Chemical Bonding Ionic Bonding and Ionic Compounds Most

Chemical Bonding Ionic Bonding and Ionic Compounds

� Most of the rocks and minerals that make up the Earth’s crust consist of positive and negative ions held together by ionic bonding.

� Ionic compounds are made up of positive and negative ions that are combined so that the numbers of positive and negative charges are equal

� Most ionic compounds exist as crystalline solids � A crystal is a 3 -D network of positive and negative ions mutually attracted to one another

�A formula unit show the simplest ratio of positive and negative ions in a crystal ◦ Na. Cl ◦ Mg. Br 2 � For ionic compounds, the subscripts are reduced to the smallest whole-number ratio. ◦ This is different from molecular compounds which show the number of atoms in one molecule which is NOT always the smallest whole-number ratio.
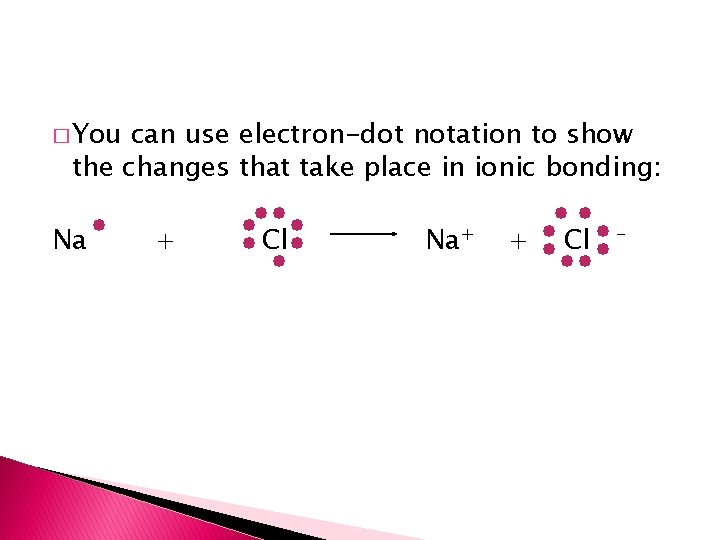
� You can use electron-dot notation to show the changes that take place in ionic bonding: Na + Cl Na+ + Cl -

� Remember that nature favors arrangements where potential energy is minimized � In ionic crystals, potential energy is minimized when the cations and anions combine in an orderly arrangement called a crystal lattice � The smallest part of an ionic crystal that shows the arrangement of the ions is the unit cell.

� Attractive forces � Repulsive forces ◦ Between oppositely charged ions ◦ Between nuclei and electrons of adjacent ions ◦ Between like-charged ions ◦ Between nuclei of adjacent ions ◦ Between electrons of adjacent ions
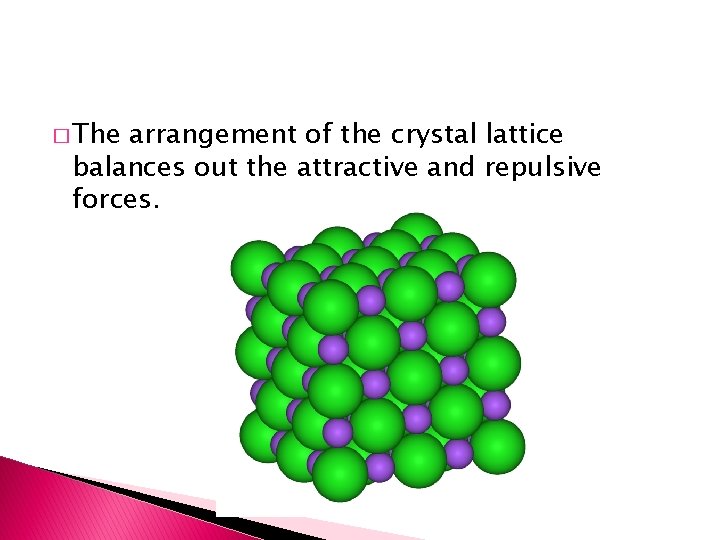
� The arrangement of the crystal lattice balances out the attractive and repulsive forces.

� The 3 -D arrangement of ions depends on the sizes and charges of the ions and the number of ions of different charges. � In Na. Cl, each sodium ion is surrounded by 6 chloride ions and each chloride ion is surrounded by 6 sodium ions. � In Ca. F 2, each calcium ion is surrounded by 8 fluoride ions, and each fluoride ion is surrounded by 4 calcium ions

� Lattice energy is the amount of energy released when one mole of an ionic crystalline compound is formed from gaseous ions. � Lattice energies are negative because energy is released as the crystal forms

� The greater the amount of energy released when the crystal forms, the stronger the ionic bonds are. � The stronger the bonds are, the higher the melting point and boiling point are.

Comparison between Ionic and Molecular Compounds � In ionic compounds, the ionic bonds are very strong attractions between the ions. � In molecular compounds, the covalent bonds within a molecule are also quite strong ◦ However, the bonds between adjacent molecules are much weaker

� Melting point, boiling point, and hardness of a compound depend on how strongly the units are attracted to each other ◦ Ions are very attracted to each other in ionic compounds so their melting point and boiling point are high and they are very hard. ◦ The bonds between adjacent molecules are not as strong, so molecular compounds have lower melting and boiling points. Many molecular compounds are gases at room temperature.

� Ionic compounds may be hard, but they are brittle. � In an ionic crystal, if one layer of the crystal shifts, there is a large buildup of repulsive forces, causing the crystal to break

� In the solid state, ionic crystals are not electrical conductors. ◦ This is because the ions cannot move – they are held in fixed positions � In the molten state, ionic compounds are electrical conductors because the ions can move freely

� Many ionic compounds can dissolve in water � When they dissolve, the ions separate and become surrounded by water molecules � Because the ions are free to move around the solution, the solutions are electrical conductors

Polyatomic Ions � Certain atoms bond covalently with each other to form a group of atoms that has both ionic and molecular characteristics � A polyatomic ion is a group of covalently bound atoms with a charge ◦ The charge results from an excess or shortage of electrons

� Lewis structures of polyatomic ions are drawn in brackets with the charge of the ion shown outside the brackets on the upper right.


� CH 3 COO-

- Slides: 22