Chemical Bonding Review valence electrons Valence electrons are

Chemical Bonding


Review: valence electrons • Valence electrons are the electrons found in the outermost energy level of the atom • Valence electrons are responsible forming chemical bonds • TO THE RIGHT: Atoms of nitrogen have 5 valence electrons

Review - Octets • When an atom has eight valence electrons it has an octet • Octets are chemically stable • Atoms want to be stable, so they will gain, lose or share electrons to have an octet ( BONDING)

Why do atoms bond? • We learned that octets (a full valence electron shell) are very stable. Atoms desire stability, so they will combine with other elements in order to achieve a full octet of valence electrons. • To become more stable by filling their outer energy level with electrons (8)

Valence Electrons and the Periodic Table • The periodic table is divided into groups which are columns of elements with similar properties • Elements in the same group will have the same number of valence electrons (exception: transition metals)

Three Main Types of Bonding • Ionic bonding –a metal and a nonmetal • Covalent bonding – a nonmetal and a nonmetal • Metallic bonding –a metal and a metal

What type of bond is it? Find the following elements on the periodic table and determine their bond type: a) Ca & S b) Li & Pt c) C & Cl

Ionic Bonding • Metal and non-metal (one is way more electronegative than the other) • Metal loses e- and becomes positive (“cation”) • Nonmetal gains e- and becomes negative (“anion”) • Opposites attract: the ions bond via opposite charges
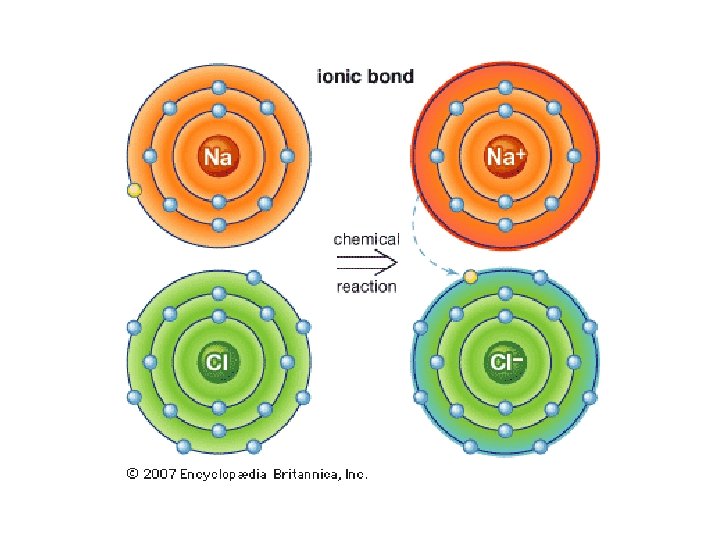

PROPERTIES OF IONIC COMPOUNDS 1. Crystalline solids – usually hard crystals 2. High melting and boiling points – because of strong forces in the bond 3. Soluble in water – water molecules attract the ions 4. Conduct electricity (electrolyte)


Covalent Bonding • Two or more nonmetals (not a large electronegativity difference) • SHARE electrons (the outer energy levels overlap) • 1, 2, or 3 electrons shared (single, double, or triple bond)
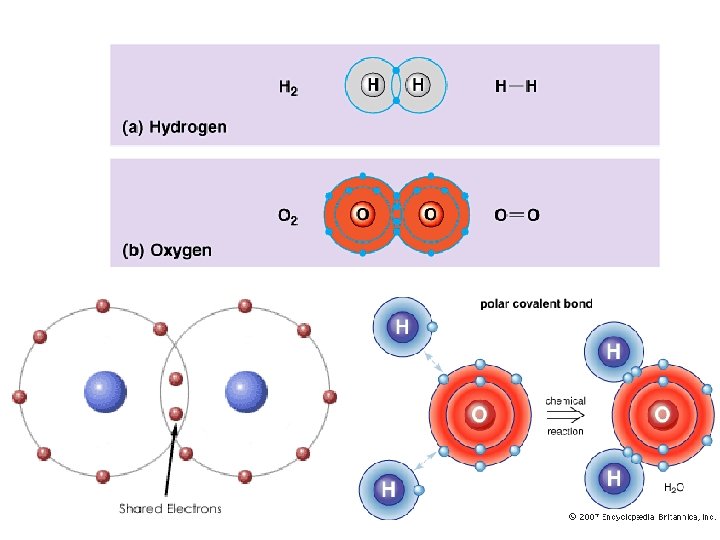

COVALENT COMPOUNDS: 1. Soft and brittle 2. Low melting and boiling points – because covalent bonds are weak 3. Insoluble in water – except if dipole moment 4. Do NOT conduct electricity non-electrolyte

Metallic Bonding • Metal and a metal • Metals are held together in rigid shapes • The nuclei are attracted to delocalized electrons from other atoms

Review Questions: 1) Why do atoms bond? 2) Which type of bond shares electrons? 3) What is a cation? Does the metal or the nonmetal become the cation? 4) What is a triple bond? 5) What types of compounds are soft? Do they conduct electricity? 6) What is an electrolyte?
- Slides: 17