Chemical Bonding Review Valence electrons Electrons in the

Chemical Bonding Review

Valence electrons • Electrons in the highest occupied energy level of an atom or ion • These electrons determine the properties of the atom • How do we know how many valence electrons an atom or ion has? • ELECTRON CONFIGURATION
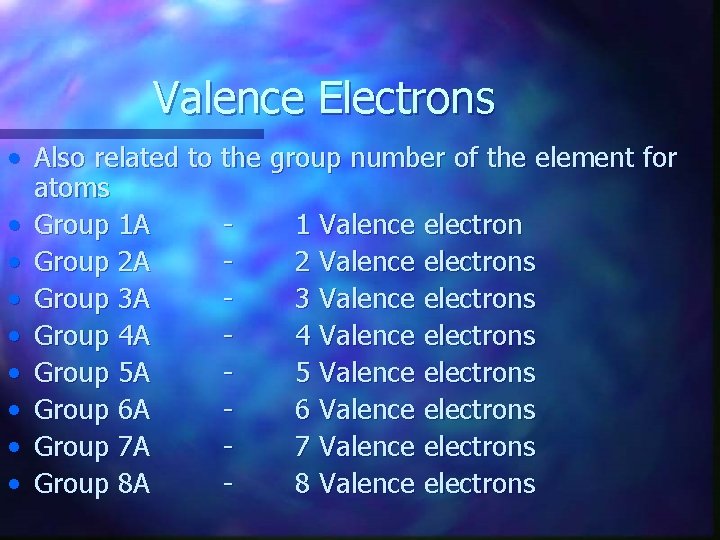
Valence Electrons • Also related to the group number of the element for atoms • Group 1 A 1 Valence electron • Group 2 A 2 Valence electrons • Group 3 A 3 Valence electrons • Group 4 A 4 Valence electrons • Group 5 A 5 Valence electrons • Group 6 A 6 Valence electrons • Group 7 A 7 Valence electrons • Group 8 A 8 Valence electrons

Can we draw those? • Yes! • • • Let’s draw the valence electrons for Mg (I will do the first one) K N O F

Lewis Structures • Valence electrons are usually the only ones used in chemical bonding • Therefore, we can represent atoms and ions using ELECTRON DOT or LEWIS NOTATION

OK, so what? • The octet rule helps us decide if an atom will gain or lose an electron, or tell us how many bonding electrons it can share in a chemical bond

The Octet Rule • Which atoms have the most stable or preferred electron configurations? • NOBLE GASES • Apart from He, how may valence electrons do each of the noble gases have? • 8 • What is the electron configuration of these valence electrons? • ns 2 np 6

The Octet Rule • So, all other atoms want to have this stable configuration with 8 valence electrons • This is the OCTET RULE • OCTET = EIGHT

Cations • Sodium – Na – What is the electron configuration? • What type of ion will it form and how?

Anions • What is the electron configuration of fluorine? • What type of ion will it form and how?

Types of Bonds • • • Ionic Bonds Electrostatic Interactions Net charge Transfer • Covalent Bonds • Sharing of one of more electron

Ionic Bonds • A large difference in electro-negativities will lead to an IONIC BOND between two atoms
![Formation of an Ionic Bond Na ( [Ne] 3 s 1 ) + Cl Formation of an Ionic Bond Na ( [Ne] 3 s 1 ) + Cl](http://slidetodoc.com/presentation_image_h2/1b2afd2e6c796819907e425f1dc1803a/image-13.jpg)
Formation of an Ionic Bond Na ( [Ne] 3 s 1 ) + Cl ( [Ne] 3 s 2 3 p 5 ) Na+ ([Ne]) + Cl- ([Ne] 3 s 2 3 p 6) • Note that each ion formed has a noble gas configuration of electrons -- Na + has the Ne configuration of 10 electrons and Cl- has the Ar configuration of 18 electrons – Na+/Ne and Cl-/Ar are said to be ISOELECTRONIC • Such electron arrangements are very stable, and accounts for the change in electron structure that Na and Cl atoms undergo.

Covalent Bonds A small difference in electro-negativities will lead to a covalent bond between two atoms.

CH 4 Electronegativity of C = 2. 55 electronegativity of H = 2. 20 DEN = 2. 55 - 2. 20 = 0. 35
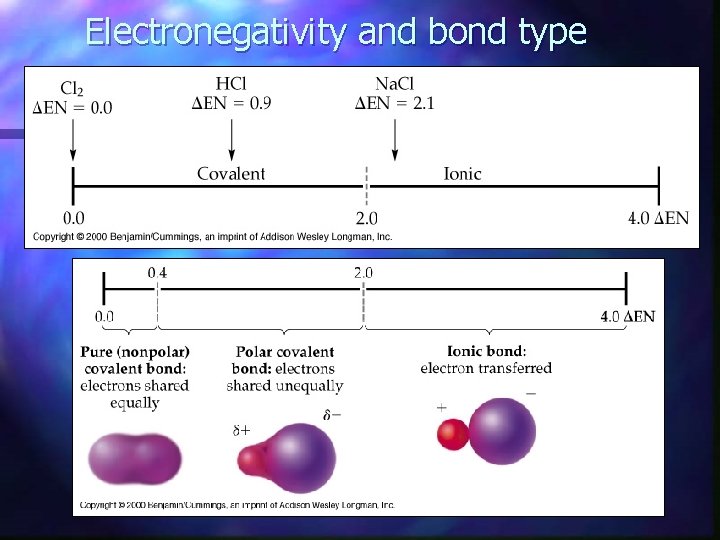
Electronegativity and bond type

Properties of Ionic Compounds • • • High Melting points Soluble in polar solvents (e. g. water) Insoluble on non-polar solvents (e. g. hexane) Molten ionic compounds conduct electricity Aqueous solutions conduct electricity Formed between elements of quite different electro negativities (metals and non-metals)

Properties of Covalent Compounds • • Usually low melting point Insoluble in polar solvents Soluble in non-polar solvents Liquid and molten compounds do not conduct electricity • Aqueous solutions are poor conductors • Often formed between elements of similar electro negativities

Properties of Metals & Alloys • To be done on the board
- Slides: 19