Chemical Bonds Notes Atoms combine to form compounds

Chemical Bonds Notes • Atoms combine to form compounds by holding pairs of electrons between them. • *(Draw the picture below) • The paired electrons are known as a chemical bond. H H C H H

Chemical Bond Classification • Chemical bonds are classified by the way the electrons are held between the atoms that are bonding to each other.

Chemical Bond Classification • Electrons can be held in a bond by one of two ways: – Electrons are equally shared by both atoms. (Covalent) – Electrons are “exchanged” from one atom to another forming ions. (Ionic)

Properties of Covalent Bonds • Covalent bonds occur between two non-metal atoms. • In covalent bonds electrons are shared by the atoms involved in the bond.

Examples of Covalent Bonds • Examples of compounds formed with covalent bonds include: – Hydrogen gas (H 2) – Ammonia (NH 3) – Large biological molecules such as glucose (C 6 H 12 O 6).

Properties of Ionic Bonds • Ionic bonds occur between a metal atom and a non-metal atom. • In ionic bonds electrons are exchanged or transferred from the metal atom to the non-metal atom. • After the transfer of electrons, both atoms become oppositely charged, keeping both atoms together (bonded).

Examples of Ionic Bonds • Examples of compounds formed with ionic bonds include: – Sodium chloride (Na. Cl) – Strontium sulfide (Sr. S)

Next steps…if you’ve mastered bonding All elements have a certain ELECTRONEGATIVITY This describes tendency of an atom to attract electrons towards itself. (aka-it’s hunger for electrons) High Electronegativity = High attraction/hunger for electrons. Low Electronegativity = Low attraction/hunger for electrons.

Electronegativity differences and bonding • If atom A is very electronegative (electron hungry) and atom B is not, the electrons will move toward atom A and this will result in an IONIC BOND where the electrons are transferred. • If atom A’s electronegativity is only a little greater than atom B, they will share the electrons unevenly (POLAR COVALENT BOND) (electrons will be closer to atom A and one end of the group is more negative than the other. ) • If atom A and atom B have the same or very similar electronegativities the electrons are evenly shared and it makes a NONPOLAR COVALENT BOND.
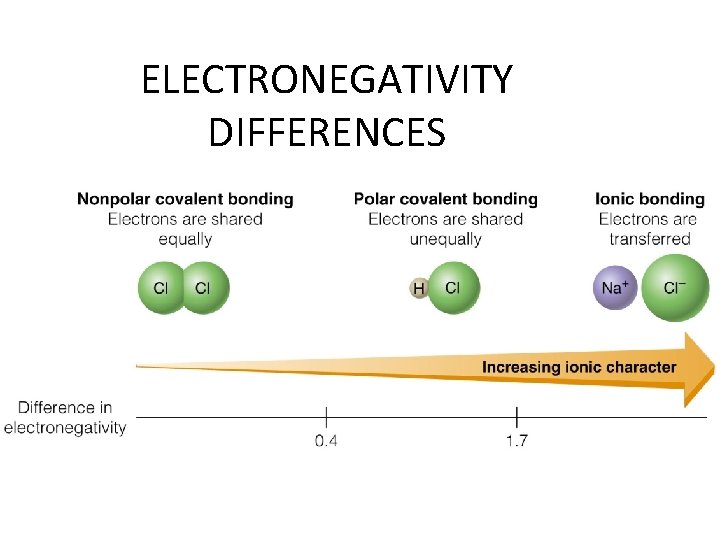
ELECTRONEGATIVITY DIFFERENCES
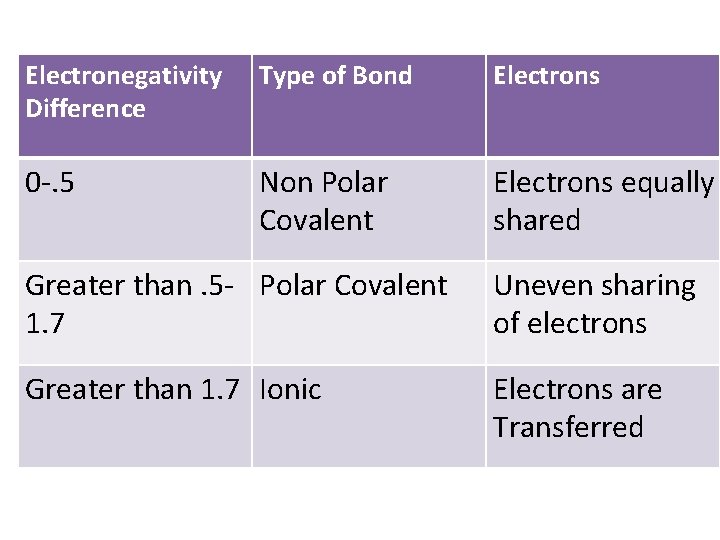
Electronegativity Difference Type of Bond Electrons 0 -. 5 Non Polar Covalent Electrons equally shared Greater than. 5 - Polar Covalent 1. 7 Uneven sharing of electrons Greater than 1. 7 Ionic Electrons are Transferred

Some periodic tables will also list the electronegativity.

Ionic Bond or Covalent Bond 1. 2. 3. 4. 5. Na. Cl Fe. Cl 3 SO 3 H 2 O Mg. O • • • Ionic Covalent Ionic How do you know? Look at the atoms and determine whethere are metal atoms present or not. (Metal & Nonmetal=Ionic!)

Practice!! Get your whiteboards Ionic Bonding • Potassium and Fluorine • Magnesium and Iodine • Sodium and Oxygen • Sodium and Chlorine • Calcium and Chlorine • Aluminum and Chlorine Covalent Bonding • Hydrogen and Hydrogen • Hydrogen and Oxygen • Chlorine and Chlorine • Carbon and Hydrogen • Oxygen and Oxygen • Carbon and Oxygen
- Slides: 14