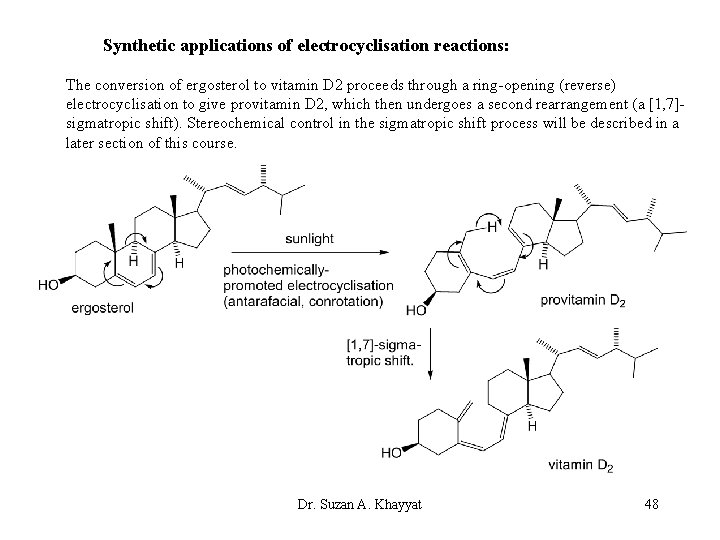
Synthetic applications of electrocyclisation reactions: The conversion of ergosterol to vitamin D 2 proceeds through a ring-opening (reverse) electrocyclisation to give provitamin D 2, which then undergoes a second rearrangement (a [1, 7]sigmatropic shift). Stereochemical control in the sigmatropic shift process will be described in a later section of this course. Dr. Suzan A. Khayyat 48

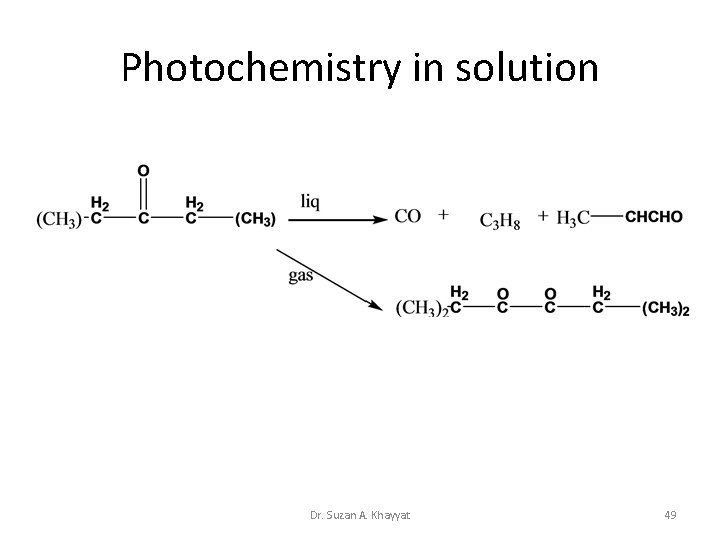
Photochemistry in solution Dr. Suzan A. Khayyat 49

Factors determining reactivity • 1 • • The excess energy possessed by the species (which may help overcome activation barriers). 2 - The intrinsic reactivity of the specific electronic arrangement. 3 - The relative efficiencies of the different competing pathways for loss of the particular electronic state. 4 - The type of orbital (s, p, σ, or, π, etc. ) and its symmetry. 5 - Explicit in the correlation rules for orbital symmetry and spin that are introduced first at the end of this section.