Chemical Bonding Valence Bond VB Molecular Orbital MO

Chemical Bonding: Valence Bond (VB) & Molecular Orbital (MO) Theories Chapter 9 Section 4 through 6 of Jespersen 7 TH ed) Dr. C. Yau Spring 2015 1

VSEPR Theory • You had previously learned how to predict the molecular geometry of a species from examining its Lewis structure. • It utilizes the concept of repulsion amongst the charge clouds of the central atoms. • This was the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory). • It does not explain how a bond is formed and how bonding relates to the s, p, d orbitals. 2

Valence Bond Theory (VB Theory) The VB Theory explains bonding as an overlap of valence orbitals. H 2 bonds are due to the overlap of their 1 s valence orbitals. 1 s 1 s A B A. Two separate H atoms B. H 2 molecule with covalent bond due to overlap of the 1 s orbitals. 3
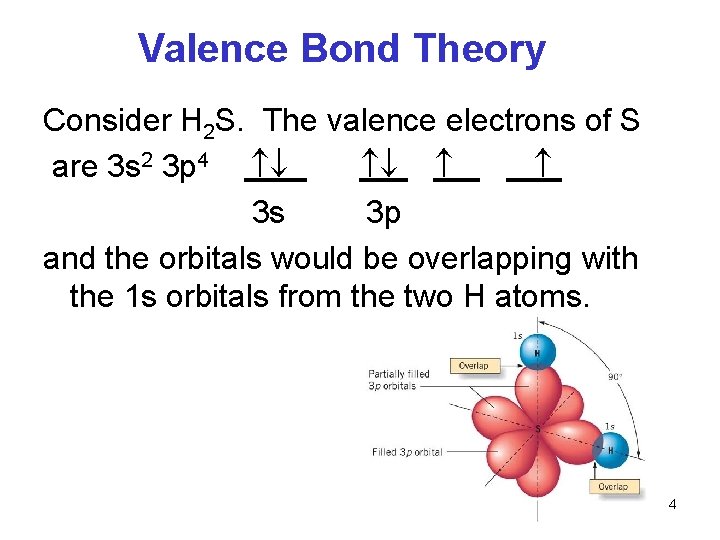
Valence Bond Theory Consider H 2 S. The valence electrons of S are 3 s 2 3 p 4 3 s 3 p and the orbitals would be overlapping with the 1 s orbitals from the two H atoms. 4

Difficulties With VB Theory • Most experimental bond angles do not support those predicted by mere atomic orbital overlap. • For example: C 1 s 22 p 2 and H 1 s 1 • One might expect C and H to form CH 2 with a bond angle of 90 o. Why might one expect that? • Experimental bond angles in methane CH 4 are in fact, 109. 5° and all angles are the same • p orbitals are 90° apart, and not all valence e- in C are in the p orbitals • Also, how can multiple bonds form? 5

Hybridization in VB Theory • Atomic orbitals are mixed to allow formation of bonds that have realistic bond angles • The newly mixed orbitals that result are called “hybrid orbitals” with specified shapes: Review: # charge clouds 2 3 4 5 6 Hybridization Bond Angles sp sp 2 sp 3 d 2 180 o 120 o 109. 5 o 120 o & 90 o & 180 o 6
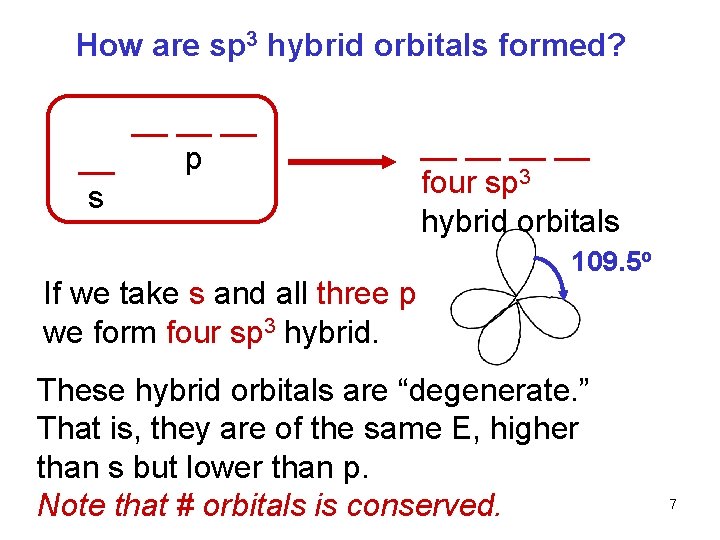
How are sp 3 hybrid orbitals formed? __ __ p s If we take s and all three p we form four sp 3 hybrid. __ __ four sp 3 hybrid orbitals 109. 5 o These hybrid orbitals are “degenerate. ” That is, they are of the same E, higher than s but lower than p. Note that # orbitals is conserved. 7
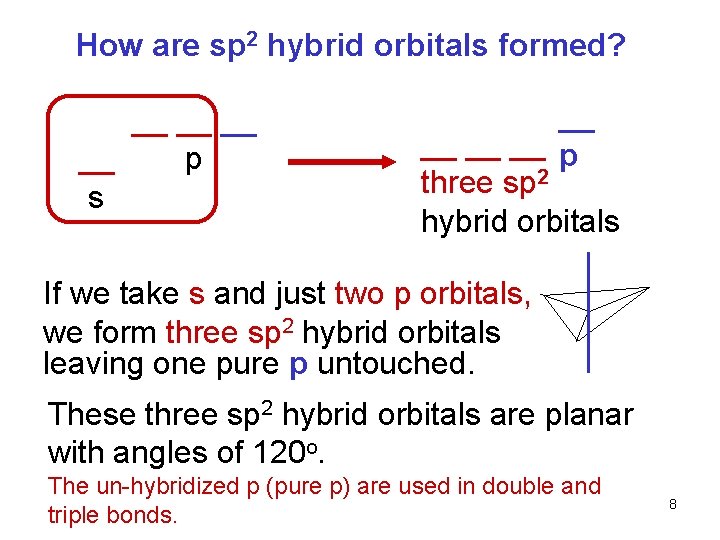
How are sp 2 hybrid orbitals formed? __ __ p s __ __ p three sp 2 hybrid orbitals If we take s and just two p orbitals, we form three sp 2 hybrid orbitals leaving one pure p untouched. These three sp 2 hybrid orbitals are planar with angles of 120 o. The un-hybridized p (pure p) are used in double and triple bonds. 8
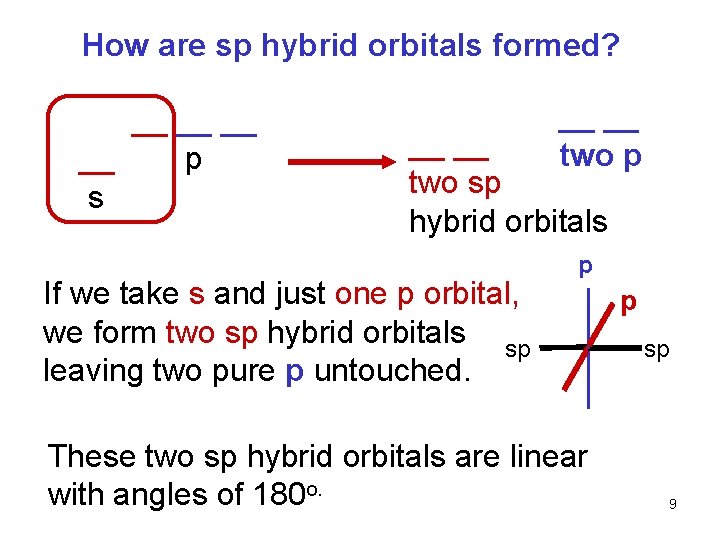
How are sp hybrid orbitals formed? __ __ p s __ __ two p __ __ two sp hybrid orbitals If we take s and just one p orbital, we form two sp hybrid orbitals sp leaving two pure p untouched. p These two sp hybrid orbitals are linear with angles of 180 o. p sp 9

How are hybrid orbitals formed? When we run out of p orbitals, we start using d orbitals, but this would not be possible for elements smaller than Ne (Period 2 and smaller) because they do not have dorbitals. Thus we have sp 3 d hybrid orbitals and sp 3 d 2 hybrid orbitals (but only for elements beyond Period 2). 10
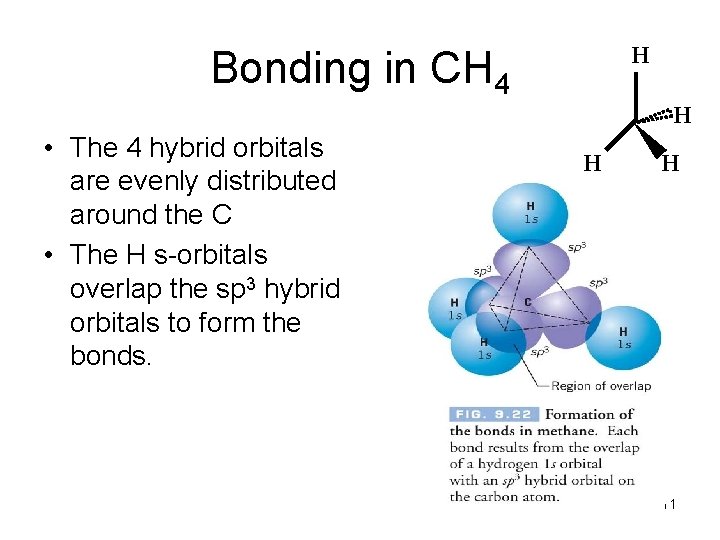
H Bonding in CH 4 H • The 4 hybrid orbitals are evenly distributed around the C • The H s-orbitals overlap the sp 3 hybrid orbitals to form the bonds. H H 11

Bonding Types • Two types of bonds result from orbital overlap: • sigma ( ) bonds – from head-on overlap – lie along the bond axis – account for the first bond • pi ( )bonds – pi bonds are perpendicular to bond axis – account for the second and third bonds in a multiple bond 12

Sigma and Pi Bonding • Please refer to lecture notes on and bonding of C 2 H 4 and C 2 H 2. • Here is what you should be able to do: • Given the structural formula of a compound, be able to specify the hybridization of each atom, state the bond angles and determine the # of sigma and pi bonds. 13

KNOW THIS WELL! X X single bond = bond X double bond = bond + bond X triple bond = bond + two bonds 14

1 2 3 4 Always start with hybridization. Give the hybridization of each C and each O. Give the bond angles. How many bonds are there? 9 bonds How many bonds are there? 3 bonds Ans. C 1 = sp C 2 = sp C 3 =sp 2 C 4 = sp 3 O = sp 3 C 1 -C 2 -C 3 = 180 o C 2 -C 3 -O = 120 o C 2 -C 3=O = 120 o 15 O-C 4 -H = 109. 5 o

Molecular Orbital Theory (MO Theory) Oh No! Yet another theory! VSEPR and VB theories do not explain everything. MO Theory considers the molecule as an entity rather than just a collection of atoms. Instead of considering atomic orbitals or hybrid orbitals, the MO theory considers molecular orbitals and molecular energy levels. 16

MO Theory • When two atomic orbitals overlap, their wave functions interact by constructive and destructive interference to form bonding and antibonding orbitals. • Electrons in bonding molecular orbitals stabilize the molecule. • Electrons in antibonding molecular orbitals destabilize the molecule. • The Bond Order is # bonds, and calculated thus: Bond order = ½ (# bonding electrons - # antibonding electrons). 17
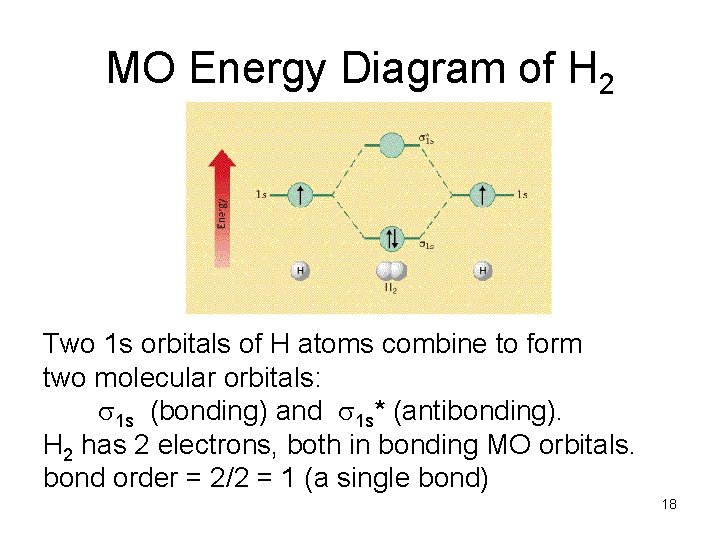
MO Energy Diagram of H 2 Two 1 s orbitals of H atoms combine to form two molecular orbitals: 1 s (bonding) and 1 s* (antibonding). H 2 has 2 electrons, both in bonding MO orbitals. bond order = 2/2 = 1 (a single bond) 18

MO Theory See handout for application of MO theory on diatomic molecules such as N 2, O 2 etc. Know how to fill electrons into the MO energy diagram and calculate bond order. 19
- Slides: 19