Chemical Bonding Polarity of Compounds Bond Polarity Nonpolar

Chemical Bonding Polarity of Compounds
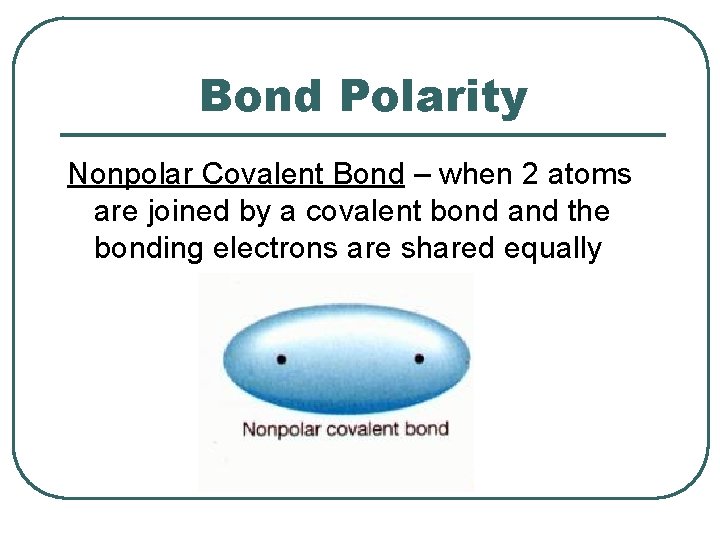
Bond Polarity Nonpolar Covalent Bond – when 2 atoms are joined by a covalent bond and the bonding electrons are shared equally
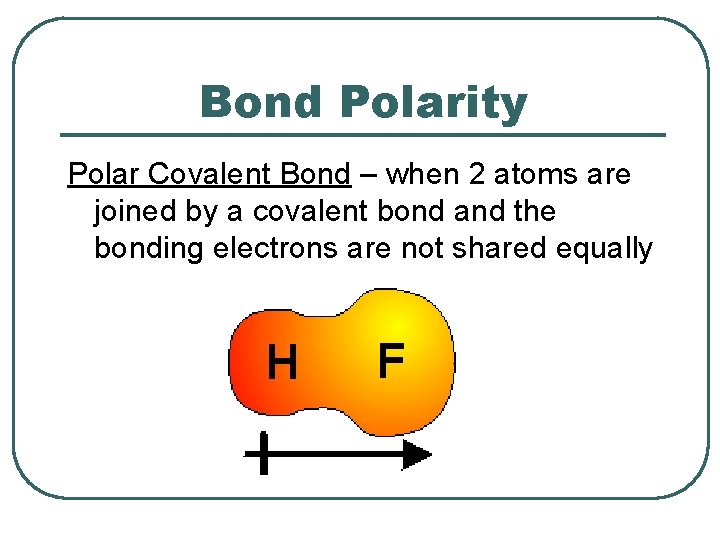
Bond Polarity Polar Covalent Bond – when 2 atoms are joined by a covalent bond and the bonding electrons are not shared equally
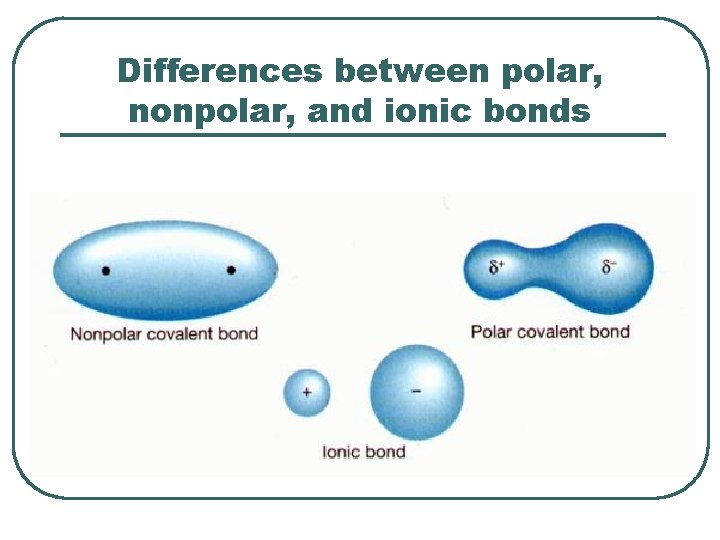
Differences between polar, nonpolar, and ionic bonds

How do you determine if a bond is polar, nonpolar, or ionic? Subtract the electronegativities of the bonding atoms
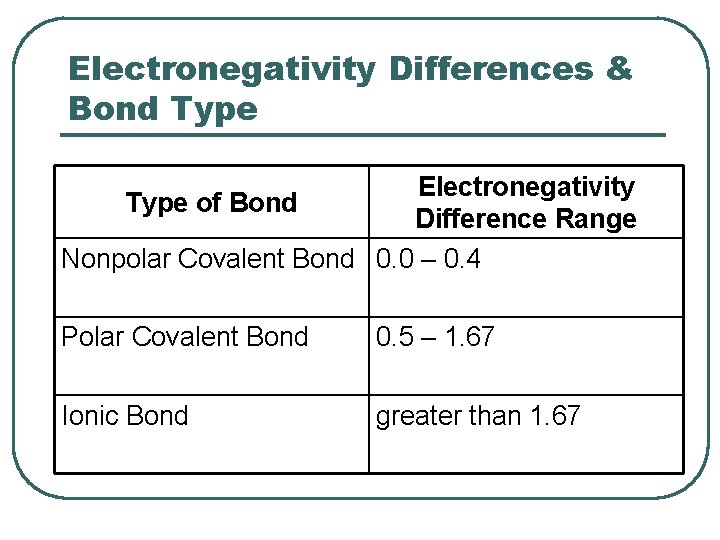
Electronegativity Differences & Bond Type Electronegativity Type of Bond Difference Range Nonpolar Covalent Bond 0. 0 – 0. 4 Polar Covalent Bond 0. 5 – 1. 67 Ionic Bond greater than 1. 67
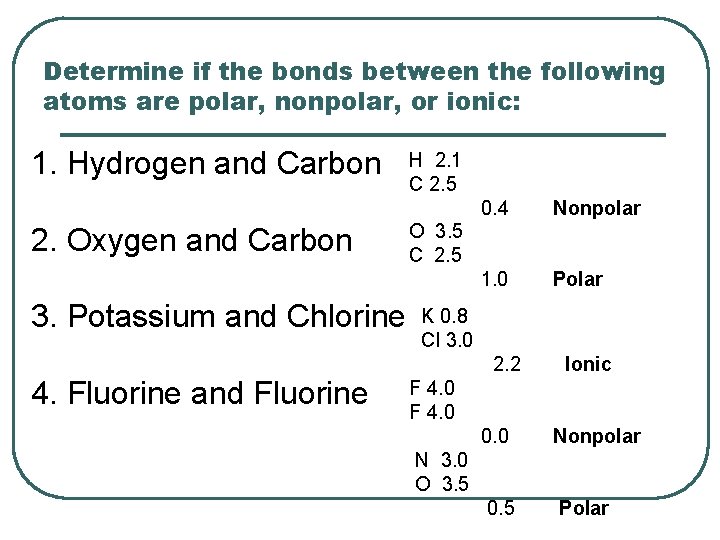
Determine if the bonds between the following atoms are polar, nonpolar, or ionic: 1. Hydrogen and Carbon 2. Oxygen and Carbon 3. Potassium and Chlorine H 2. 1 C 2. 5 0. 4 Nonpolar 1. 0 Polar O 3. 5 C 2. 5 K 0. 8 Cl 3. 0 2. 2 4. Fluorine and Fluorine Ionic F 4. 0 0. 0 Nonpolar 0. 5 Polar N 3. 0 O 3. 5
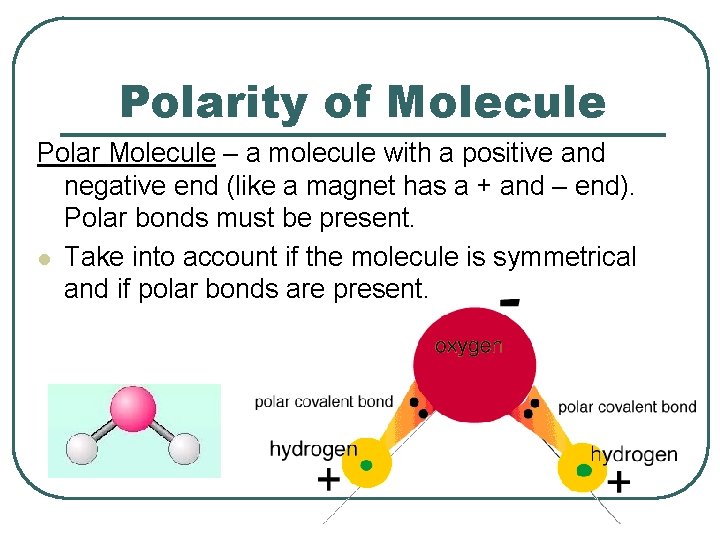
Polarity of Molecule Polar Molecule – a molecule with a positive and negative end (like a magnet has a + and – end). Polar bonds must be present. l Take into account if the molecule is symmetrical and if polar bonds are present.
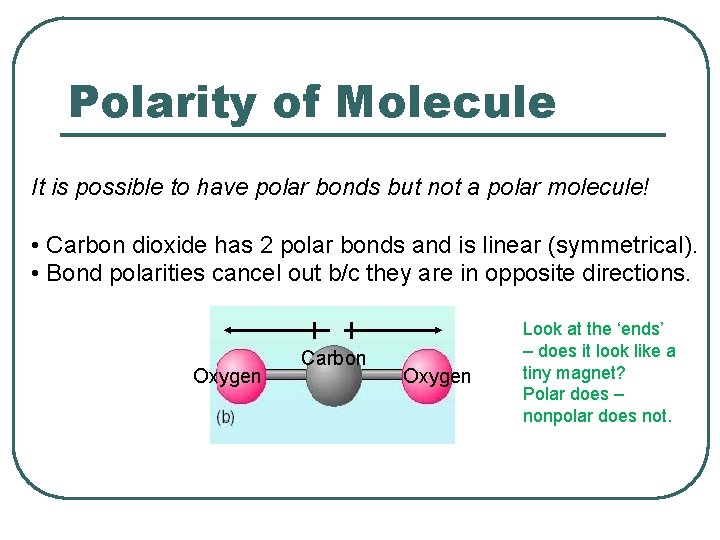
Polarity of Molecule It is possible to have polar bonds but not a polar molecule! • Carbon dioxide has 2 polar bonds and is linear (symmetrical). • Bond polarities cancel out b/c they are in opposite directions. Oxygen Carbon Oxygen Look at the ‘ends’ – does it look like a tiny magnet? Polar does – nonpolar does not.
- Slides: 9