Chemical Bonds Gilbert Newton Lewis surrounded by pairs

Chemical Bonds

Gilbert Newton Lewis (surrounded by pairs of electrons)
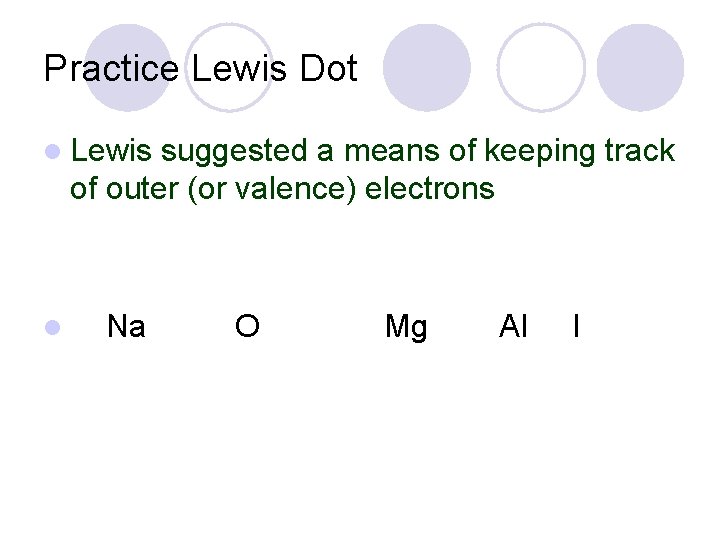
Practice Lewis Dot l Lewis suggested a means of keeping track of outer (or valence) electrons l Na O Mg Al I

Overview: Types of Bonds There are 2 bond types : ionic and covalent l In ionic bonding one atom has a stronger attraction for electrons than the other, and “steals” an electron from a second atom l In covalent bonding the attraction for electrons is similar for two atoms. They share their electrons to obtain an octet. l l Mg. O (ionic), Ca. Cl 2 (ionic), SO 2 (covalent), Pb. Cl 2 (ionic), CCl 4 (covalent), CH 4 (covalent)
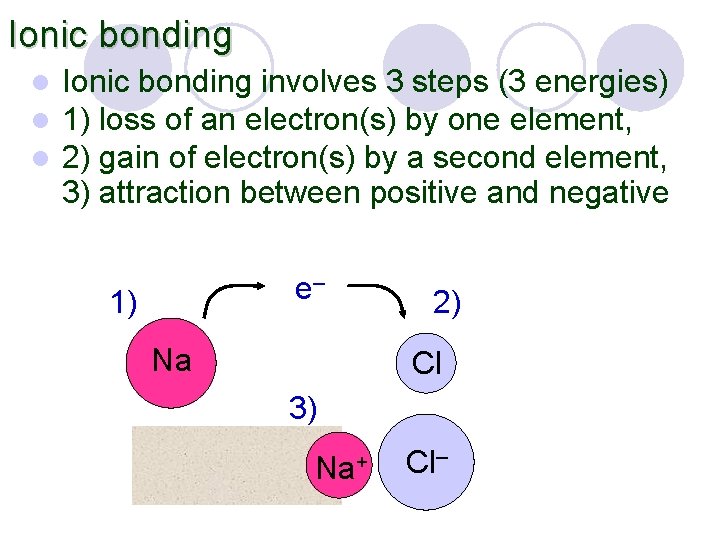
Ionic bonding l l l Ionic bonding involves 3 steps (3 energies) 1) loss of an electron(s) by one element, 2) gain of electron(s) by a second element, 3) attraction between positive and negative e– 1) Na 2) Cl 3) Na+ Cl–

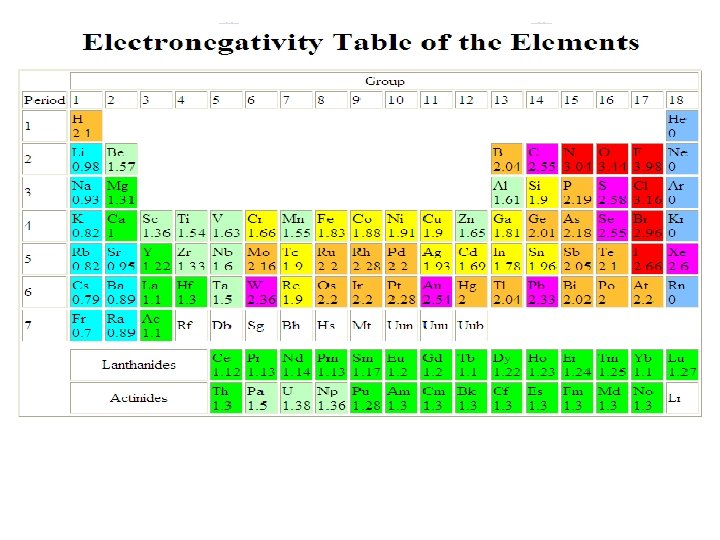
When is the bond Ionic? l Metal and non-metal (alkali & Halogens) l Electronegativity Difference l The rule is that when the electronegativity difference is greater than 1. 7, the bond is ionic.

HOW TO DO An Ionic Bond l Step 1 find out valence electrons for each Element. l Draw diagram (Lewis Dot Diagram) l Show the transfer l Write the answer without the electron dots
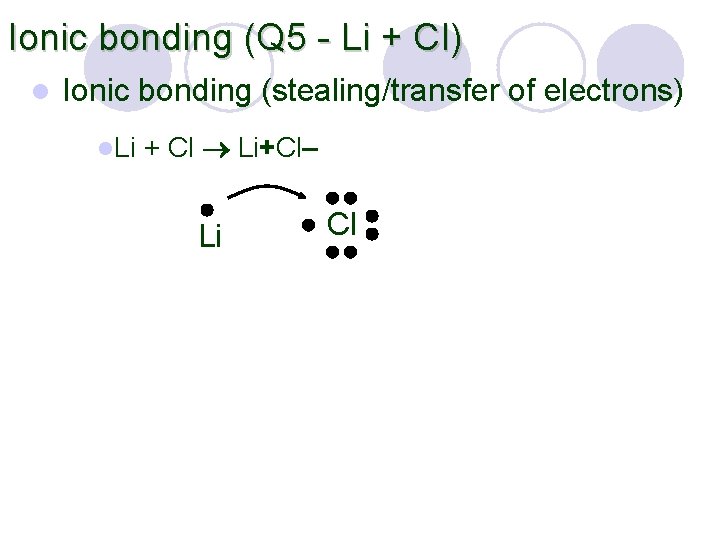
Ionic bonding (Q 5 - Li + Cl) l Ionic bonding (stealing/transfer of electrons) l. Li + Cl Li+Cl– Li Cl
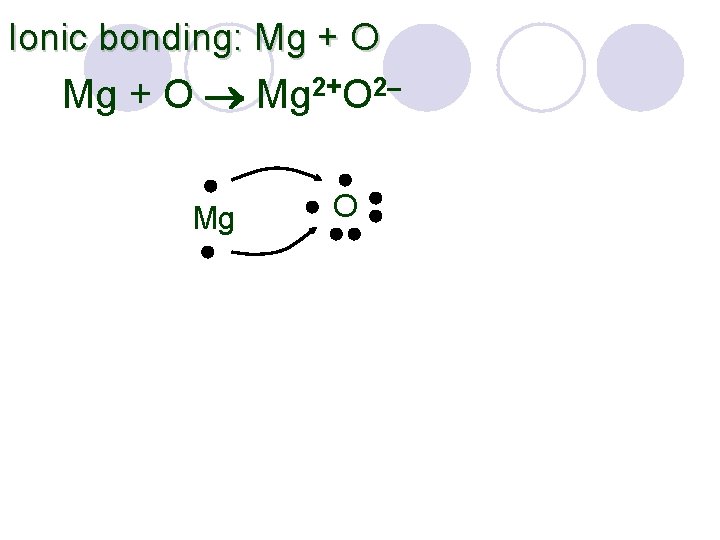
Ionic bonding: Mg + O Mg 2+O 2– Mg O

Ionic bonding: Al + 3 Cl

COVALENT BONDS l Step 1 find out valence electrons for each Element. l Draw diagram (Lewis Dot Diagram) l Connect the Dots l Draw the circles to show sharing of electrons l Draw the bond structure using symbols and Lines l Write the answer without the electron dots

F 2

CS 2
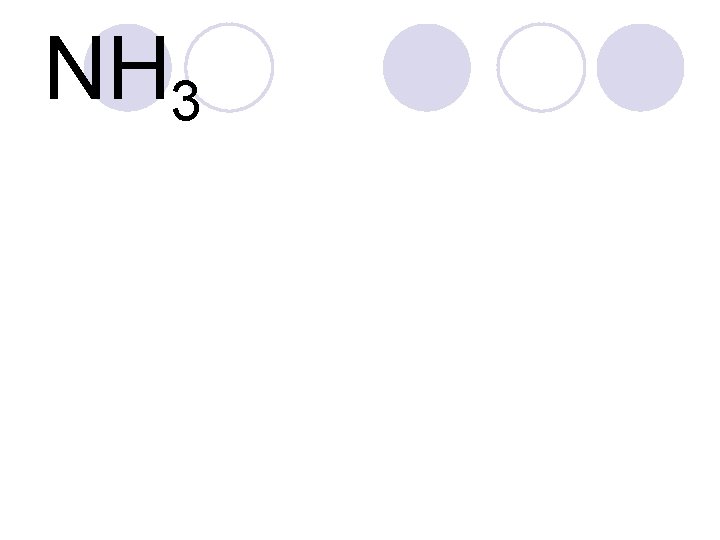
NH 3
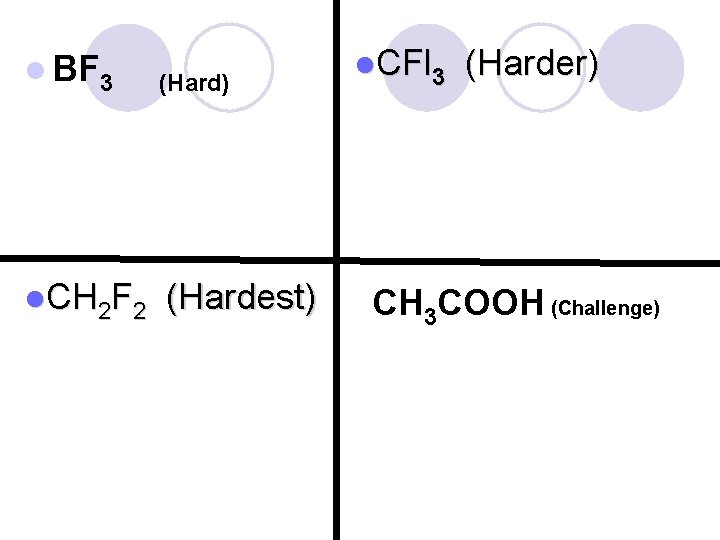
l BF 3 (Hard) l. CH 2 F 2 (Hardest) l. CFl 3 (Harder) CH 3 COOH (Challenge)
- Slides: 16