Chemical Bonding The Covalent Bond Covalent or Ionic

Chemical Bonding The Covalent Bond

Covalent or Ionic ? ? ? Electronegativity – the attraction that an atom has for the electrons that it is sharing w/ another atom n Scale devised by Nobel Prize winner Linus Pauling n Based on scale with fluorine assigned 4. 0, the highest value n Material from karentimberlake. com and H. Stephen Stoker

Forming Chemical Bonds • According to the Lewis model – an atom may lose or gain enough electrons to acquire a filled valence shell and become an ion. An ionic bond is the result of the force of attraction between a cation and an anion. – an atom may share electrons with one or more other atoms to acquire a filled valence shell. A covalent bond is the result of the force of attraction between two atoms that share one or more pairs of electrons. Material from karentimberlake. com and H. Stephen Stoker

Covalent Bonds Two nonmetal atoms form a covalent bond because they have less energy after they bonded H + H H : H = H 2 hydrogen molecule Material from karentimberlake. com and H. Stephen Stoker
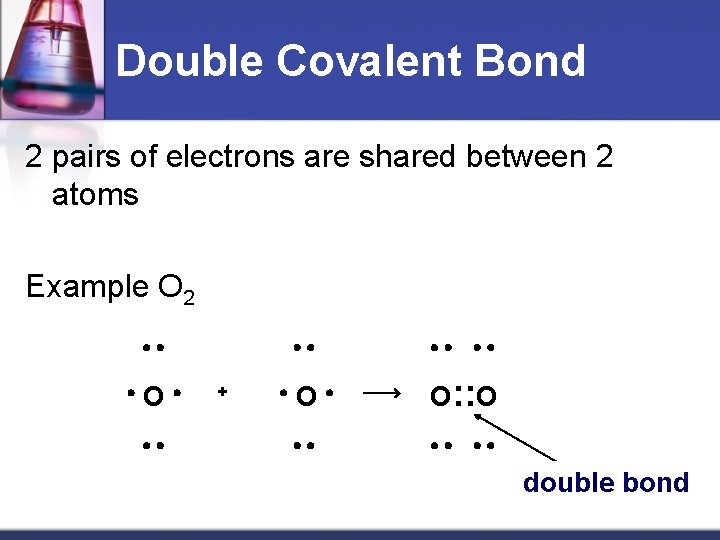
Double Covalent Bond 2 pairs of electrons are shared between 2 atoms Example O 2 O + O O: : O double bond Material from karentimberlake. com and H. Stephen Stoker
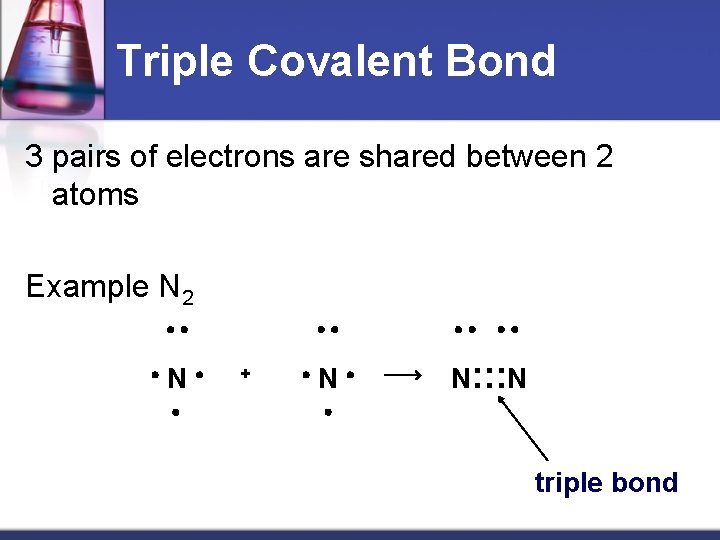
Triple Covalent Bond 3 pairs of electrons are shared between 2 atoms Example N 2 N + N N: : : N triple bond Material from karentimberlake. com and H. Stephen Stoker

Diatomic Elements n n Elements that are naturally in molecules with 2 atoms each. H, O, N, Cl, Br, I, F Existing as diatomic molecule yields a stable octet Gases that exist as diatomic molecules are H 2, F 2, N 2, O 2, Cl 2, Br 2, I 2 Material from karentimberlake. com and H. Stephen Stoker

Learning Check Use the name of the element to name the following diatomic molecules. H 2 hydrogen N 2 nitrogen Cl 2 ________ O 2 ________ I 2 Material from karentimberlake. com and H. Stephen Stoker ________

Solution Use the name of the element to name the following diatomic molecules. H 2 hydrogen N 2 nitrogen Cl 2 chlorine O 2 oxygen I 2 iodine Material from karentimberlake. com and H. Stephen Stoker

Lewis Structures Other molecules having single covalent bonds H 2 O The hydrogens share their electrons w/ oxygen so that O has 8 e- and each H has 2 e- Material from karentimberlake. com and H. Stephen Stoker
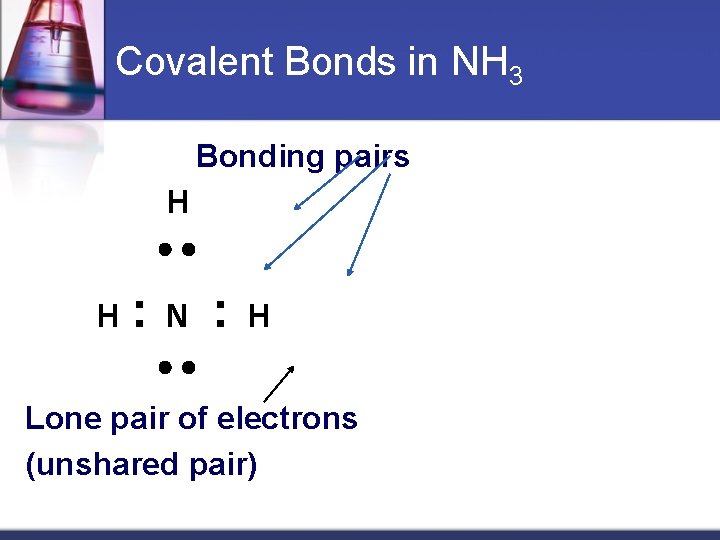
Covalent Bonds in NH 3 Bonding pairs H H : N : H Lone pair of electrons (unshared pair) Material from karentimberlake. com and H. Stephen Stoker

Chemical Bonding: The Covalent Bond Model ← © Bettman/CORBIS Fig. 5. 10 Linus Pauling received the Nobel Prize in chemistry in 1954 for his work on the nature of the chemical bond. Material from karentimberlake. com and H. Stephen Stoker

Bond Polarity: Nonpolar covalent bond l Electrons are shared between atoms with the same or very close electronegativity values. l Examples: N 2 Br 2 Material from karentimberlake. com and H. Stephen Stoker

Bond Polarity: Polar covalent bond l Electrons are shared between different nonmetal atoms; Examples: O-Cl O-S N-Cl Material from karentimberlake. com and H. Stephen Stoker

Differences in E-Negativity n Your covalent bond will be Non-polar if the e-negativity difference is between 0 &. 4 n Your covalent bond will be polar if the enegativity difference is between >. 4 < 1. 7
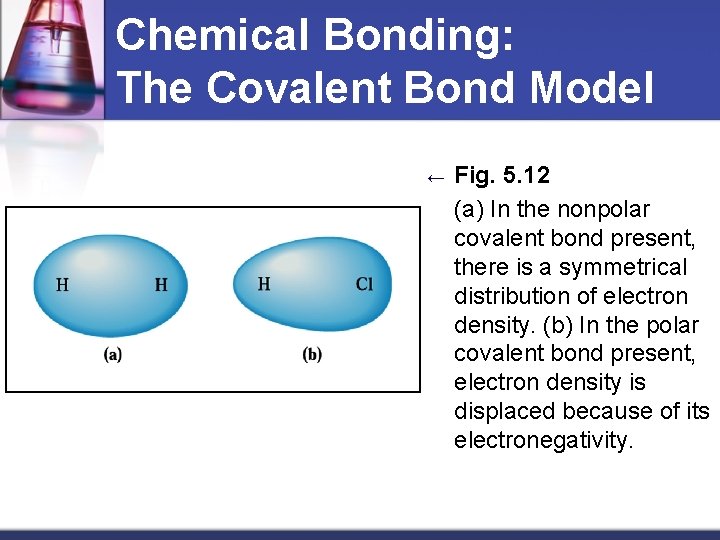
Chemical Bonding: The Covalent Bond Model ← Fig. 5. 12 (a) In the nonpolar covalent bond present, there is a symmetrical distribution of electron density. (b) In the polar covalent bond present, electron density is displaced because of its electronegativity. Material from karentimberlake. com and H. Stephen Stoker

Learning Check Identify the type of bond between the following atoms A. K-N 1) nonpolar 2) polar 3) ionic B. N-O 1) nonpolar 2) polar 3) ionic C. Cl-Cl 1) nonpolar Material from karentimberlake. com and H. Stephen Stoker

Solution A. K-N 3) ionic B. N-O 2) polar, covalent C. Cl-Cl 1) nonpolar, covalent Material from karentimberlake. com and H. Stephen Stoker
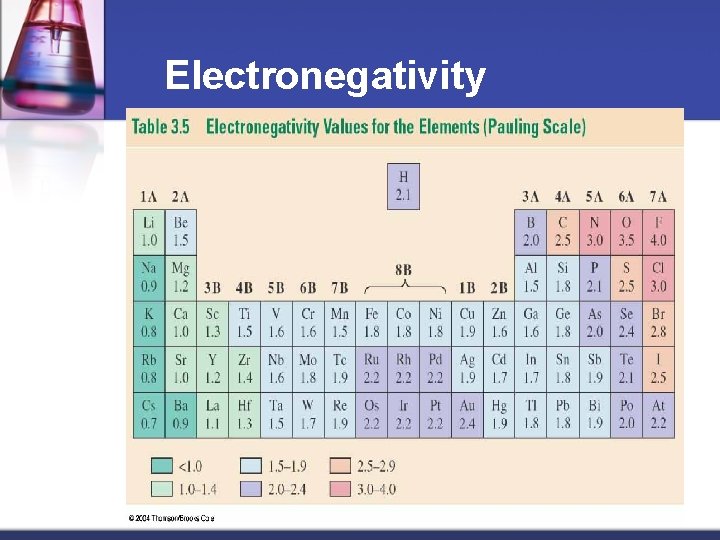
Electronegativity Material from karentimberlake. com and H. Stephen Stoker

Writing Formulas Nonmetal/Nonmetal n In covalent bonds, the element with the lowest electronegativity is written first Material from karentimberlake. com and H. Stephen Stoker
Chemical Bonding: The Covalent Bond Model Material from karentimberlake. com and H. Stephen Stoker

Chemical Bonding: The Covalent Bond Model Material from karentimberlake. com and H. Stephen Stoker

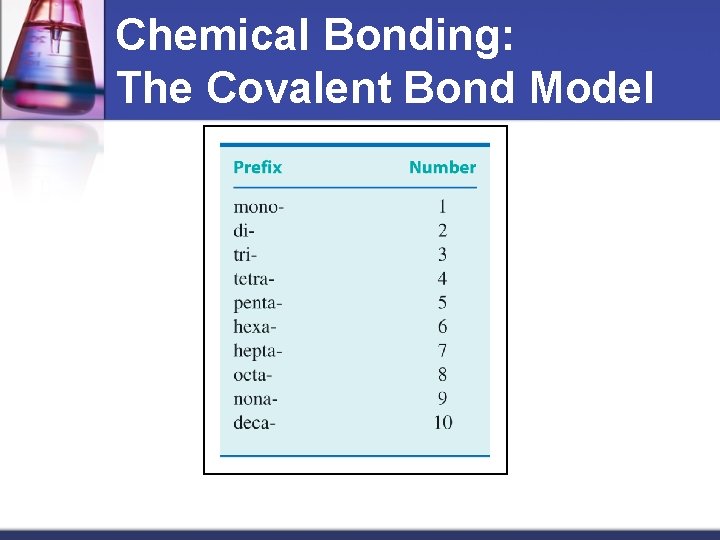
Naming of 2 Nonmetals 1. 2. 3. Name each element End the last element in –ide Add prefixes to show more than 1 atom Prefixes mon di tri tetra pent 1 2 3 4 5 hexa hepta octa nona deca 6 7 8 9 10 Material from karentimberlake. com and H. Stephen Stoker

Learning Check Fill in the blanks to complete the following names of covalent compounds. © Karen Timberlake CO carbon ______oxide CO 2 carbon ________ PCl 3 phosphorus _______chloride CCl 4 carbon ____chloride N 2 O _____nitrogen _____oxide Material from karentimberlake. com and H. Stephen Stoker

Learning Check A. B. C. P 2 O 5 Cl 2 O 7 Cl 2 Material from karentimberlake. com and H. Stephen Stoker
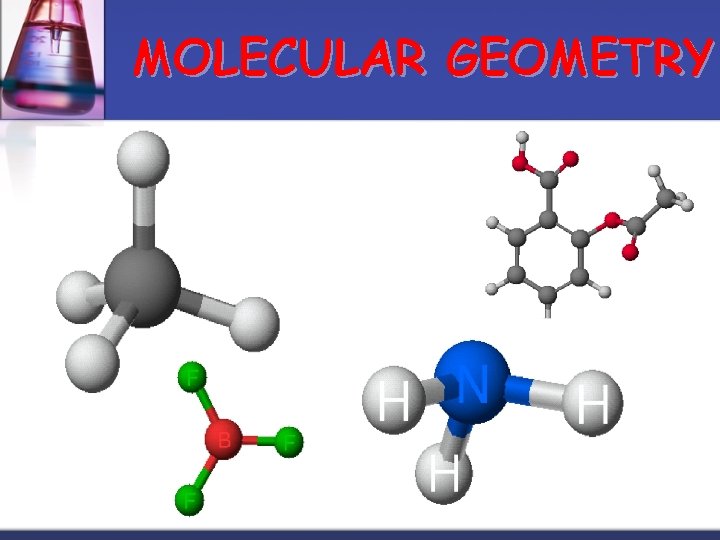
MOLECULAR GEOMETRY

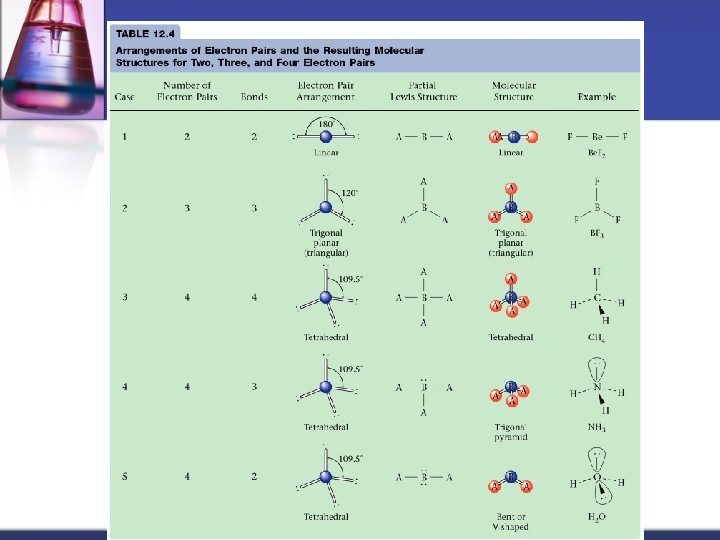
MOLECULAR GEOMETRY VSEPR n Valence Shell Electron Pair Repulsion theory. n Most important factor in determining geometry is relative repulsion between electron pairs. Molecule adopts the shape that minimizes the electron pair repulsions.
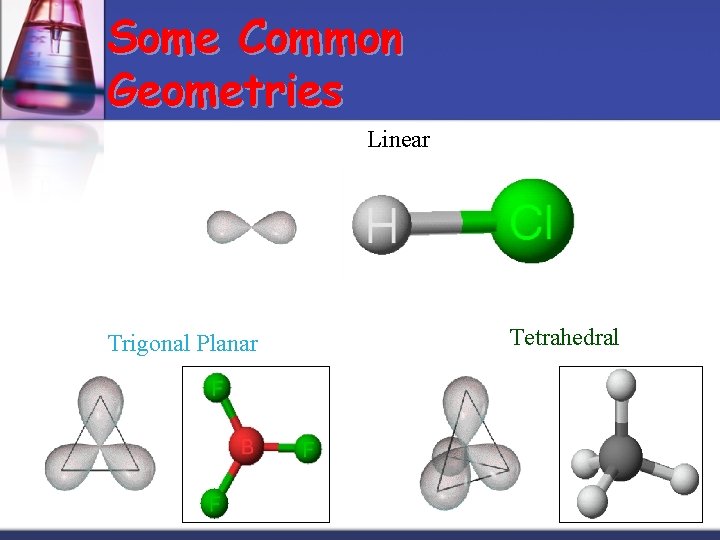
Some Common Geometries Linear Trigonal Planar Tetrahedral

VSEPR charts n n n Use the Lewis structure to determine the geometry of the molecule Electron arrangement establishes the bond angles Molecule takes the shape of that portion of the electron arrangement Charts look at the CENTRAL atom for all data! Think REGIONS OF ELECTRON DENSITY rather than bonds (for instance, a double bond would only be 1 region)
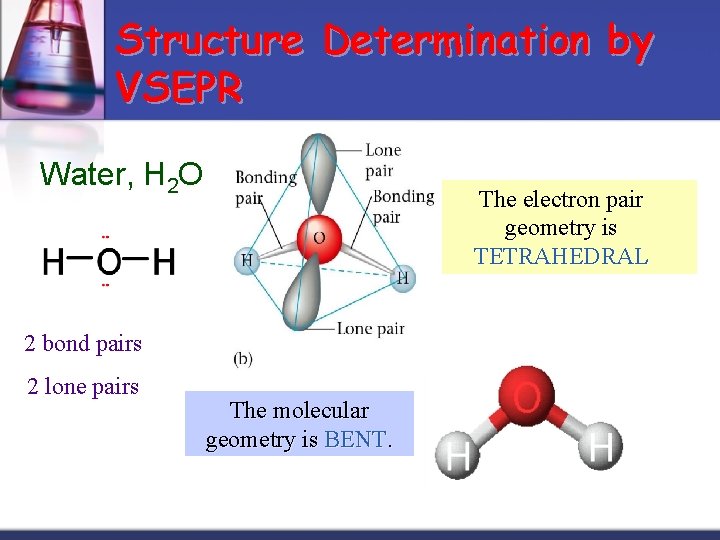
Structure Determination by VSEPR Water, H 2 O The electron pair geometry is TETRAHEDRAL 2 bond pairs 2 lone pairs The molecular geometry is BENT.
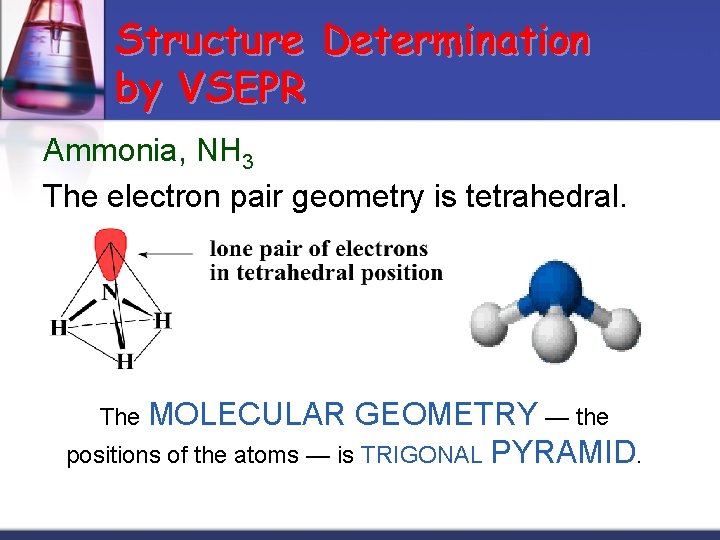
Structure Determination by VSEPR Ammonia, NH 3 The electron pair geometry is tetrahedral. The MOLECULAR GEOMETRY — the positions of the atoms — is TRIGONAL PYRAMID.
- Slides: 32