Chemical Bonding Why do bonds form to lower
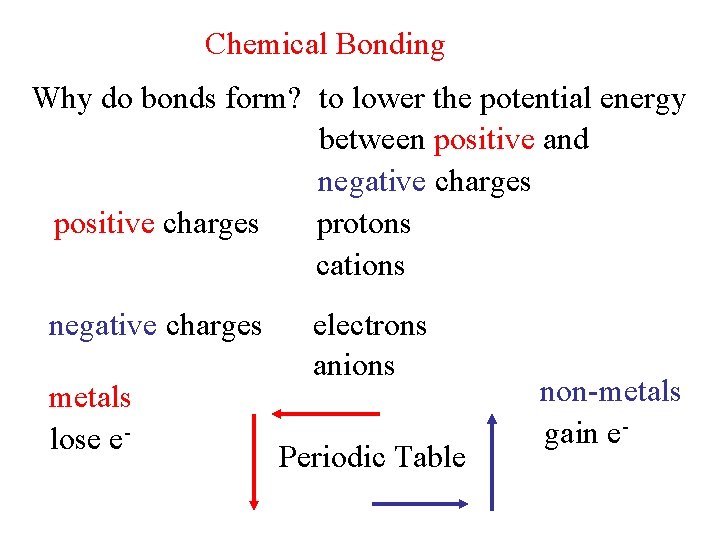
Chemical Bonding Why do bonds form? to lower the potential energy between positive and negative charges positive charges protons cations negative charges metals lose e- electrons anions Periodic Table non-metals gain e-

Lewis electron-dot symbols element symbol = nucleus + core eone “dot” = valence e- metals dot = e- it loses to form cation non-metal unpaired dot = e- paired through e- gain or sharing
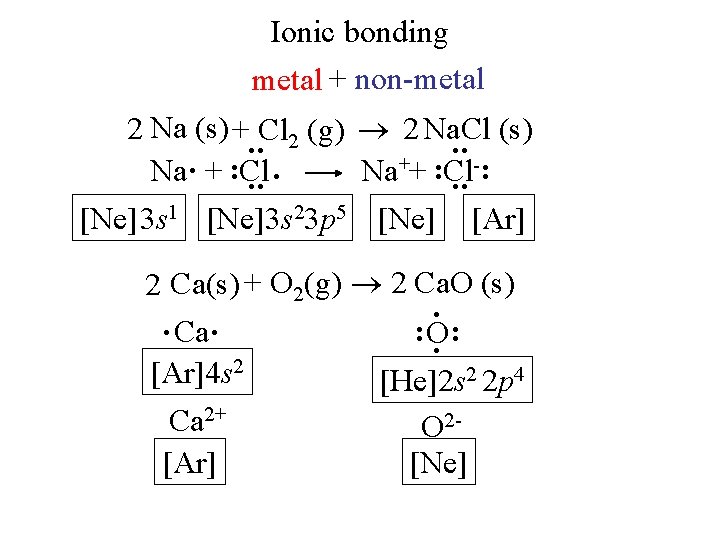
Ionic bonding metal + non-metal : : 2 Na (s) + Cl 2 (g) 2 Na. Cl (s) Na. + : Cl. Na++ : Cl-: [Ne] 3 s 1 [Ne]3 s 23 p 5 [Ne] [Ar] 2 Ca(s) + O 2(g) 2 Ca. O (s). . Ca. : O. : [Ar]4 s 2 [He]2 s 2 2 p 4 Ca 2+ O 2[Ar] [Ne]
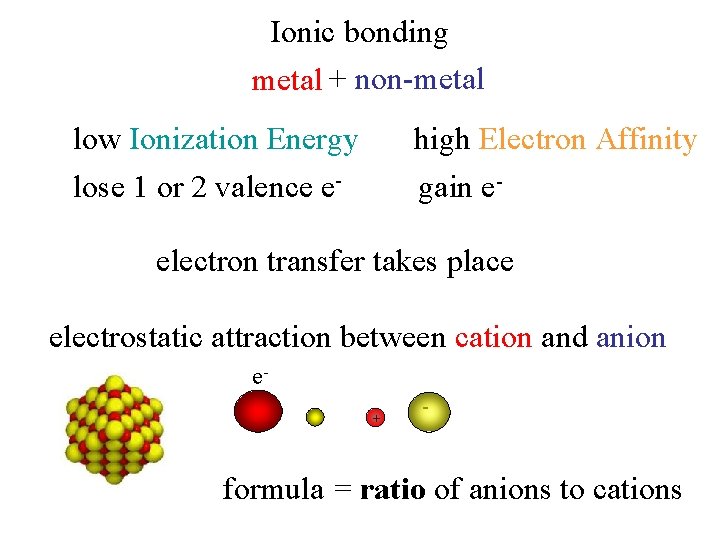
Ionic bonding metal + non-metal high Electron Affinity low Ionization Energy lose 1 or 2 valence e- gain e- electron transfer takes place electrostatic attraction between cation and anion e+ - formula = ratio of anions to cations
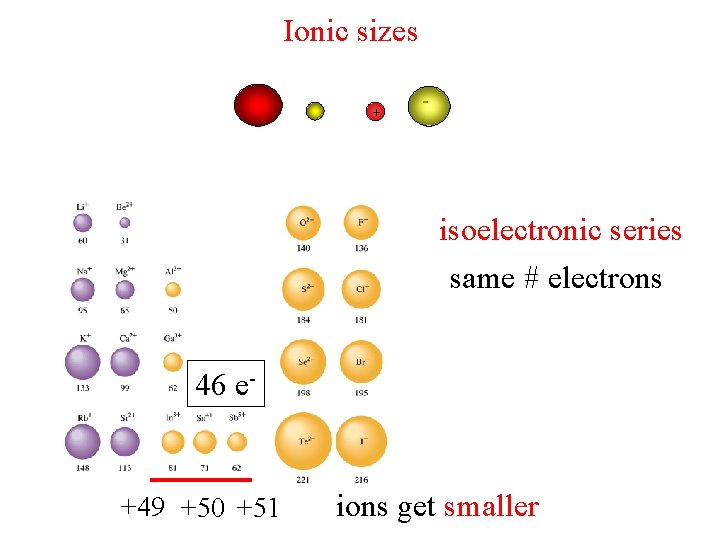
Ionic sizes + - isoelectronic series same # electrons 46 e- +49 +50 +51 ions get smaller

Ionic bonding metal + non-metal 2 Na (s) + Cl 2 (g) 2 Na. Cl (s)
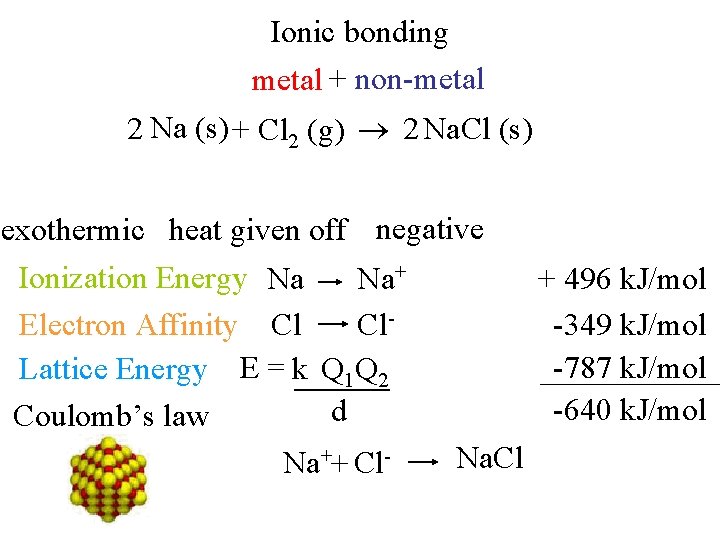
Ionic bonding metal + non-metal 2 Na (s) + Cl 2 (g) 2 Na. Cl (s) exothermic heat given off negative Ionization Energy Na Na+ Electron Affinity Cl Cl. Lattice Energy E = k Q 1 Q 2 d Coulomb’s law Na. Cl Na++ Cl- + 496 k. J/mol -349 k. J/mol -787 k. J/mol -640 k. J/mol
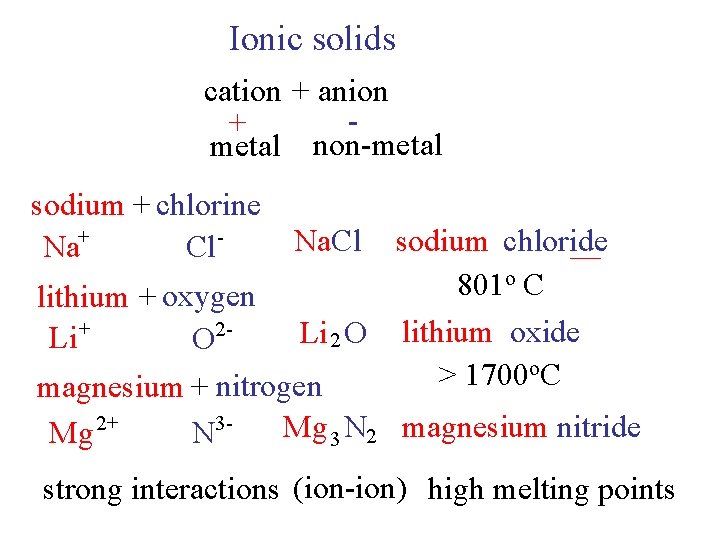
Ionic solids cation + anion + metal non-metal sodium + chlorine Na+ Cl - sodium chloride o. C 801 lithium + oxygen 2 Li 2 O lithium oxide Li + O o. C > 1700 magnesium + nitrogen Mg 3 N 2 magnesium nitride Mg 2+ N 3 Na. Cl strong interactions (ion-ion) high melting points
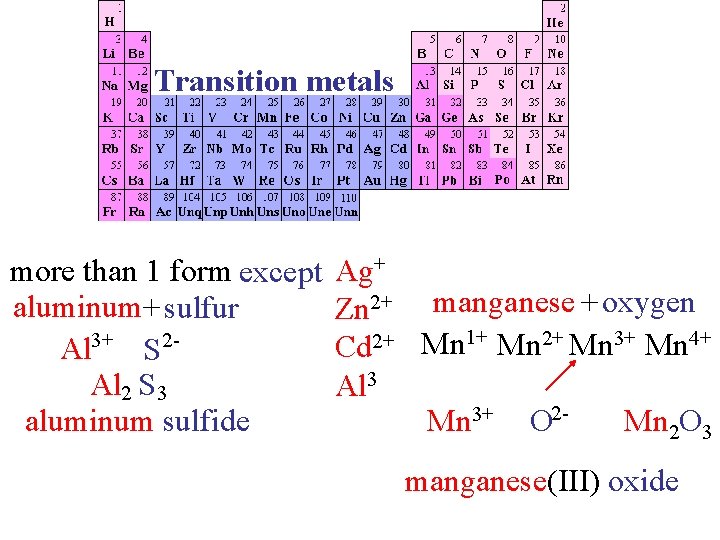
Transition metals more than 1 form except aluminum+ sulfur Al 3+ S 2 Al 2 S 3 aluminum sulfide Ag+ Zn 2+ manganese + oxygen Cd 2+ Mn 1+ Mn 2+ Mn 3+ Mn 4+ Al 3+ 23+ 4+ 2 Mn Mn 22 O 3 O Mn Mn. O O manganese(IV) oxide manganese(III) oxide
Covalent bonding non-metal + non-metal electrons shared between atoms high Ionization Energies high Electron Affinities electron density between the atoms distance between atoms = bond length formula = actual # atoms
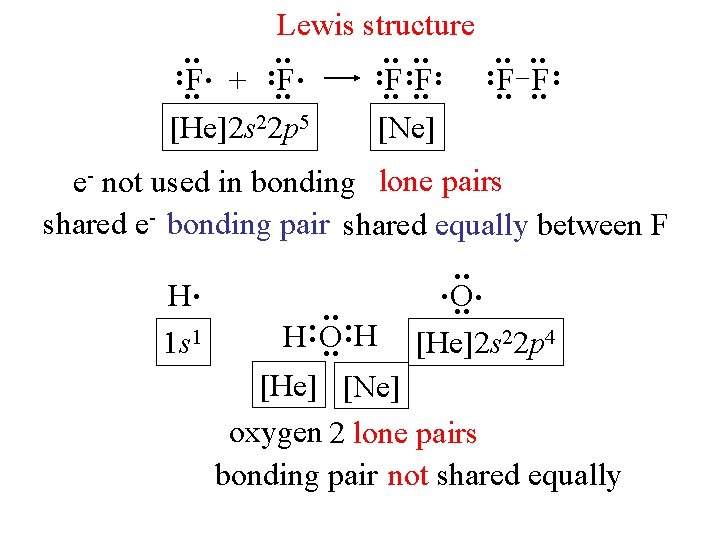
: : : : Lewis structure : F. + : F: F. . : F F : [He]2 s 22 p 5 [Ne] e- not used in bonding lone pairs shared e- bonding pair shared equally between F. O. : : . . H H O : : H. 1 s 1 [He]2 s 22 p 4 [He] [Ne] oxygen 2 lone pairs bonding pair not shared equally
![Covalent bonding non-metal + non-metal H. + H. H. . H [He] 1 s Covalent bonding non-metal + non-metal H. + H. H. . H [He] 1 s](http://slidetodoc.com/presentation_image/4c9e727decf2764f4a3fb1bbda9a34fa/image-13.jpg)
Covalent bonding non-metal + non-metal H. + H. H. . H [He] 1 s 1

Covalent compounds non-metal + non-metal share valence electrons = chemical bonds carbon + chlorine 1 mono 4+ Cl C 2 di 3 tri CCl 4 4 tetra carbon tetrachloride 5 penta nitrogen + oxygen ? N O 2 NO nitrogen monoxide 2+ 4+ NO 2 nitrogen dioxide N 2 O 4 dinitrogen tetroxide 4+ 6 hexa 7 hepta 8 octa
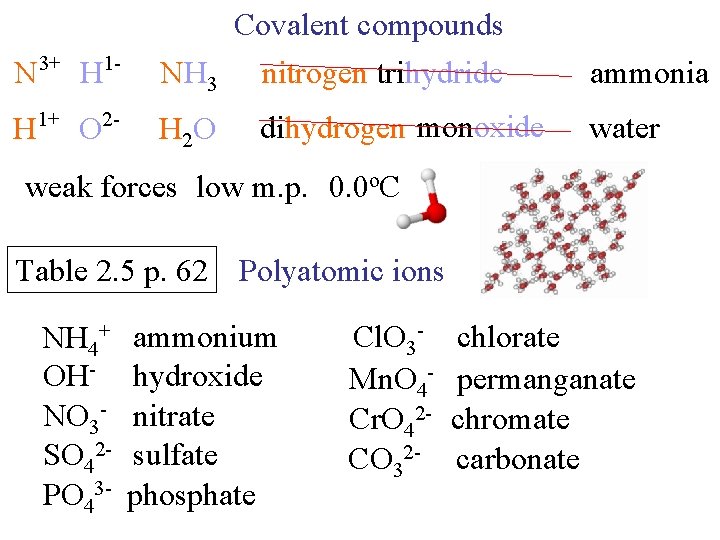
N 3+ H 1 - Covalent compounds NH 3 nitrogen trihydride H 1+ O 2 - H 2 O dihydrogen monoxide ammonia water weak forces low m. p. 0. 0 o. C Table 2. 5 p. 62 Polyatomic ions NH 4+ OHNO 3 SO 42 PO 43 - ammonium hydroxide nitrate sulfate phosphate Cl. O 3 Mn. O 4 Cr. O 42 CO 32 - chlorate permanganate chromate carbonate
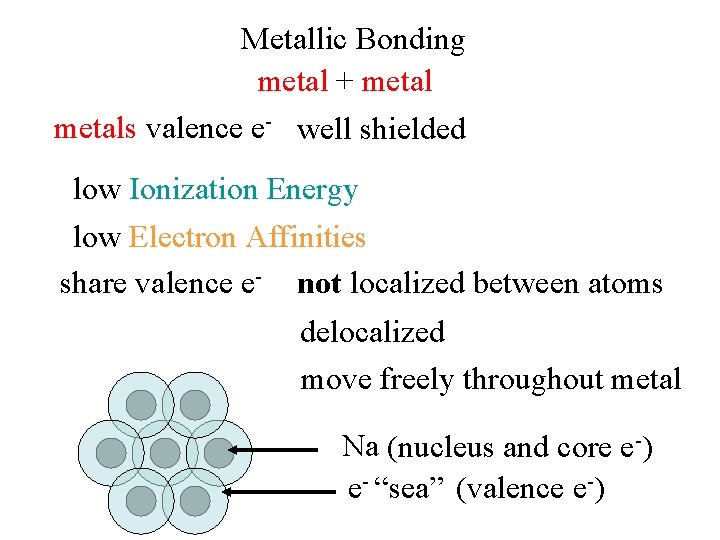
Metallic Bonding metal + metals valence e- well shielded low Ionization Energy low Electron Affinities share valence e- not localized between atoms delocalized move freely throughout metal Na (nucleus and core e-) e- “sea” (valence e-)
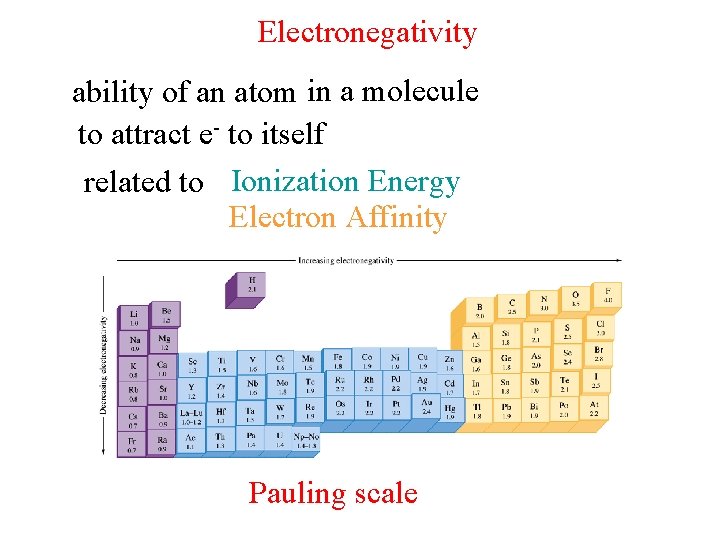
Electronegativity ability of an atom in a molecule to attract e- to itself related to Ionization Energy Electron Affinity Pauling scale
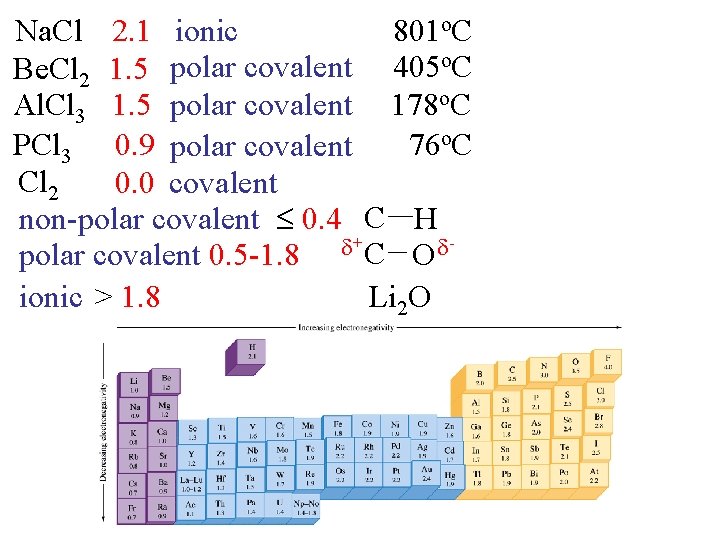
Na. Cl 2. 1 ionic 801 o. C Be. Cl 2 1. 5 polar covalent 405 o. C Al. Cl 3 1. 5 polar covalent 178 o. C PCl 3 0. 9 polar covalent 76 o. C Cl 2 0. 0 covalent non-polar covalent 0. 4 C H + C polar covalent 0. 5 -1. 8 O ionic > 1. 8 Li 2 O
- Slides: 18