Chemical Bonding Notes Valence electrons are used in
Chemical Bonding Notes

Valence electrons are used in bonding. • Stable elements want to achieve 8 electrons similar to the noble gases • A metal wants to achieve the configuration for the noble gas before. • A nonmetal wants to achieve the configuration for the noble gas after.

Bond- force that holds groups of two or more atoms together and makes them function as a unit Bond energy- energy required to break the bond (tells the bond strength) Ionic bonding- between ionic compounds which contain a metal and a nonmetal • Atoms that lose electrons relatively easily react with an atom that has a high affinity for electrons • Transfer of electrons Covalent bonding- between two nonmetals • Electrons are shared by nuclei Polar Covalent bonding- unequal sharing of electrons • positive end attracted to the negative end • (delta) indicates partial charge

• Electronegativity-(p. 402) relative ability of an atom in a molecule to attract shared electrons to itself • The higher the atom’s electronegativity value, the closer the shared electrons tend to be to that atom when it forms a covalent bond Increases – across a period Decreases- down a group Electronegativity difference Bond type Zero (0 - 0. 4) Covalent Intermediate (0. 4 – 1. 4) Polar covalent Large (>1. 4) Ionic

Bond Type Ex. Determine if each bond is covalent, polar covalent or ionic. H-H, O-H, Cl-H, S-H, F-H H-H = 2. 1 - 2. 1 = 0 covalent O-H = 3. 5 - 2. 1 = 1. 4 polar covalent Cl-H = 3. 0 - 2. 1 =. 9 polar covalent S-H = 2. 5 - 2. 1 =. 4 covalent F-H = 4. 0 - 2. 1 = 1. 9 ionic

Molecule (molecular compound) – term used to describe covalent compounds, made from two nonmetals Naming Molecular Compounds • Prefixes: (MEMORIZE) Mono-1 tetra-4 hepta-7 di-2 penta-5 octa-8 tri-3 hexa-6 non-9 deca-10 • prefixes are used with both the first named and second named element. Exception: mono- is not used on the first word • second word ends in –ide • If a two syllable prefix ends in a vowel, the vowel is dropped before the prefix is attached to a word beginning with a vowel monooxide Writing Molecular Formulas Translate prefixes

Molecule Names & Formulas Examples: N 2 O = Dinitrogen monoxide dihydrogen monoxide = H 2 O Si 8 O 5 = Octasilicon pentoxide tetrasulfur hexachloride = S 4 Cl 6 NH 3 = Nitrogen trihydride carbon monoxide = CO P 3 I 10 = Triphosphorus deciodide carbon dioxide = CO 2

Lewis Structure- representation of a molecule Shows how the valence electrons are arranged among the atoms in the molecule. For an element: s Oxygen 1 s 22 p 4 pz X px py • • • • O • • For a compound: Li + Cl For a molecule: • • F F • • [Li]+1 + [ Cl ]-1

Duet rule- He & He only need 2 electrons for a full, stable valence shell Octet rule- eight electrons needed for a full, stable valence shell Bonding pair- electrons shared with other atom Line (─) = 2 shared electrons Lone pair - not involved in bonding dots ( • • ) = 2 unshared electrons
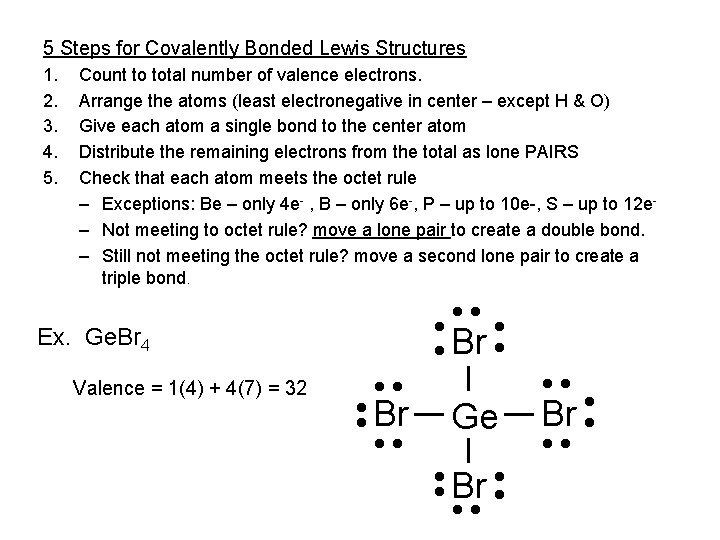
5 Steps for Covalently Bonded Lewis Structures 1. 2. 3. 4. 5. Count to total number of valence electrons. Arrange the atoms (least electronegative in center – except H & O) Give each atom a single bond to the center atom Distribute the remaining electrons from the total as lone PAIRS Check that each atom meets the octet rule – Exceptions: Be – only 4 e- , B – only 6 e-, P – up to 10 e-, S – up to 12 e– Not meeting to octet rule? move a lone pair to create a double bond. – Still not meeting the octet rule? move a second lone pair to create a triple bond. Ex. Ge. Br 4 Valence = 1(4) + 4(7) = 32 • • Br • • • • Br • • Ge • • Br • • • Br • • •
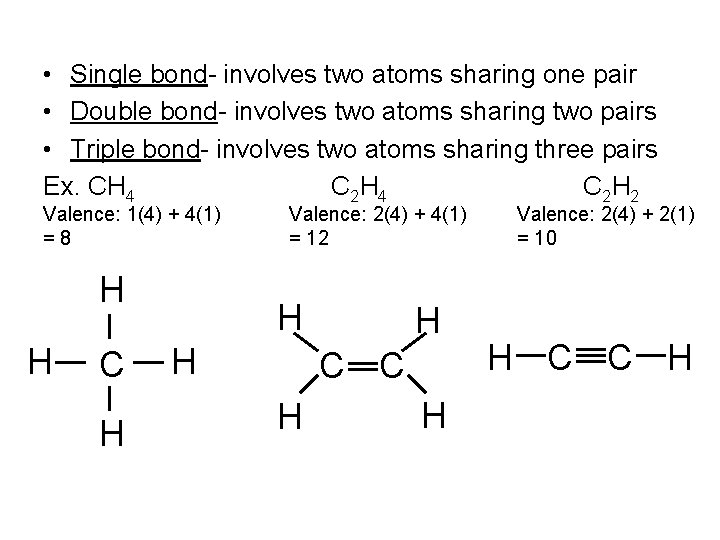
• Single bond- involves two atoms sharing one pair • Double bond- involves two atoms sharing two pairs • Triple bond- involves two atoms sharing three pairs Ex. CH 4 C 2 H 2 Valence: 1(4) + 4(1) =8 H H C H Valence: 2(4) + 4(1) = 12 H H H C C H H Valence: 2(4) + 2(1) = 10 H C C H
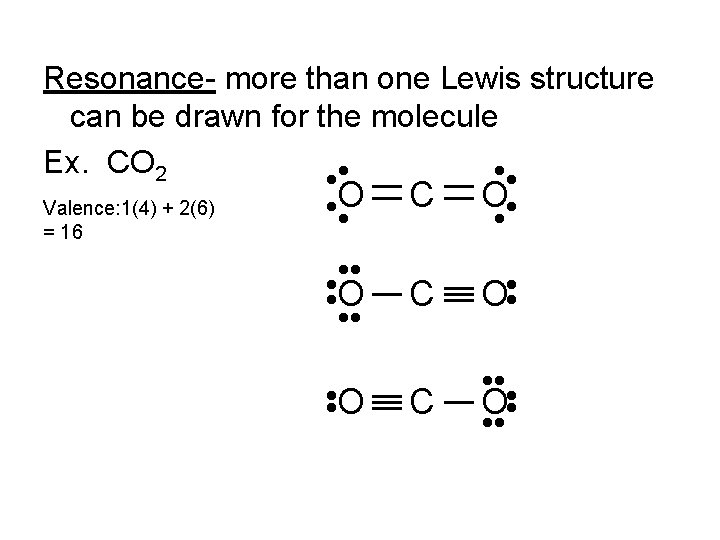
Resonance- more than one Lewis structure can be drawn for the molecule Ex. CO 2 • • Valence: 1(4) + 2(6) = 16 • O • • O • • O C O • • • C O • • C • • • O • • •
- Slides: 12