Chemical Bonds Mr Conkey Physical Science Ch 6

Chemical Bonds Mr. Conkey Physical Science Ch. 6

Stable Electron Configurations • When the highest energy level of an atom is filled with e−’s, it is stable and unlikely to react • Group 8 all have stable e− configurations (including He) • Chemical properties depend on the # of valence e−’s • Electron dot diagram – a model of an atom in which each dot represents a valence e−
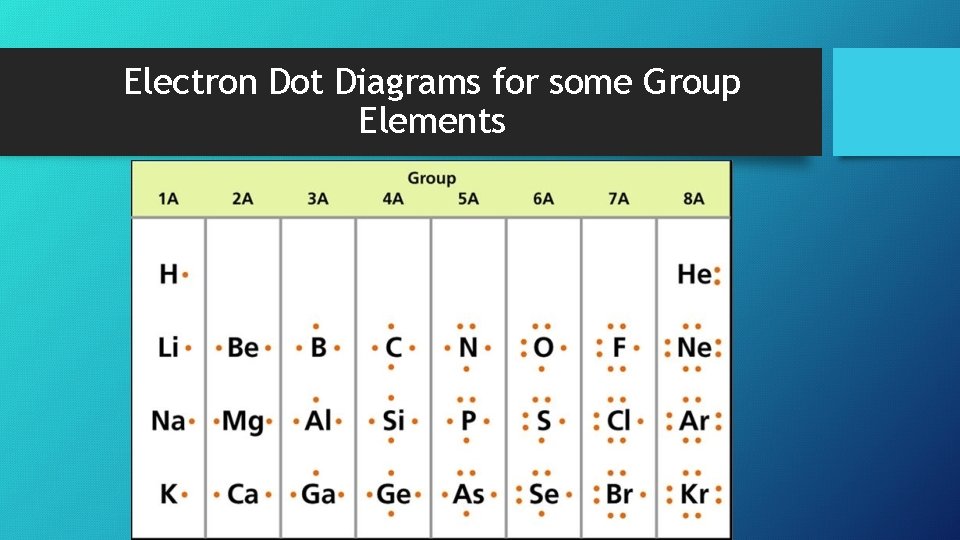
Electron Dot Diagrams for some Group Elements

Ionic Bonds (e− Donors and e− Acceptors) • Elements with incomplete sets of valence e−’s tend to react • Some elements achieve a stable electron configuration through the transfer of electrons between atoms • Example of electron transfer: Full Octet e− acceptor (now stable) Donated e− (also now e− donor stable

Formation of Ions • Ions – form when an atom gains or loses an electron and the number of protons and electrons become ≠ • The charge on an ion is shown by a plus or minus sign (i. e. Na+ and Cl ‾ ) • Cation – an ion with a positive charge (+) • Anion – an ion with a negative charge ( ‾ )
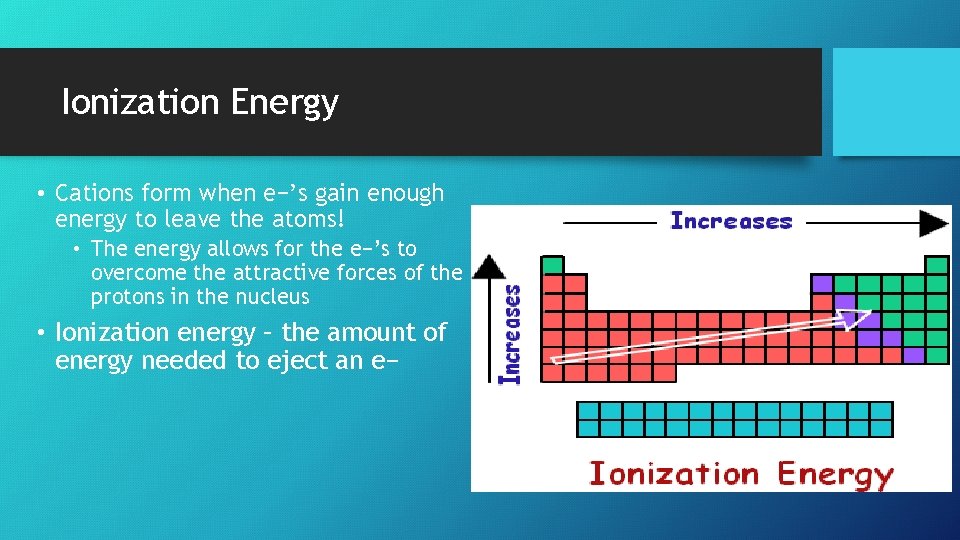
Ionization Energy • Cations form when e−’s gain enough energy to leave the atoms! • The energy allows for the e−’s to overcome the attractive forces of the protons in the nucleus • Ionization energy – the amount of energy needed to eject an e−

Ionic Compounds • Ionic compounds – compounds that contain ionic bonds • Chemical formula – a notation that shows the ratio of the atoms/ions of elements in a compound as well as what elements are in the compound • Example: Mg. Cl₂

Properties of Ionic Compounds • Ionic compounds have high melting points, good conductors of electric current (when melted), and are brittle • Ionic compounds tend to shatter when struck because the ions move from their fixed positions and like charges repel each other causing them to shatter

Covalent Bonds (sharing of e−’s) • Nonmetals bond together using covalent bonds (two atoms sharing a pair of valence e−’s) and form a molecule • Two Hydrogen atoms share their valence e−’s to form a stable e− configuration • Molecule – a neutral group of atoms joined together by one or more covalent bonds H₂ Molecule

Molecules and Multiple Covalent Bonds • Attractions between the shared e−’s and the protons in the nucleus hold atoms together in covalent bonds • The amount of covalent bonds needed in a molecule depends on the # of valence e−’s of the atoms
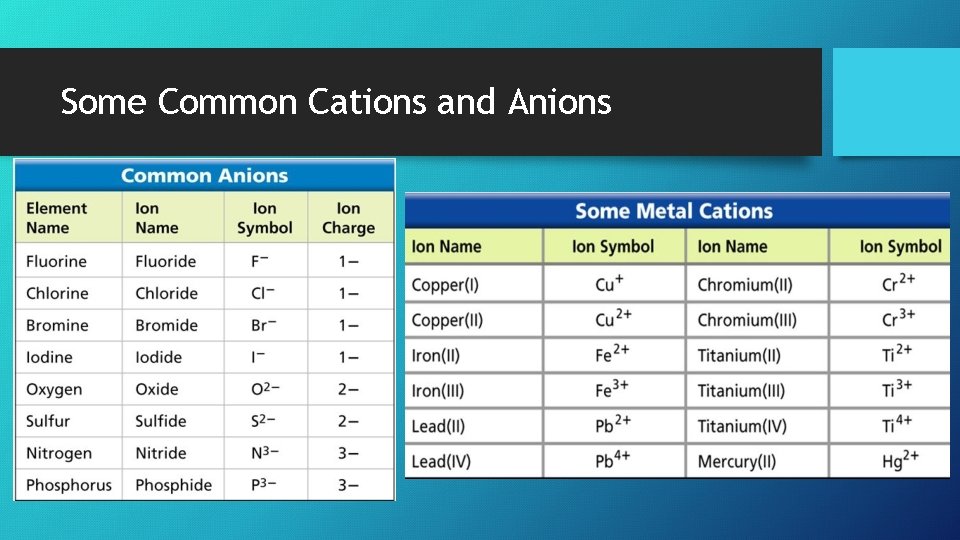
Some Common Cations and Anions

Metallic Bonds • Metallic bond – the attraction between a metal cation and the shared e−’s surrounding it • Metallic bonds are relatively strong • Electric current and malleability are due to the ability for the e−’s to move • Pool of e−’s allow for good electric conductivity • Cation and e−’s shifting allow for malleability

Alloys • Alloy – a mixture of two or more elements (at least one has to be a metal!) • Cu alloys: • Bronze (Cu and Sn); two soft metals when mixed are much harder/stronger • Brass (Cu and Zn); softer than bronze and easier to shape and shinier Bronze Brass
You Made it, its the end of the notes!!!! WOOHOOOOOO!!!!!!!
Now the Battle of the Finals Begins!
- Slides: 15