Chemical Bonding Lewis Structures Forming Chemical Bonds According

Chemical Bonding Lewis Structures

Forming Chemical Bonds • According to the Lewis model – an atom may lose or gain enough electrons to acquire a filled valence shell and become an ion. An ionic bond is the result of the force of attraction between a cation and an anion. – an atom may share electrons with one or more other atoms to acquire a filled valence shell. A covalent bond is the result of the force of attraction between two atoms that share one or more pairs of electrons. Material from karentimberlake. com and H. Stephen Stoker

Ionic Compounds Vs Covalent (Molecules) Compounds

• Ionic Compounds – Made up of a metal and a non-metal – Have a difference in electronegativity of more than 1. 7 (Figure 20 pg. 161) – Formed from ions – Electrons are transferred

• Covalent Molecules – Made up of two non-metals – Have a difference in electronegativity of less than 1. 7 (Figure 20 pg. 161) – Ions are not formed – Electrons are shared

Bond Polarity: Nonpolar covalent bond l Electrons are shared between atoms with the same electronegativity values. l Difference = 0 l Examples: N 2 Br 2

Bond Polarity: Polar covalent bond l Electrons are shared between different nonmetal atoms Examples: O-Cl O-S N-Cl
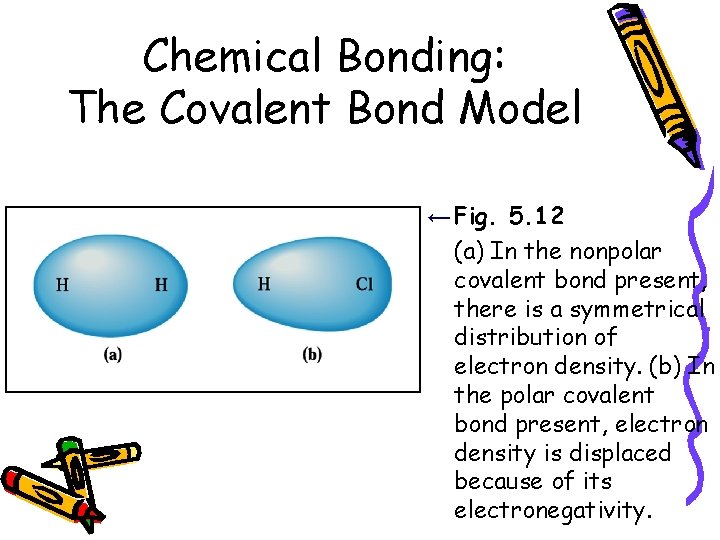
Chemical Bonding: The Covalent Bond Model ← Fig. 5. 12 (a) In the nonpolar covalent bond present, there is a symmetrical distribution of electron density. (b) In the polar covalent bond present, electron density is displaced because of its electronegativity.

Valence electrons for Elements Recall that the valence electrons for the elements can be determined based on the elements position on the periodic table. Lewis Dot Symbol 9 Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures 1) Find your element on the periodic table. 2) Determine the number of valence electrons. 3) This is how many electrons you will draw. 10 Lewis Structure Tutorial 10. 7. 00 6: 16 PM
Lewis Structures • Find out which group (column) your element is in. • This will tell you the number of valence electrons your element has. • You will only draw the valence electrons. www. chem 4 kids 11 Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures C 12 1) Write the element symbol. 2) Carbon is in the 4 th group, so it has 4 valence electrons. 3) Starting at the right, draw 4 electrons, or dots, counter-clockwise around the element symbol. Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures C 13 1) Check your work. 2) Using your periodic table, check that Carbon is in the 4 th group. 3) You should have 4 total electrons, or dots, drawn in for Carbon. Lewis Structure Tutorial 10. 7. 00 6: 16 PM
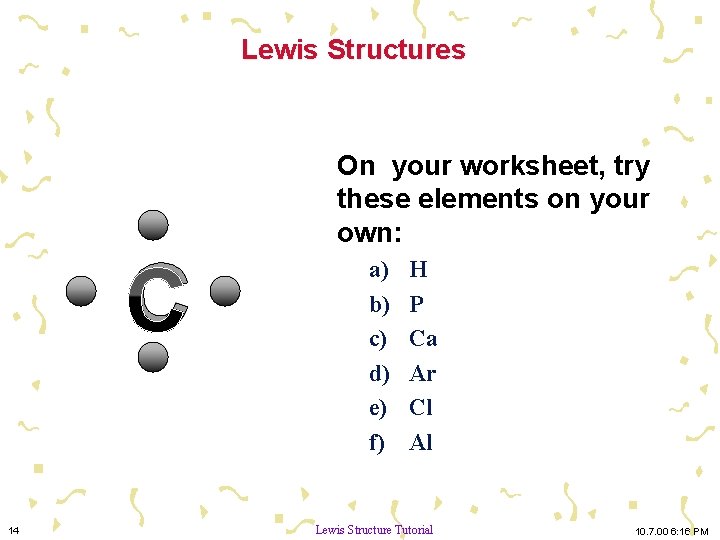
Lewis Structures C 14 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures H 15 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM
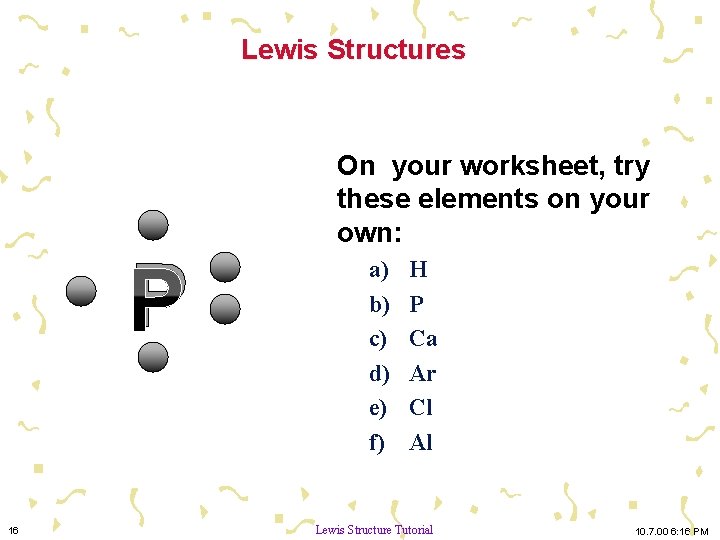
Lewis Structures P 16 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures Ca 17 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM
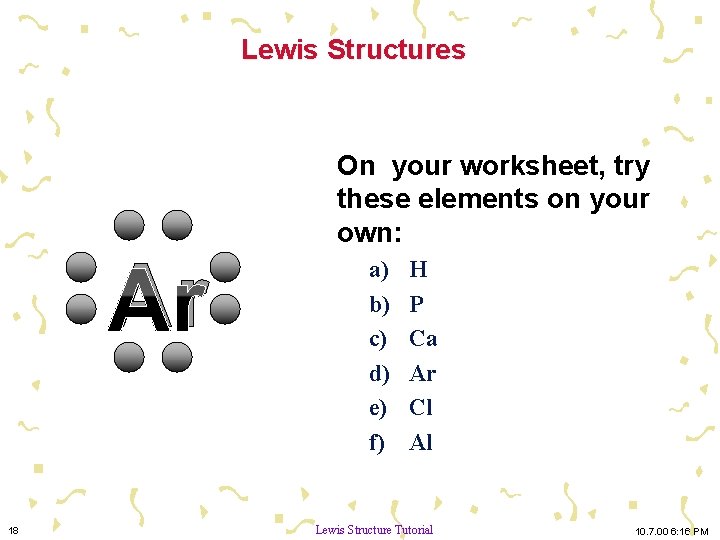
Lewis Structures Ar 18 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures Cl 19 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structures Al 20 On your worksheet, try these elements on your own: a) b) c) d) e) f) H P Ca Ar Cl Al Lewis Structure Tutorial 10. 7. 00 6: 16 PM
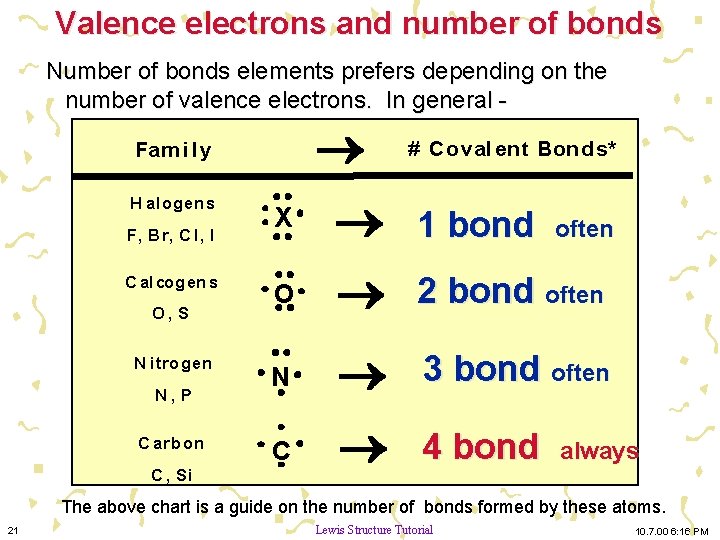
Valence electrons and number of bonds Number of bonds elements prefers depending on the number of valence electrons. In general - Fam i l y H alogen s F, Br, C l, I C alcogen s O, S N itrogen N, P C arb on C , Si # C oval ent Bonds* 1 bond O N 3 bond often C 4 bond X often 2 bond often always The above chart is a guide on the number of bonds formed by these atoms. 21 Lewis Structure Tutorial 10. 7. 00 6: 16 PM

Lewis Structure, Octet Rule Guidelines When compounds are formed they tend to follow the Octet Rule: Atoms will share electrons (e-) until it is surrounded by eight valence electrons. Rules of the (VSEPR) gamei) The Octet Rule works mostly for second period elements. Many exceptions especially with 3 rd period elements (d-orbitals) ii) H prefers 2 e- (electron deficient) iii) : C: . . N: . . : O: . . . : F: . 4 unpaired 3 unpaired 2 unpaired 1 unpaired up = unpaired e 4 bonds 3 bonds 2 bonds 1 bond O=C=O N N O=O F-F iv) H & F are terminal in the structural formula (Never central) 22 Lewis Structure Tutorial 10. 7. 00 6: 16 PM
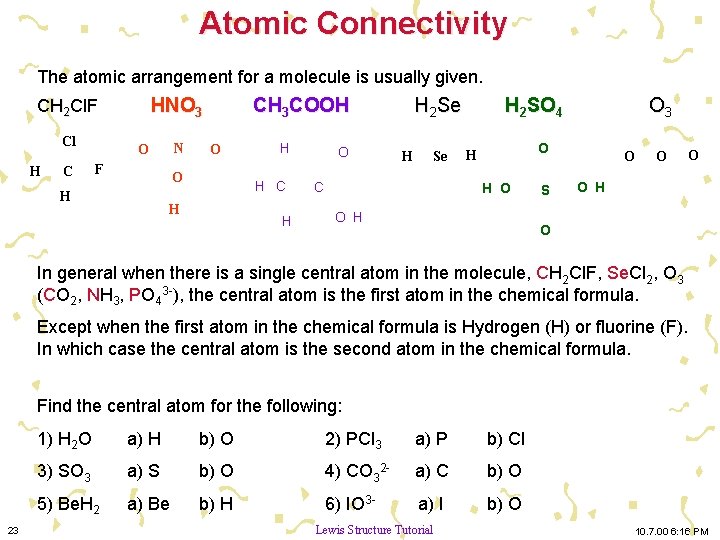
Atomic Connectivity The atomic arrangement for a molecule is usually given. HNO 3 CH 2 Cl. F Cl H C N O F CH 3 COOH O O H H H C H H O H 2 Se H Se C H 2 SO 4 O 3 O H H O O H S O O H O In general when there is a single central atom in the molecule, CH 2 Cl. F, Se. Cl 2, O 3 (CO 2, NH 3, PO 43 -), the central atom is the first atom in the chemical formula. Except when the first atom in the chemical formula is Hydrogen (H) or fluorine (F). In which case the central atom is the second atom in the chemical formula. Find the central atom for the following: 23 1) H 2 O a) H b) O 2) PCl 3 a) P b) Cl 3) SO 3 a) S b) O 4) CO 32 - a) C b) O 5) Be. H 2 a) Be b) H 6) IO 3 - a) I b) O Lewis Structure Tutorial 10. 7. 00 6: 16 PM
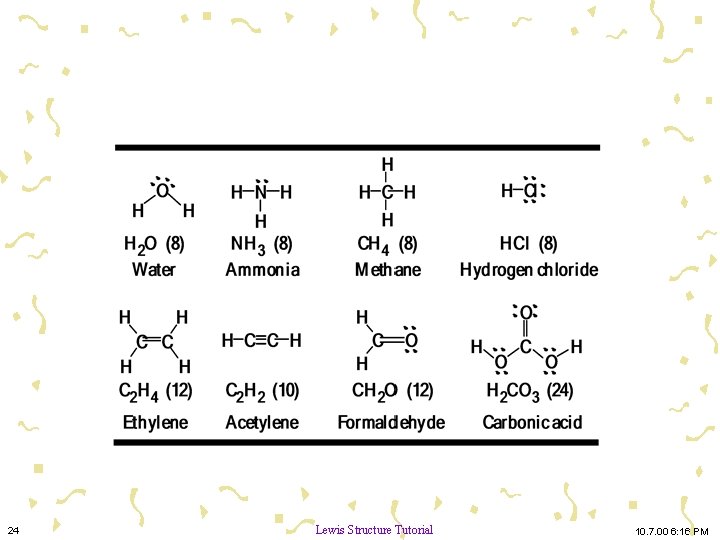
Lewis Structures 24 Lewis Structure Tutorial 10. 7. 00 6: 16 PM
- Slides: 24