Chemical BondsReview Objectives I can differentiate the 5

Chemical Bonds/Review

Objectives: -I can differentiate the 5 types of reactions

How many covalent bonds does nitrogen prefer to form?

3
What occurs in an exothermic reaction?

-Heat is released -Chemical bonds are broken

How many electrons can fit in the 1 st 2 nd 3 rd Energy levels?

2 8 18

How is a polar covalent bond different from a nonpolar covalent bond?

-Polar covalent bond is unequal sharing of electrons, creating a partial charge -Nonpolar covalent bond is equal sharing of electrons, no charge at all

What are the key parts of a potential energy diagram?

X axis: Reaction Progress Y axis: Potential Energy Labels: reactants, products, activation complex, activation energy
What is an ion? NOT AN IONIC BOND

It is an atom that lost or gained an electron

What subatomic particle has the least mass?

Electron

What occurs in an endothermic reaction?

-Energy is absorbed -Chemical bonds are formed
What has the largest mass in an atom?

Nucleus

What is ionization energy?

Ability to lose electrons -Lowest in Metals, which form cations

How is atomic mass different from atomic number?

Atomic Mass: sum of masses of all isotopes, in amu Mass number: protons plus neutrons, rounded atomic mass
Why is a metallic bond so conductive?

-Due to the sea of electrons that is produced in a metallic bond

Where are electrons found?

Electron cloud
What do isotopes have in common?

Number of electrons and protons

What is the -Molecular Geometry Name AND the -Electron pair geometry name of CO₂
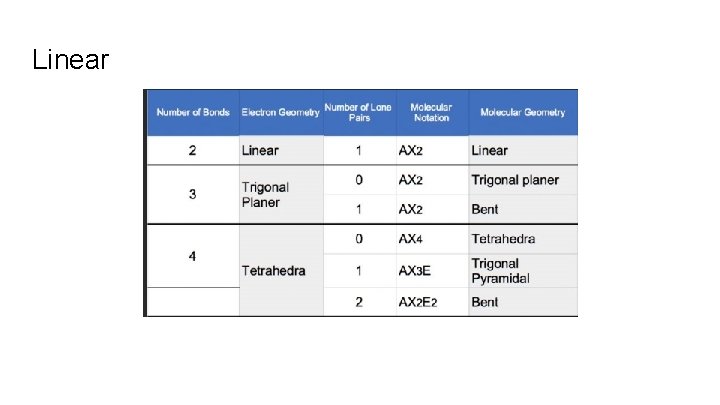
Linear

Where are electrons located on the Bohr Atom Diagram?

Energy Levels Specific Orbitals
What is different between isotopes?
Number of Neutrons Mass Number

What is the name of the compound below Al₂O₃

Aluminum Oxide

What is the Lewis Dot structure for NH₃

What type of covalent bond, requires the most energy to break?

Triple Bond
What is electronegativity? What difference value results in a polar covalent bond?

-Ability to attract electrons -0 -. 39 Nonpolar -. 4 -1. 69 Polar -1. 7 Ionic
What is the lowest energy level in an atom, for an electron?

Ground state
What type of covalent bond is the longest?

Single
How do intermolecular forces compare to intramolecular forces?

Inter: weaker, attractive force Intra: stronger, chemical bond

When is a roman numeral used to name a compound?
-Ionic compound with a transition metal Ex Copper (II) Chloride

How does an atom release ER?

-Electron moves to a lower excited state, releasing photons, which is the smallest energy unit of light

What is the name of the following? C₆O₈

Hexacarbon octoxide
What occurs to reactivity as you move across the periodic table?

It decreases

Group Mini Project: 15 HW points -Develop a poster/whiteboard with the following -5 types of reactions -general equations -how to identify each type -model reaction (using beads) Time: 30 min

Balancing Equations Check In 1) Solve questions 2) Exchange 3) Peer Review

ACT Discussion: -Areas for improvement -What other supports are needed? -What did you need more practice on?
- Slides: 61