Chemical Bonds Chapter 6 Smart Starter What is

Chemical Bonds Chapter 6

Smart. Starter • What is the octet rule?

Smart. Starter • Draw the electron dot diagram for ionic compound Mg. Cl 2

Compare – Eyes on Chemistry • • Epsom Salts Rock Salt Sucrose One more… • WHAT DO YOU NOTICE ABOUT THEIR STRUCTURE?
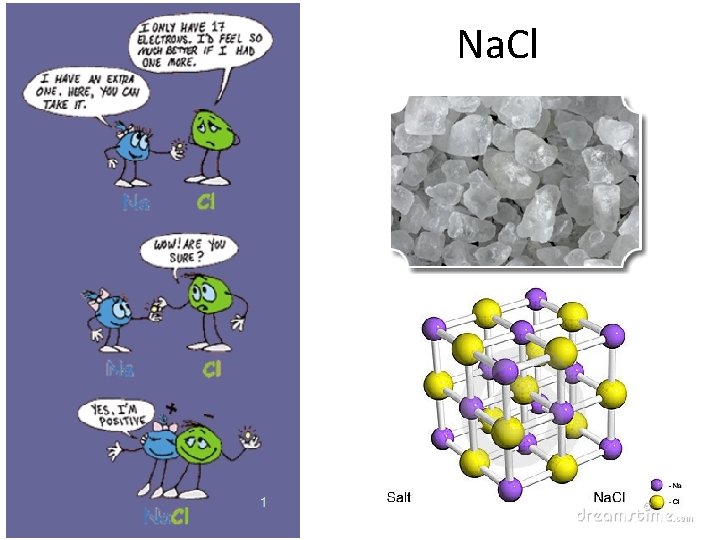
Na. Cl

Valence Electrons and Lewis Dot Structure (AKA Electron Dot Diagram) • Remember: Octet Rule? Valence Electrons? • How many valence electrons in… Example: Sulfur • Your turn 1 -20 – count off!
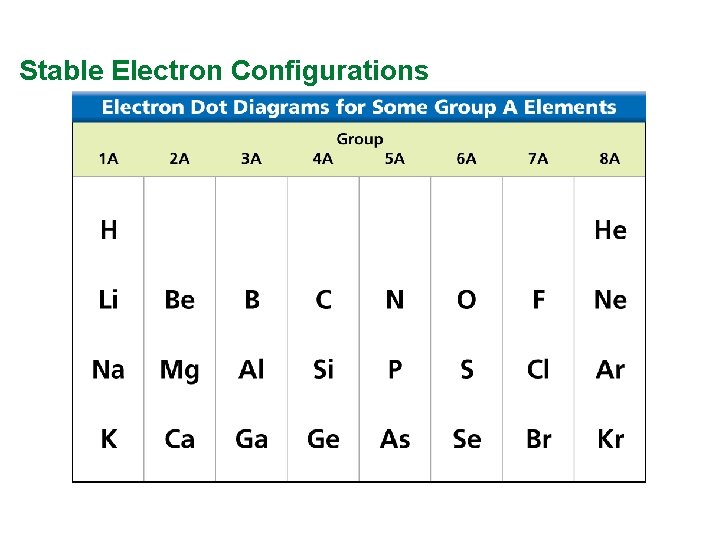
Stable Electron Configurations
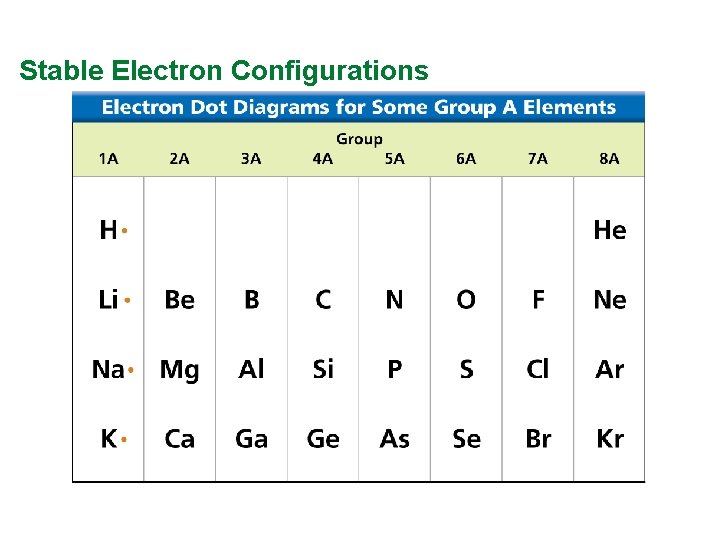
Stable Electron Configurations
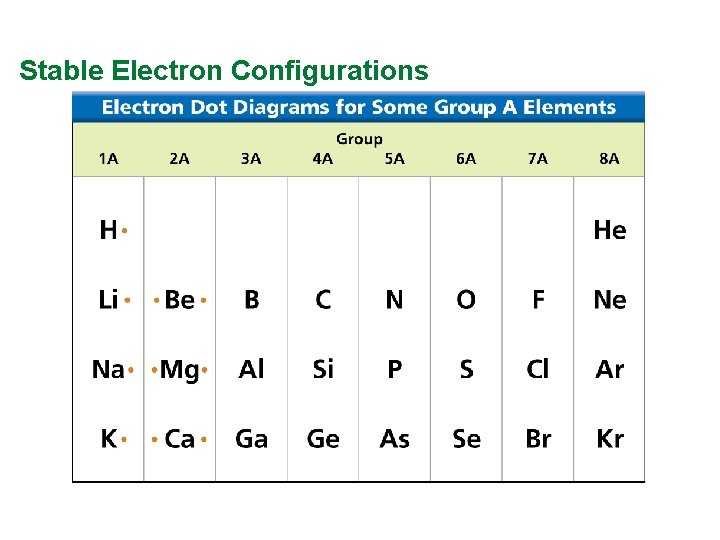
Stable Electron Configurations
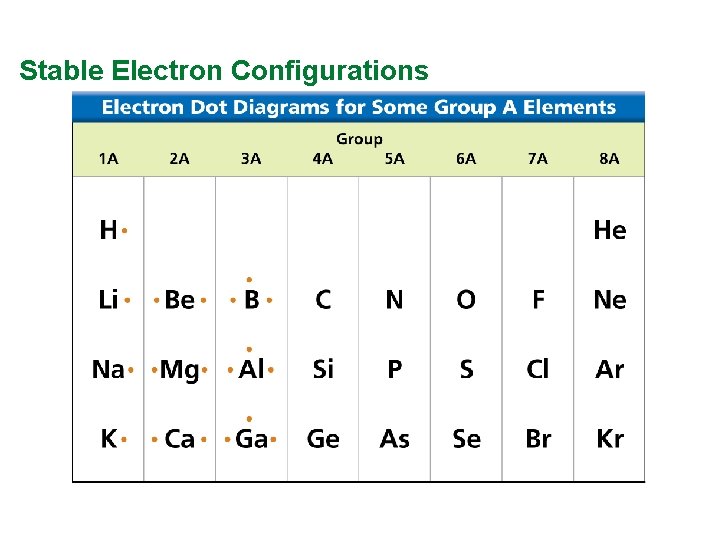
Stable Electron Configurations
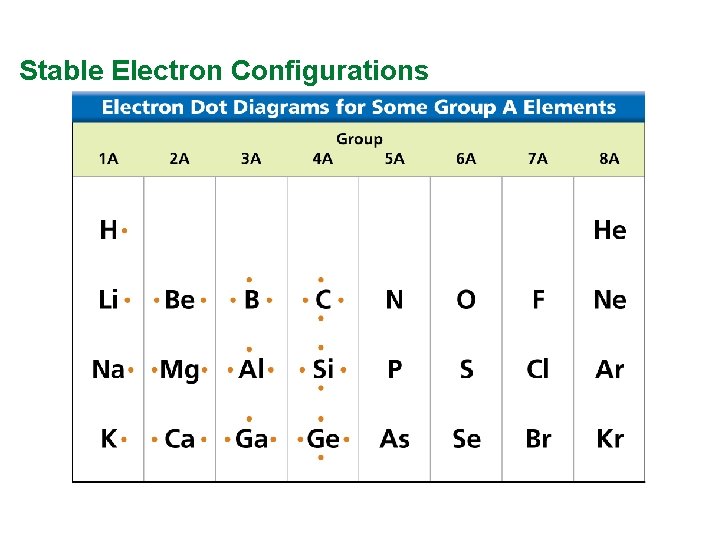
Stable Electron Configurations
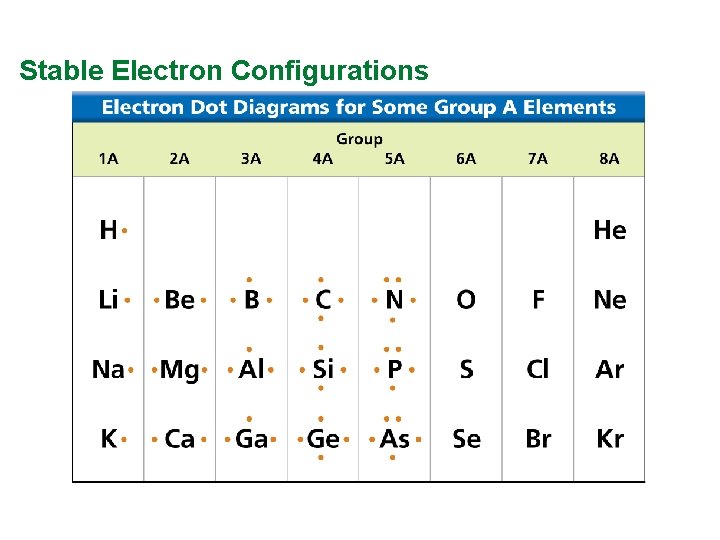
Stable Electron Configurations
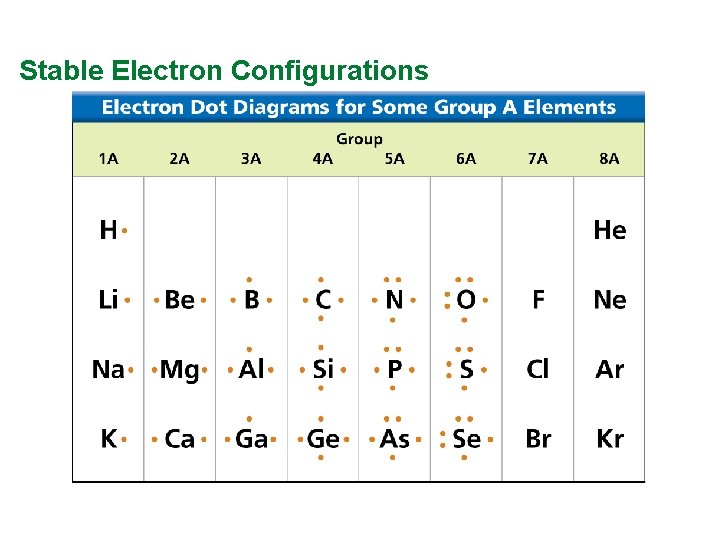
Stable Electron Configurations
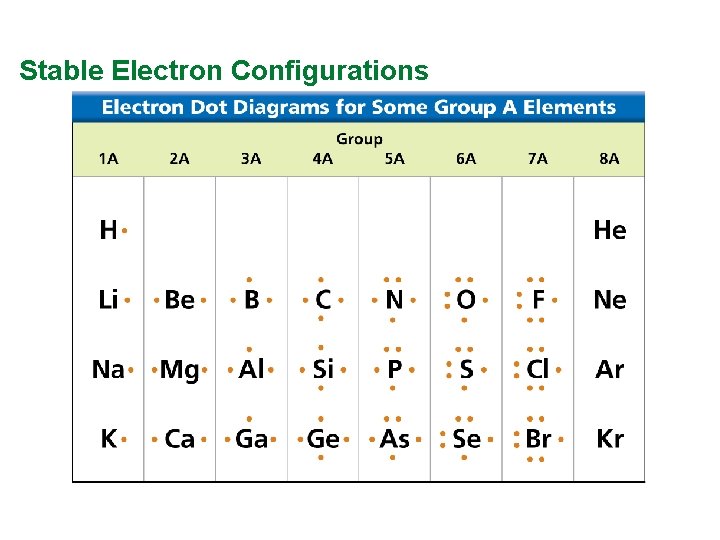
Stable Electron Configurations
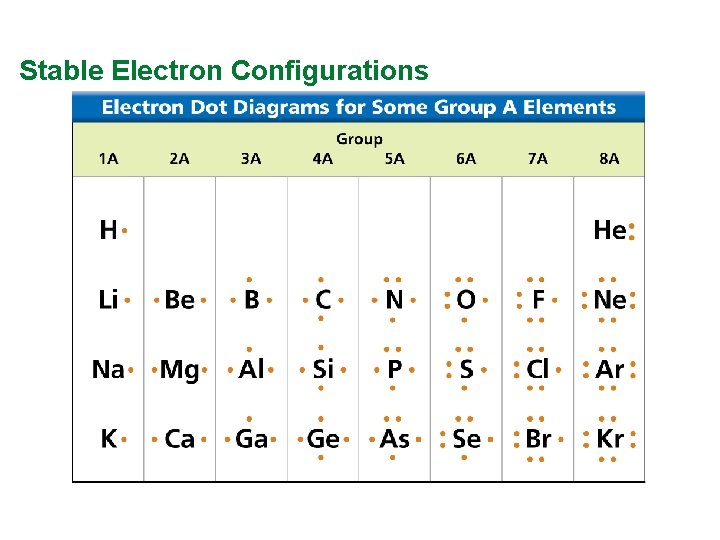
Stable Electron Configurations
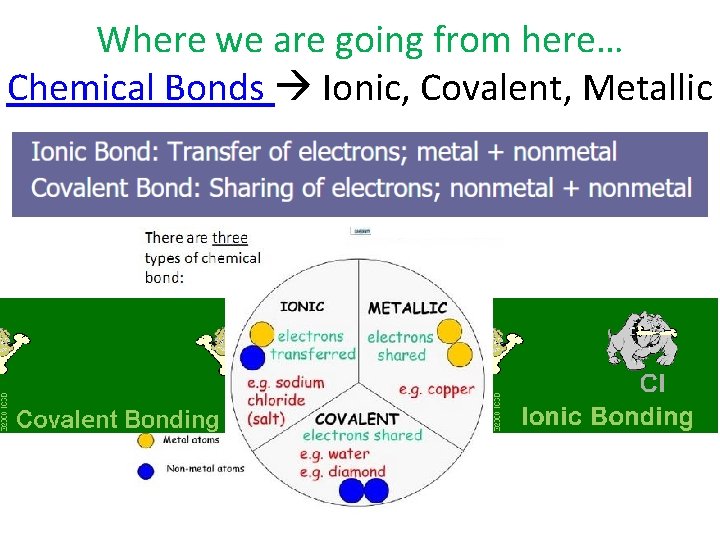
Where we are going from here… Chemical Bonds Ionic, Covalent, Metallic

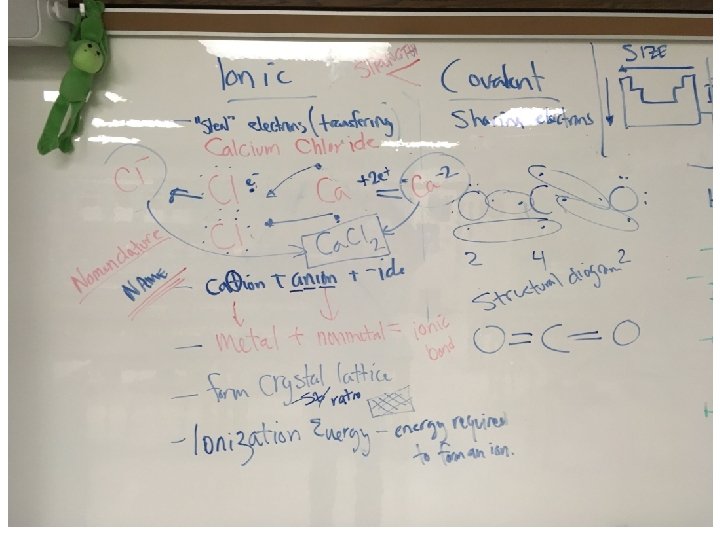
Ionic Bonding • Relationships are weaker when you – take/borrow/steal… • It means someone had to – give/lend/be robbed …Where the analogy breaks down… – Who is left feeling more negative? – Who is more positive? • What happens when negatives and positive are near each other?
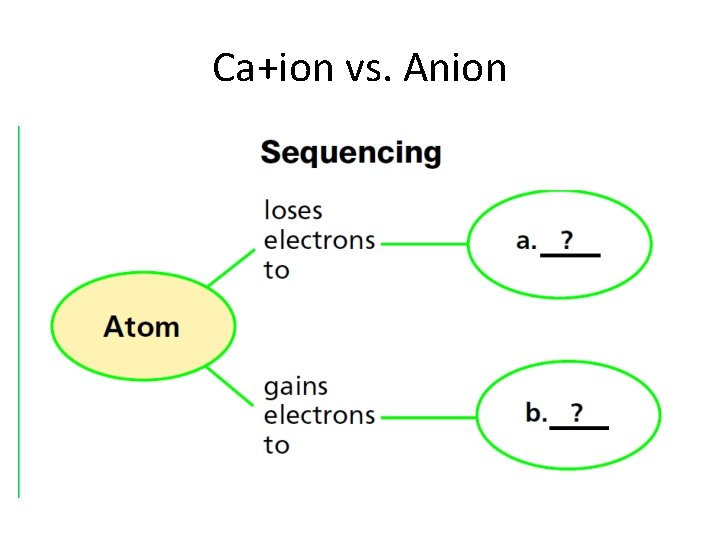
Ca+ion vs. Anion
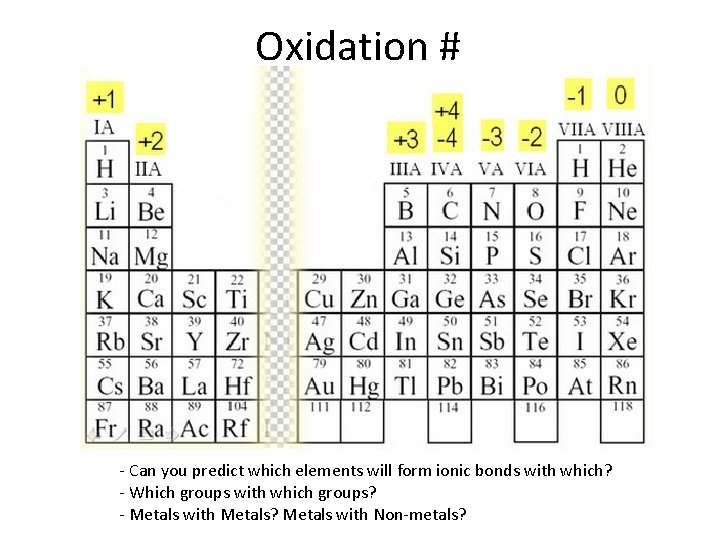
Oxidation # - Can you predict which elements will form ionic bonds with which? - Which groups with which groups? - Metals with Metals? Metals with Non-metals?

Mg. Cl 2 RATIO = 1: 2 • Lattice shape depends on the arrangement of the atoms. • 2 Factors: Size of atoms and ratio of elements
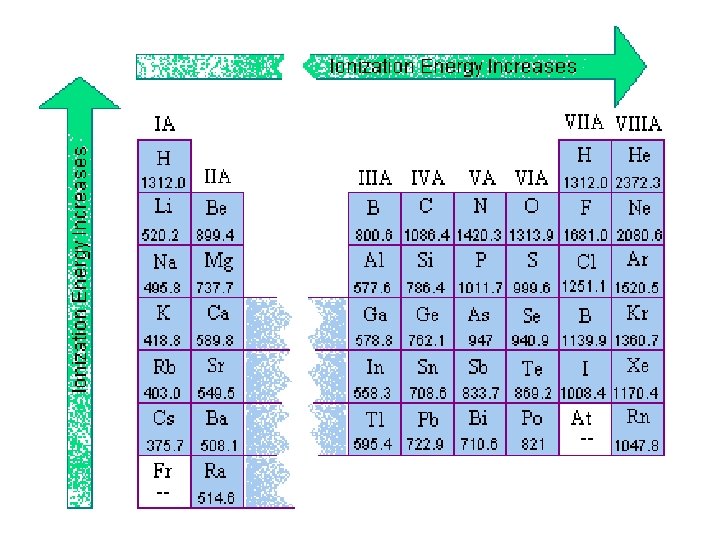

IONIC BONDS
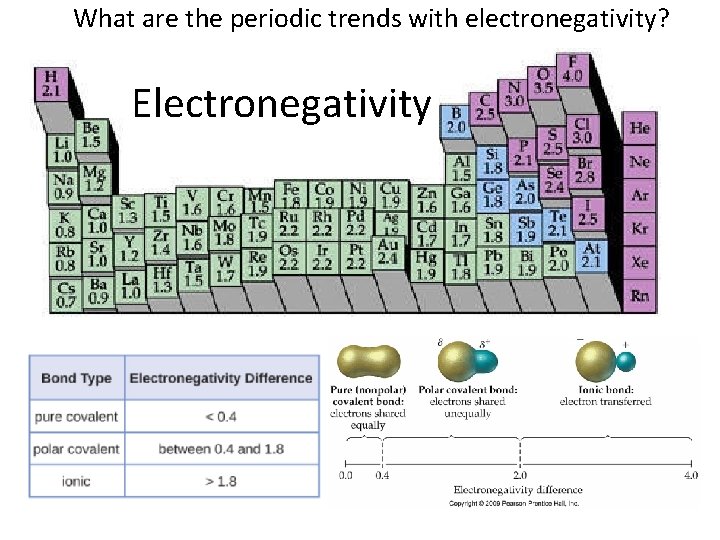
What are the periodic trends with electronegativity? Electronegativity
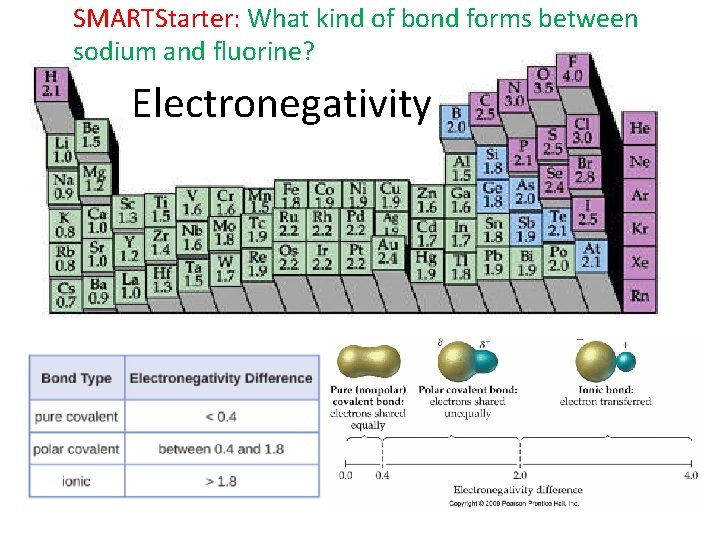
SMARTStarter: What kind of bond forms between sodium and fluorine? Electronegativity
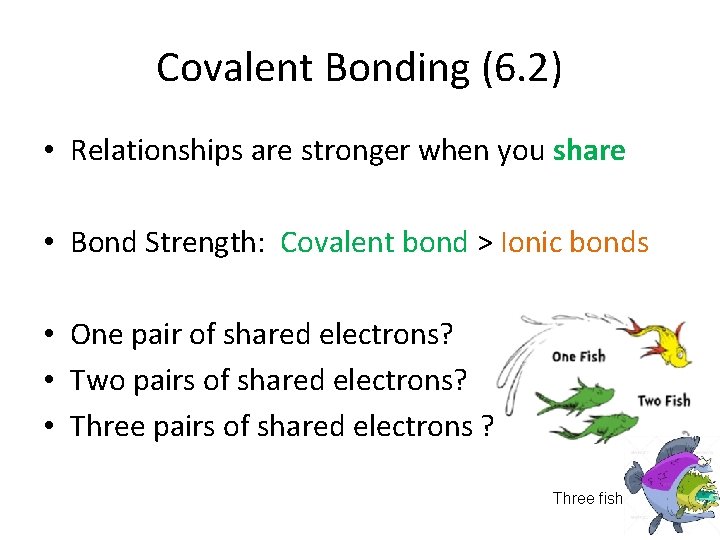
Covalent Bonding (6. 2) • Relationships are stronger when you share • Bond Strength: Covalent bond > Ionic bonds • One pair of shared electrons? • Two pairs of shared electrons? • Three pairs of shared electrons ? Three fish
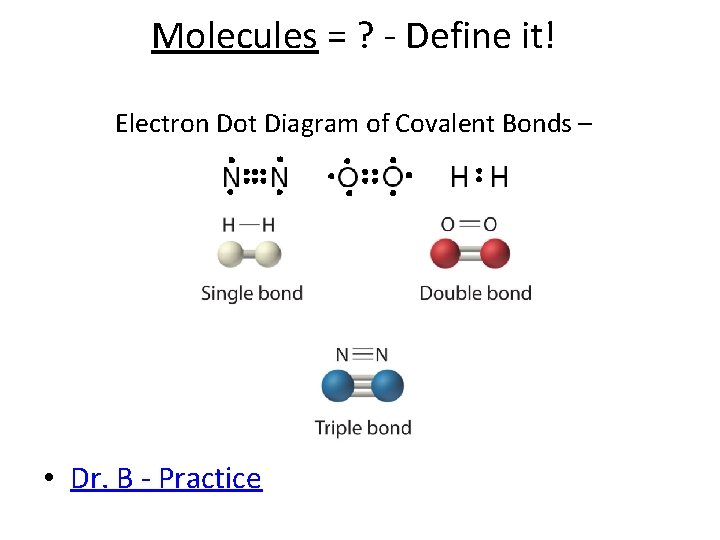
Molecules = ? - Define it! Electron Dot Diagram of Covalent Bonds – • Dr. B - Practice

Polar Covalent Bond Share…but not equally Results in partial charges = polarity EXAMPLE: Water
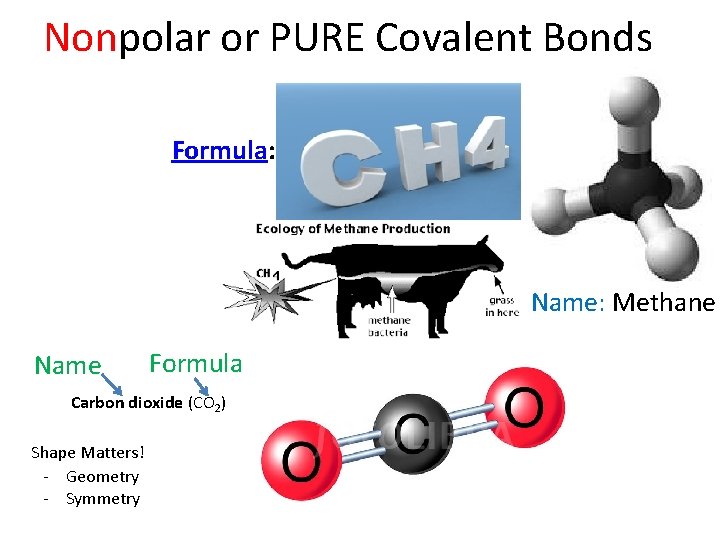
Nonpolar or PURE Covalent Bonds Formula: CH 4 Name: Methane Name Formula Carbon dioxide (CO 2) Shape Matters! - Geometry - Symmetry

Molecular Compound – bonded molecules • Forces of attraction that hold molecules together in a liquid or solid - Stronger in polar or nonpolar molecules? Ex. Hydrogen bonds (Van der Waals Forces)

POLYATOMIC IONS: A polyatomic ion, also known as a molecular ion, is a charged chemical species (ion) composed of two or more atoms covalently bonded or of a metal complex that can be considered to be acting as a single unit. The prefix poly- means "many, " in Greek, but even ions of two atoms are commonly referred to as polyatomic -ite and -ate Some polyatomic anions contain oxygen. When an element forms two oxygen containing anions they are named with different suffixes/endings: -ite = the one with less oxygen -ate = the one with more oxygen Examples: NO 2 - Nitrite NO 3 - Nitrate SO 32 - Sulfite SO 42 - Sulfate

Nomenclature -- How do you name or talk about compounds and molecules? – Formulas - describes the ratio of ions in the compound Formula Name – words to describe/identify it. Carbon dioxide (CO 2) Binary compound = exactly two different elements Metal + Nonmetal = ? Nonmetal + Nonmetal = ? Metal + Metal = ? Examples: calcium chloride (Ca. Cl 2), sodium fluoride (Na. F), and magnesium oxide (Mg. O) water (H 2 O), and sulfur hexafluoride (SF 6)

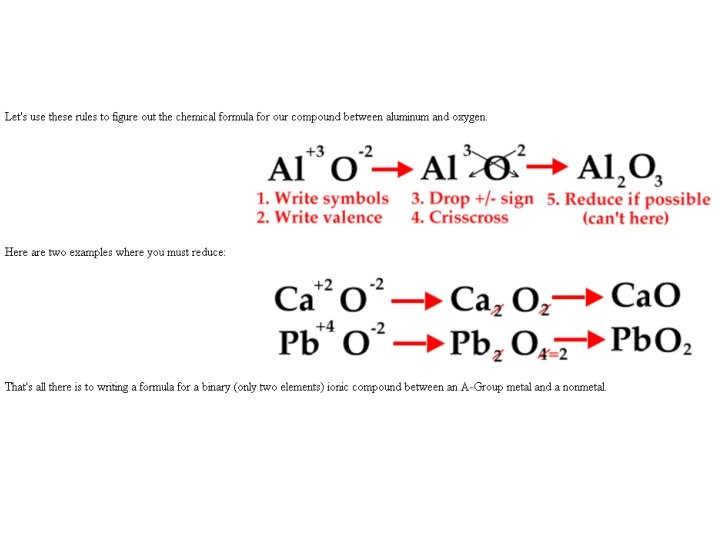
Talking Ionically…Formula When writing an ionic FORMULA between a metal and a nonmetal follow these 5 steps: 1. 2. 3. 4. 5. Write the symbols for the metal and the nonmetal. Write the valences as superscripts above each symbol. Drop the + and - sign. Crisscross the valences so they become the subscript for the other element. Reduce subscripts whenever possible. Only when both are divisible by a number greater than one.
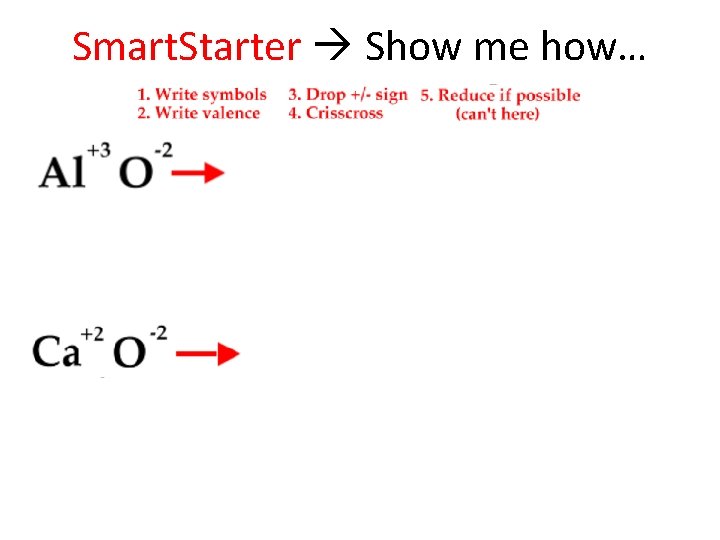
Smart. Starter Show me how…
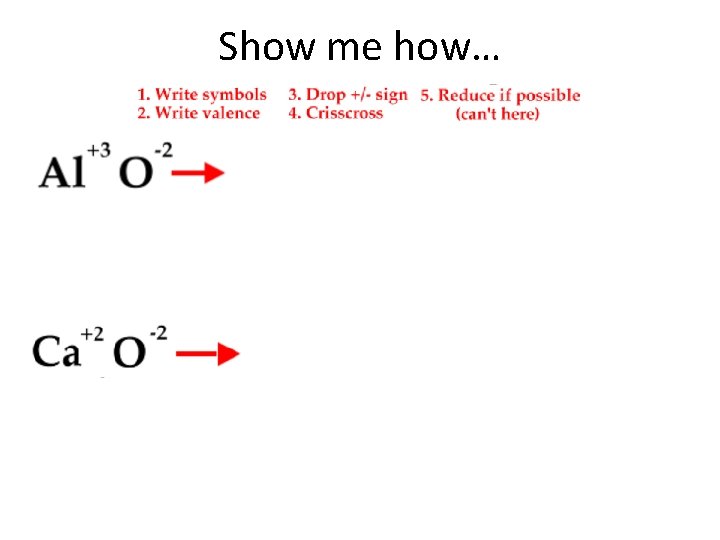
Show me how…

Pattern/Rules for Naming Ionic Compounds: NAME… name of cation + name of anion + “-ide” Ionic Compounds - Naming of anions -ide The -ide ending is added to the name of a monoatomic ion of an element. H- Hydride F- Fluoride O 2 - Oxide S 2 - Sulfide N 3 - Nitride P 3 - Phosphide

Practice Using a periodic table & oxidation numbers find the formula and name of these ionic compounds: 1. 2. 3. 4. 5. 6. 7. 8. 9. magnesium and fluorine aluminum and sulfur Nitrogen and calcium* barium and iodine bromine and potassium* aluminum and phosphorus strontium and oxygen nitrogen and magnesium* cesium and phosphorus • Check your answers

When the metal (ca+ion) is a Transition Metal… • More than one type of ions possible • • A Roman numeral in parentheses, following the name of the element, is used for elements that can form more than one positive ion. This is usually seen with metals. You can use a chart to see the possible valences for the elements. • Fe 2+ Iron (II) Cu+ Copper (I) Fe 3+ Iron (III) Cu 2+ Copper (II) • Use Roman Numerals to indicate the charge – Ex. Copper (II) ion + Chlorine Cu 2+ + __Cl- Cu. Cl? Cu+ Cuprous Cu 2+ Cupric
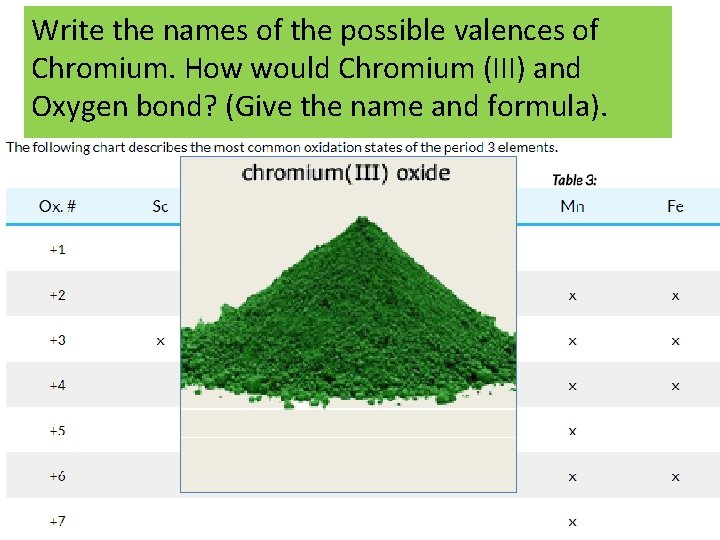
Write the names of the possible valences of Chromium. How would Chromium (III) and Oxygen bond? (Give the name and formula).

Practice Using a periodic table try these then check your answers. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. iron(III) and phosphorus copper(II) and fluorine silver(I) and sulfur manganese(II) and nitrogen lead(IV) and iodine copper(II) and bromine gold(II) and phosphorus lead(IV) and oxygen silver(I) and nitrogen copper(II) chloride Check your answers

Compounds containing POLYATOMIC IONS Polyatomic Ion: covalently bonded group of atoms that has a positive or negative charge and acts as a unit When you write formulas for compounds containing a polyatomic ion: 1. Write the symbols for the metal and the polyatomic ion. * 2. Write the valences as superscripts above each. 3. Drop the + and - sign. 4. Crisscross the valences so they become the subscript for the other element. 5. If you have more than one of the polyatomic ion, you must encase it in parenthesis and place the subscript outside. 6. You can only reduce a subscript outside the parenthesis of a polyatomic ion - you cannot change the formula the polyatomic ion. *In the case of ammonium (the only polyatomic cation) you would write it first and then the anion
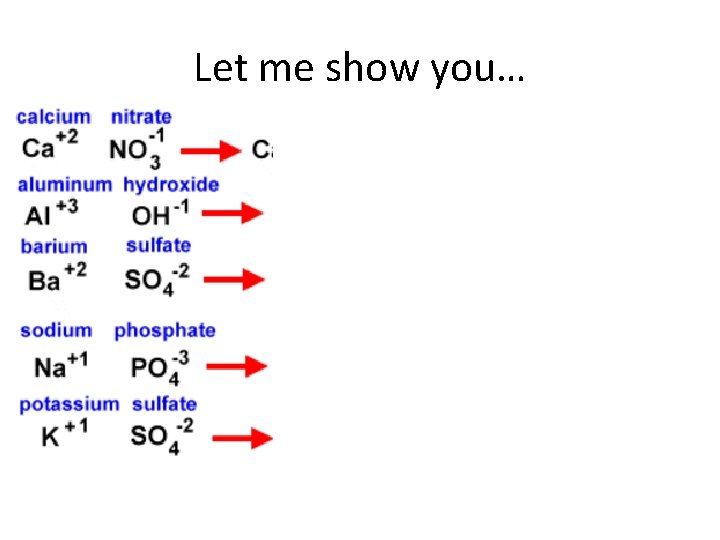
Let me show you…
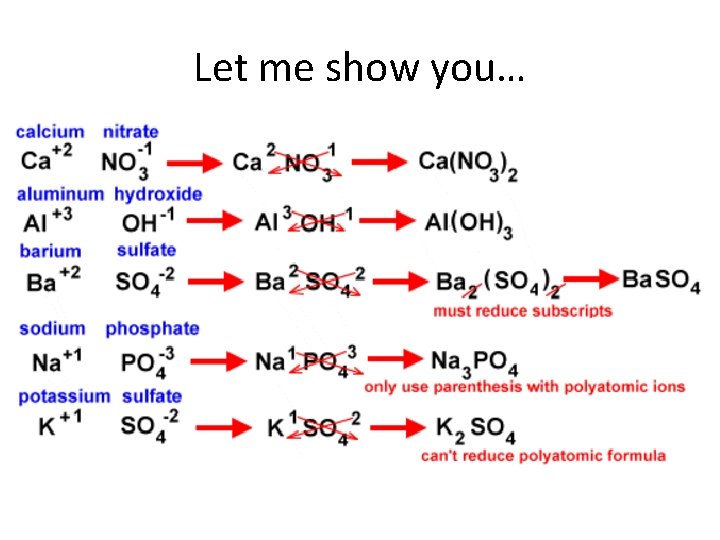
Let me show you…

Practice…You show me! Try these using a periodic table and a list of polyatomic ions. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. aluminum and sulfate ammonium and sulfur barium and hydroxide magnesium and phosphate lead(IV) and sulfite strontium and carbonate zinc(II) and phosphate ammonium and oxygen calcium and nitrate tungsten(II) and sulfate Check Answers

Covalent (Molecular) Compounds

COVALENT FORMULAS: TWO NONMETALS Because covalent compounds share electrons they can share in different ways and can form many compounds between the same two elements. IMPORTANT: You never crisscross valences to determine covalent (two nonmetals) formulas. So just how do you write the formulas? Prefixes - that's how. Here are six covalent compounds that form between nitrogen and oxygen: nitrogen monoxide = NO nitrogen dioxide = NO 2 dinitrogen oxide = N 2 O dinitrogen trioxide = N 2 O 5 dinitrogen tetroxide = N 2 O 4 dinitrogen pentoxide = N 2 O 5

NAMES *Lowest electronegativity goes first or Left to right on the P-Table

Practice – GIVE THE FORMULA Just look at the name and you have the formula for a covalent compound! Go ahead and try these: 1. 2. 3. 4. 5. sulfur dioxide dihydrogen oxide phosphorus pentafluoride carbon tetrachloride aluminum trichloride Check your answers

6. 4 Metallic Bonds From this video you should learn: – How they form…. common pool of electrons – Metallic Lattice structure ? – How they give metal its strength…? – Alloys…combined properties

Kinesthetic Learning: Bonding Charades Four Types of Bonds = four possible answers Ionic Bonds Pure Covalent Bond Polar Covalent Bond Metallic Bonds Procedure: 1. 2. 3. 4. RULES: No noise No letters/words/numbers/Symbols Form your group of 6 -8 Get your assignment 3 minutes to plan your skit Perform when called on GROUP WHO IS IDENTIFIED FASTEST FOR EACH CATEGORY GETS A PRIZE!

Metallic Bonds • Bond that keeps metal atoms together is called metallic bond. In metals; – Number of valence electrons is smaller than number of valence orbitals. So, they have many empty valence orbitals. – Their ionization energies are small and they are weakly attracted by nucleus. – Valence electrons of metals can jump to the other atom's valence orbitals. This free movement of electrons makes metals good conductor of electricity and heat. All valence electrons of metals can behave like this. • P-TABLE TRENDS – In periodic table, as we go from top to bottom in metal group, strength of metallic bond and melting point decrease. – In periodic table, as we go from left to rşght in same period, strength of metallic bond and melting point increase.

Lab IDEA Ionic compounds have very high melting and boiling points, are soluble in water, and almost always conductive when dissolved in a solution or melted. Covalent compounds have low melting and boiling points (they melted almost immediately) and low conductivity. Sugar dissolves Salt dissolves

MORE Practice/Review • Quia. com
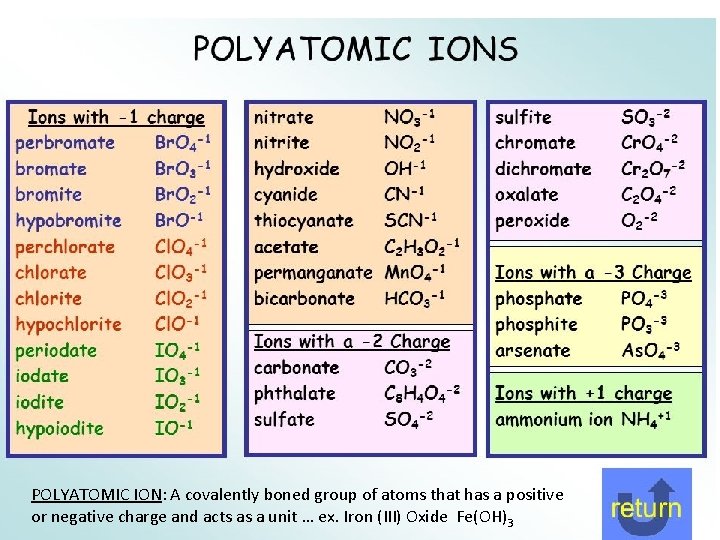
POLYATOMIC ION: A covalently boned group of atoms that has a positive or negative charge and acts as a unit … ex. Iron (III) Oxide Fe(OH)3
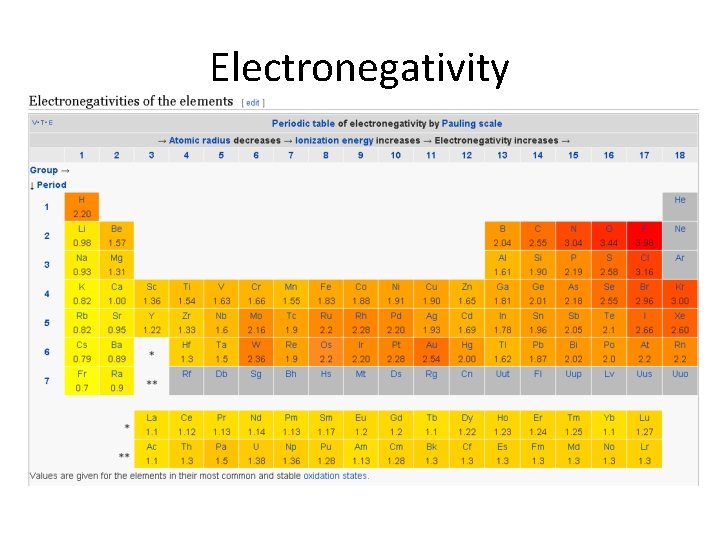
Electronegativity
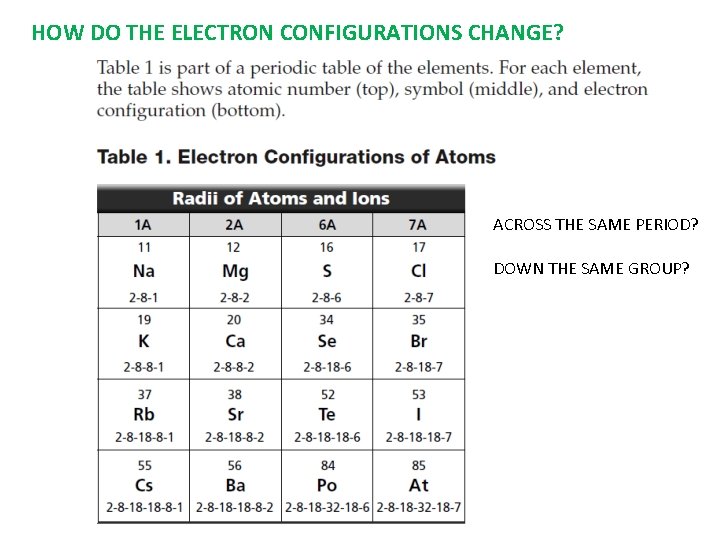
HOW DO THE ELECTRON CONFIGURATIONS CHANGE? ACROSS THE SAME PERIOD? DOWN THE SAME GROUP?

HOW DOES THE RADIUS OF AN ATOM CHANGE WHEN IT BECOMES AN ION? How does the radius change… When it becomes a cation? When it becomes an anion? Radius of the atom Charge of the ion (# electrons lost/gained) Radius of an ion of the atom Predict the radius change and # of valence electrons for Francium (Fr)

Not learned in this class…. but interesting: hypo- and per. In the case where there is a series of four oxyanions, the hypo- and per- prefixes are used in conjunction with the -ite and -ate suffixes. The hypo- and per- prefixes indicate less oxygen and more oxygen, respectively. Cl. O- Hypochlorite Cl. O 2 - Chlorite Cl. O 3 - Chlorate Cl. O 4 - Perchlorate bi- and di- hydrogen Polyatomic anions sometimes gain one or more H+ ions to form anions of a lower charge. These ions are named by adding the word hydrogen or dihydrogen in front of the name of the anion. It is still common to see and use the older naming convention in which the prefix bi-is used to indicate the addition of a single hydrogen ion. HCO 3 - Hydrogen carbonate or bicarbonate HSO 4 - Hydrogen sulfate or bisulfate H 2 PO 4 - Dihydrogen phosphate
- Slides: 59