Chemical Bonds Chapter 6 Chemical Bonding n Combining

Chemical Bonds Chapter 6

Chemical Bonding n Combining of elements to form new substances 1 st energy level holds 2 electrons 2. 2 nd and 3 rd energy levels hold 8 electrons v Valence electrons = electrons found in the outermost energy level 1.

Electron Configuration n Refers to the arrangement of the valence (outer) electrons in an atom. n The group number usually tells us how many valence electrons are present in an atom n Example: magnesium (group IIA)

Electron-Dot diagrams: n. A shorthand method to show valence electrons.

n When the outermost energy level is complete, or full, the atom is considered happy or satisfied. It is more stable.

“Happy” atoms The inert or noble gases all have a complete outer shell. Group VIIIA They are unreactive for the most part. Very stable
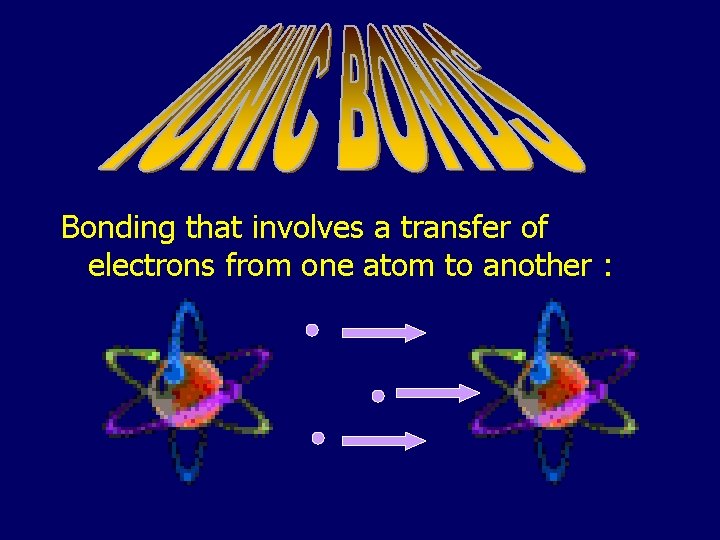
Bonding that involves a transfer of electrons from one atom to another :

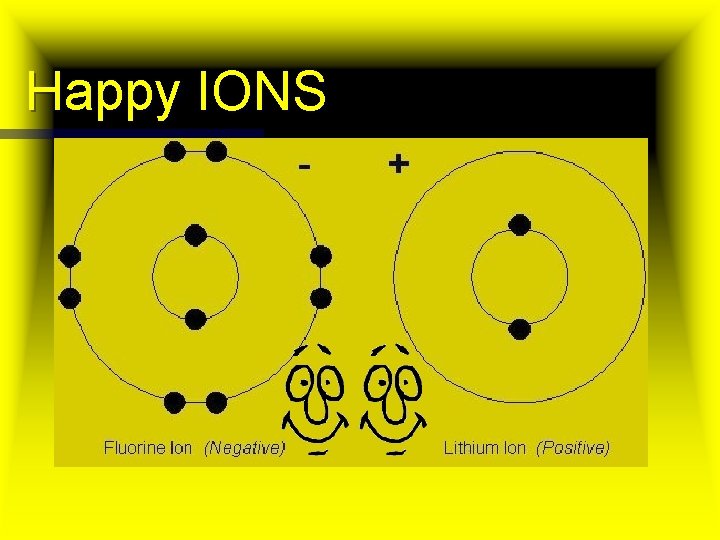
Happy IONS

Anions and Cations n Anion n Cation An ion with a negative charge positive charge n Negative ions have n Positive ions have GAINED electrons LOST electrons n Usually non-metals. n Usually metals. Left Right side of periodic table n

What happens when atoms lose electrons?

Ionization n The process of removing electrons and forming ions n Ionization energy = the energy needed to remove an electron n Different amounts of energy needed for different elements and different electrons of the same element.

Low and High Ionization Energies n LOW Ionization Energy n High ionization energy n Many valence Very few valence electrons : n Lose electrons easily n Gain electrons easily n

Crystal Lattice n Ions in an ionic compound are arranged in a specific way. A regular repeating pattern. n Very stable; High Melting points
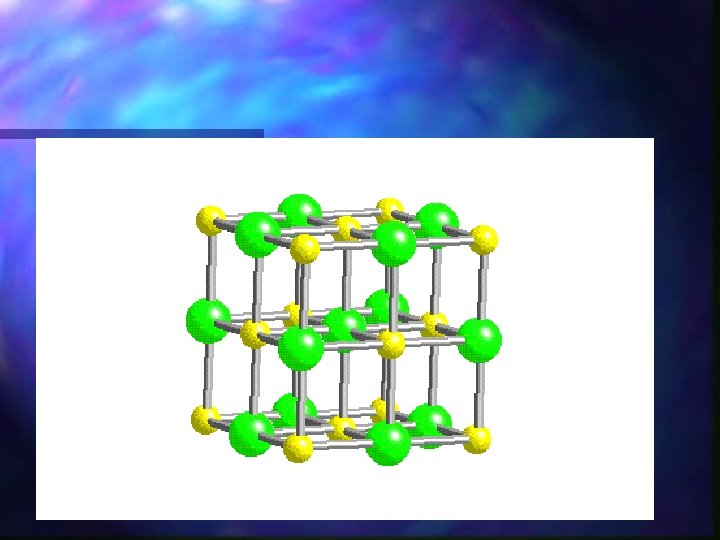

Properties of Ionic Compounds n Usually very high melting points n Good conductors of electricity when dissolved in water n Ionic crystals often shatter when struck.

Bonding in which electrons are shared. v each atom will still attempt to fill its outermost energy level v The positively charge nucleus of each atom simultaneously attracts the negatively charged electrons being shared. v


Covalent Bonds between more than two atoms:
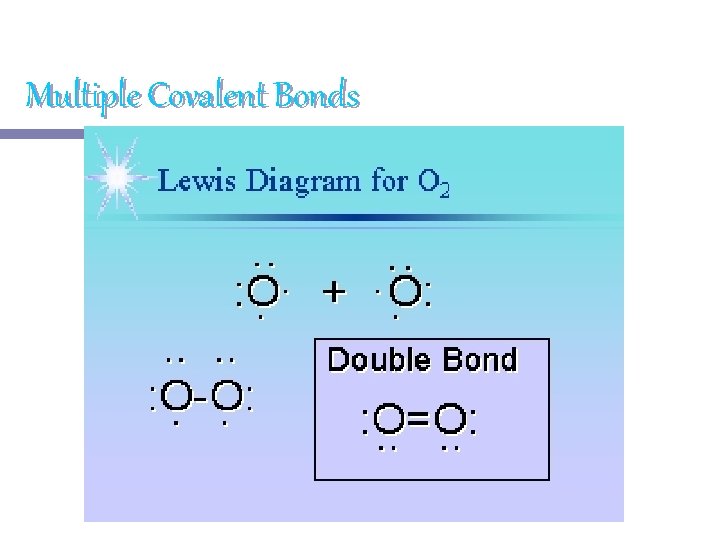
Multiple Covalent Bonds
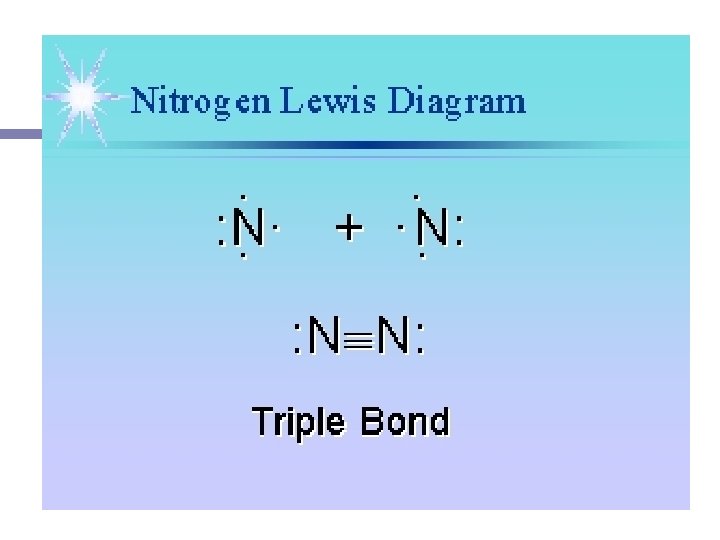

n Elements that exist in nature as two identical atoms covalently bonded to each other (know these seven!!!) n Equal sharing.

Molecules Ø The smallest piece of a covalently bonded substance Ø Very low melting points Ø Only a small amount of energy is needed to separate molecules from each other. Ø examples: (on white board)

Unequal sharing of electrons n In general, elements on the right tend to have a greater attraction for electrons. n Because of this, they “want” the electrons more. They are “stronger” in the electron tug-of-war.

Polar covalent bonds n Occurs when an atom has a greater attraction for the shared electrons than the other atom involved in the bond n Results in partial positive and partial negative charges ( + and -)


What determines if a compound is polar or non-polar? The types of atoms 2) The shape of the molecule 1. Example: 1)

Naming ionic compounds n Binary compounds: n Only consists of two elements. n The name of the cation followed by the name of the anion. n Example: sodium chloride (Na. Cl) n Not sodium chlorine n See page 171

Some metal ions have more than one charge. n Examples on page 172 n A Roman numeral is placed after the name of the metal to identify the charge.

Polyatomic ions n. A covalently bonded group of atoms that act like a single ion with a net charge. n P. 173

Naming molecular compounds n Often will use Greek prefixes. (figure 20) n Examples: n SO 3 n CO 2 n P 4 S 10

Metallic Bonds n. A special type of covalent bond where the nuclei of metal atoms are surrounded by a “sea’ of electrons n The mobility of the electrons in a metal lattice explains some of the properties of metals

Alloys n. A solid solution of two or more metals.

Common alloys n Bronze n Brass n Steel n Stainless steel n Gold jewelery n Copper and tin n Copper and zinc n Iron and carbon n Iron and chromium n Gold, silver, nickel, copper, and zinc

Assignment: n Page 187 n 1 -10, 12, 15, 25 n Page 189 (1 -7)
- Slides: 35