Chemical Bonds the attractive intramolecular forces ie the

Chemical Bonds: the attractive (intramolecular) forces, ie. “ the glue”, strong enough to maintain a group of atoms together for an indefinite amount of time. There are three basic types of bonds: Ionic, Covalent, & Metallic.
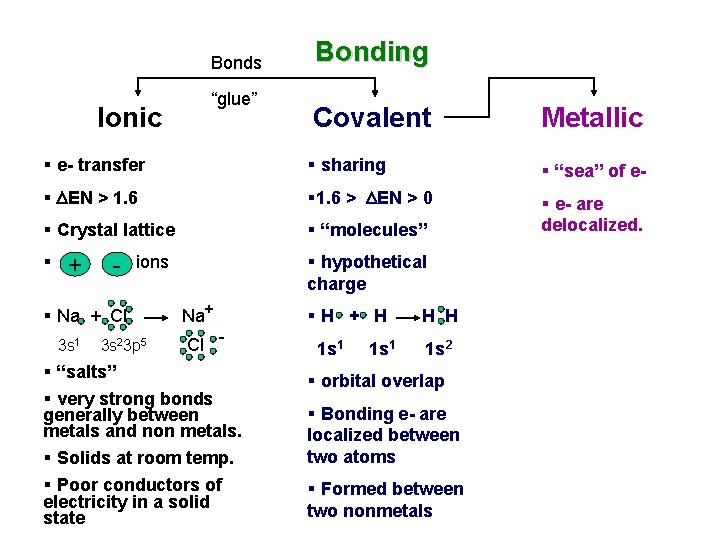
Bonds Ionic “glue” Bonding Covalent Metallic § e- transfer § sharing § “sea” of e- § DEN > 1. 6 § 1. 6 > DEN > 0 § Crystal lattice § “molecules” § e- are delocalized. § + - § Na + Cl 3 s 1 § hypothetical charge ions 3 s 23 p 5 Na+ Cl - § “salts” § very strong bonds generally between metals and non metals. § Solids at room temp. § Poor conductors of electricity in a solid state §H + H 1 s 1 H H 1 s 2 § orbital overlap § Bonding e- are localized between two atoms § Formed between two nonmetals
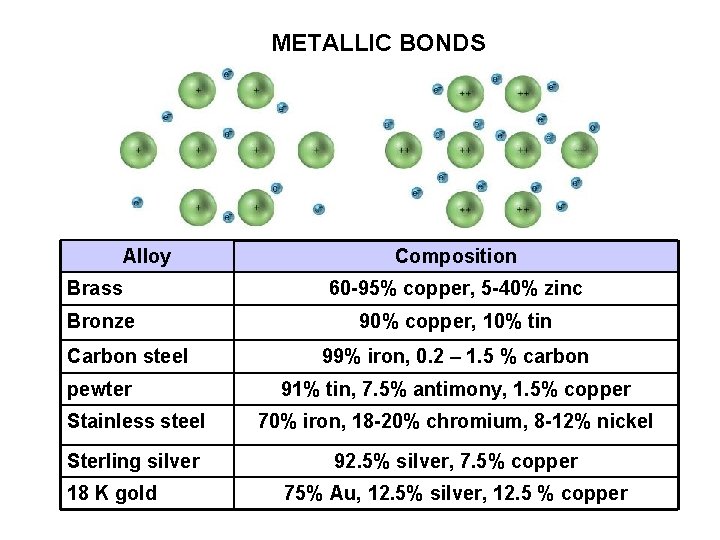
METALLIC BONDS Alloy Brass Bronze Carbon steel pewter Composition 60 -95% copper, 5 -40% zinc 90% copper, 10% tin 99% iron, 0. 2 – 1. 5 % carbon 91% tin, 7. 5% antimony, 1. 5% copper Stainless steel 70% iron, 18 -20% chromium, 8 -12% nickel Sterling silver 92. 5% silver, 7. 5% copper 18 K gold 75% Au, 12. 5% silver, 12. 5 % copper
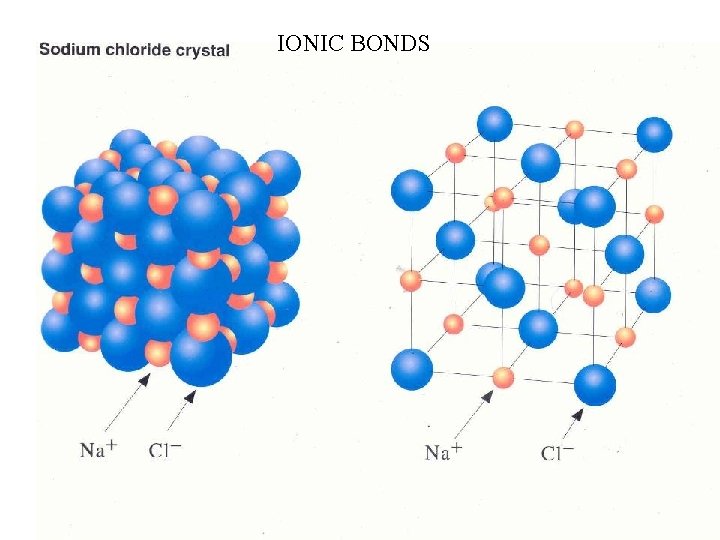
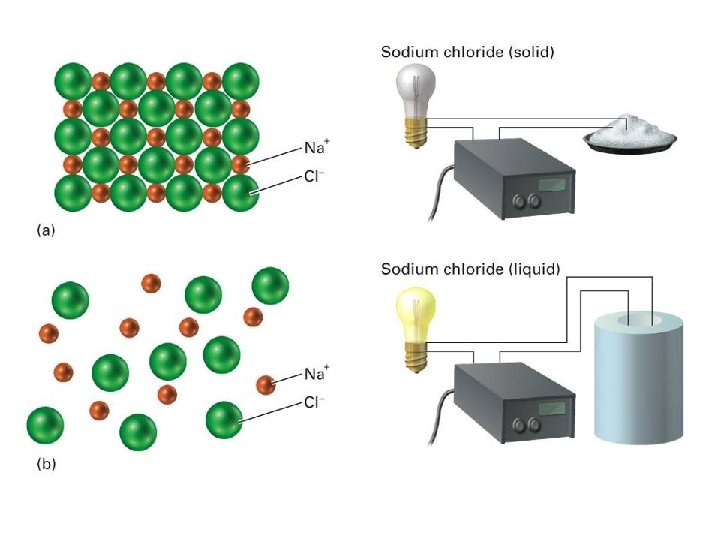
IONIC BONDS


DIPOLE MOMENT & POLARITY Polarity is a term that describes the charge distribution about either a bond or a molecule. If the electrons involved in a bond are unequally distributed along the bond axis and the electrons reside closer to the more electronegative atom, this bond is refered to as a POLAR covalent bond If the electrons are equally distributed (shared) along the bond axis then this type of bond is called NONPOLAR covalent bonds A dipole moment can be used to visualize the polarity of the bond and is represented by an arrow pointing towards the more electronegative atom and a “+” on the tail.

COVALENT BONDS POLAR COVALENT BONDS
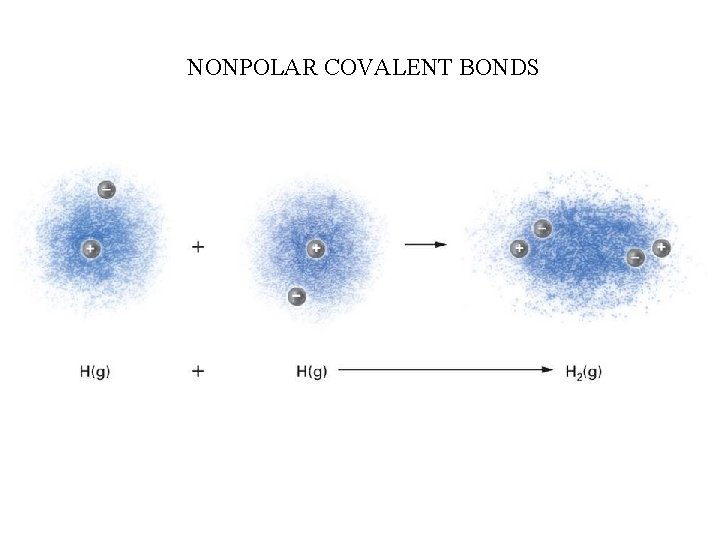
NONPOLAR COVALENT BONDS
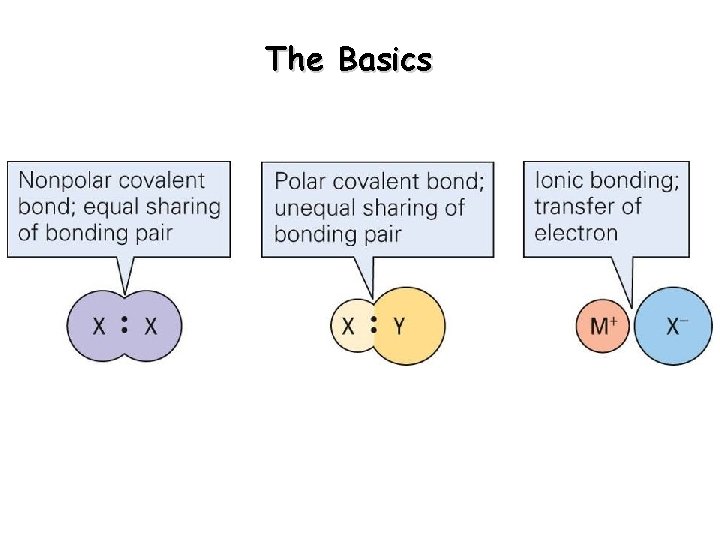
The Basics

POLARITY OF A BOND + + + H-Cl S-N C-O 2. 1 - 3. 0 2. 5 - 3. 5 D = 0. 9 D = 0. 5 D = 1. 0 If bonds were formed between any of the above examples, the bond would be refered to as a polar covalent bond. A dipole moment can be drawn in each case because the difference in electronegativity is greateer than zero. The example below has zero difference in electronegativity and no dipole can be drawn. S-C nonpolar covalent bond 2. 5 - 2. 5 • D=0
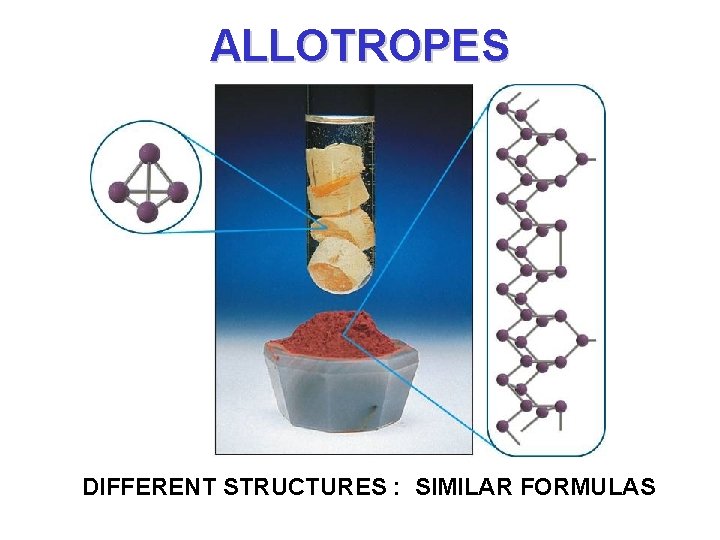
ALLOTROPES DIFFERENT STRUCTURES : SIMILAR FORMULAS
- Slides: 12