Chemical Bonding Introduction Introduction Introduction The Law of
Chemical Bonding

Introduction
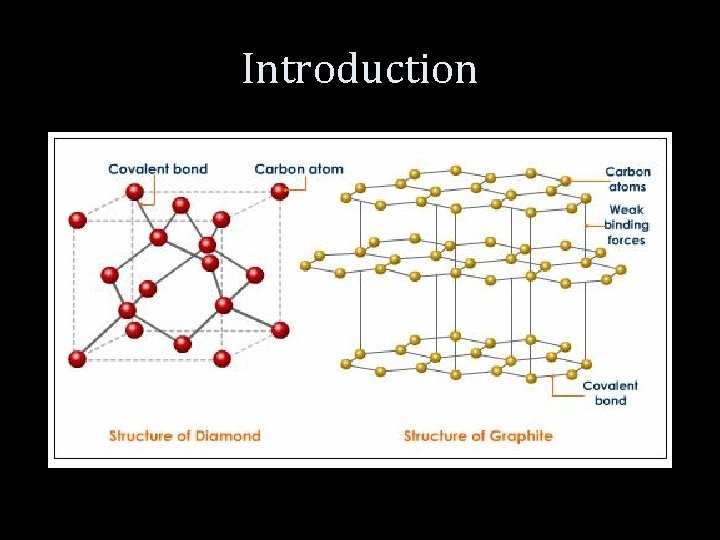
Introduction

Introduction • The Law of Definite Proportions (Joseph Louis Proust, 1799) and Dalton’s development of atomic theory (1803) lead to the recognition that atoms of an element had a characteristic combining ability with other atoms, which came to be called valence. • Existence of atoms suggested that compounds were composed of collections of atoms bound together by chemical bonds.

• Atoms combine to form molecules. • The combining power of atoms to form molecules is called valency. • All atoms having unstable or incomplete outer shell have a tendency to gain or lose electrons tendency of atoms to complete and hence stabilize their outermost orbit of electrons which is mainly responsible for chemical combination between the atoms.

• According to ‘electronic theory of valency’, a chemical bond is formed as a result of electronic interactions. • However, a molecule is formed only when electrons of the constituent atoms interact in such a way that the potential energy is lowered.

• A bond as a force that holds groups of two or more atoms together and makes them function as a unit. • A chemical bond as an effect that causes the energy of two atoms close together to be markedly lower (by about 100 k. J per mole or more) than when they are far apart. • The forces that hold bonded atoms together are basically just the same kinds electrostatic attractions that bind the electrons of an atom to its positivelycharged nucleus.

Lewis electron – dot formulas • Lewis originated the idea of the electron pair bond. • In 1902, Lewis developed the concept of valence electrons and realized that all elements known to form simple ions by losing or gaining whatever number of electrons is needed to leave eight in the valence shell of each. • In 1916, Lewis published shared electron-pair theory.

• Lewis Symbol/Structure --- representing atom singly or in combination • Only the valence electrons are shown.

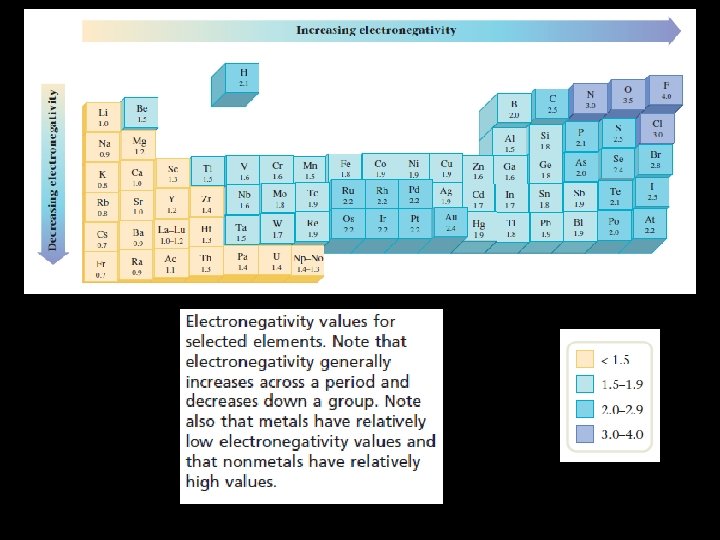
• Atoms having a tendency to lose electrons are called electropositive whereas the atoms which gain electrons are called electronegative. • The number of electrons gained or lost by an atom in order to acquire an inert gas configuration gives numerical value of the electrovalency of the atom. • A Lewis dot symbol consists of the symbol of an element and one dot for each valence electron in an atom of the element.

Type of Bonds • Electrovalent or ionic bond Electropositive elements + Electronegative elements • Covalent bond Electronegative elements + Electronegative elements • Coordinate bond Electropositive elements + Electropositive elements
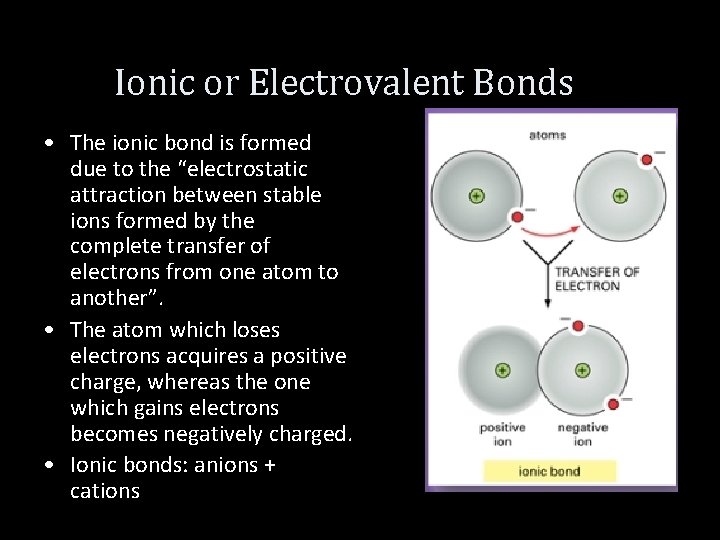
Ionic or Electrovalent Bonds • The ionic bond is formed due to the “electrostatic attraction between stable ions formed by the complete transfer of electrons from one atom to another”. • The atom which loses electrons acquires a positive charge, whereas the one which gains electrons becomes negatively charged. • Ionic bonds: anions + cations

Ionic Bonds • These charged atoms are called ions and held together by electrostatic attraction forces. • Such a mode of combination of atoms is called electrovalency and the bond formed between the atom is called electrovalent or ionic bond. • The compound thus formed is called an electrovalent or ionic compound.

Ionic Bonds • Ionic substances are formed when an atom that loses electrons relatively easily reacts with an atom that has a high affinity for electrons. • In other words, an ionic compound results when a metal reacts with a nonmetal.

Ionic Bonds Example 1: Na. CL • The electronic arrangement of Na and Cl atoms are: Na (11) (1 s 2, 2 p 6, 3 s 1) Cl (17) (1 s 2, 2 p 6, 3 s 2, 3 p 5) • The electron from the outermost orbit of Na is completely transferred to the outermost orbit of Cl atom. As a result of this transfer, both atoms acquire inert gas structure.
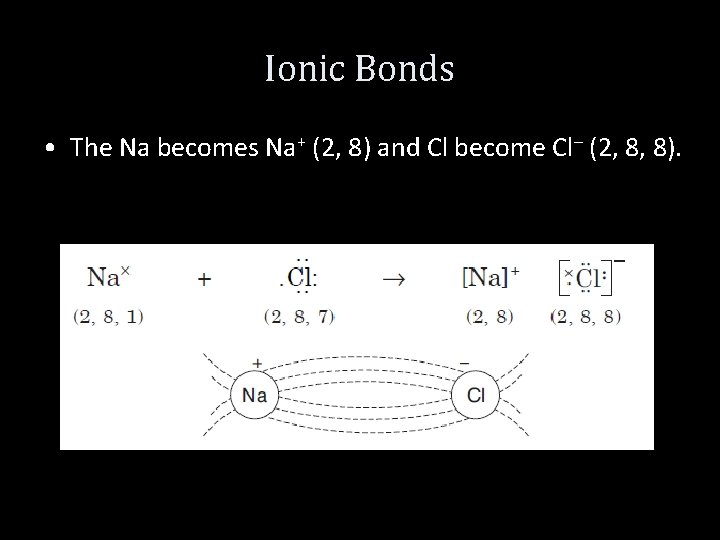
Ionic Bonds • The Na becomes Na+ (2, 8) and Cl become Cl– (2, 8, 8).
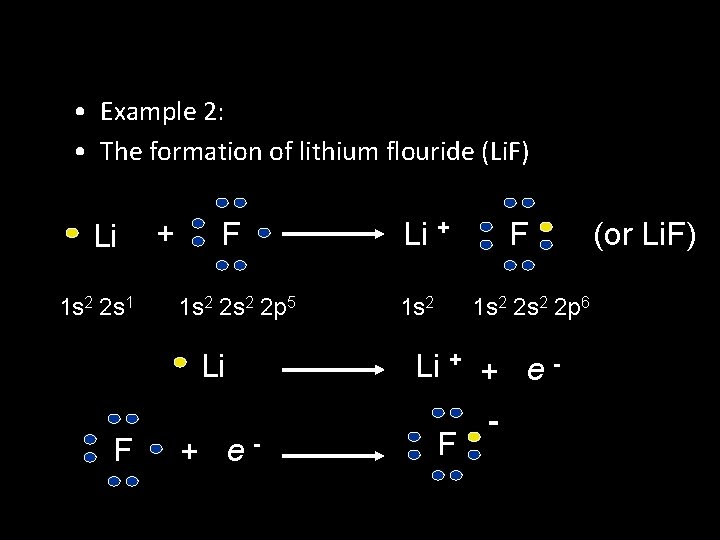
• Example 2: • The formation of lithium flouride (Li. F) Li 1 s 2 2 s 1 + F 1 s 2 2 p 5 Li F + e- Li + 1 s 2 F 1 s 2 2 p 6 Li + + e F - (or Li. F)

Ionic Bonds • Electrovalent compounds exhibit following properties: § Electrovalent compounds are generally hard solids. § They have high melting and boiling point. § Electrovalent compounds are generally sparingly soluble in organic solvents. § Electrovalent compounds in solid state are poor conductors of electricity. But when dissolved in solvents of relatively high dielectric constant, they exhibit a strong electrical conductivity. They also conduct electricity in the molten state.

Lattice energy of ionic compounds • The stability of solid ionic compounds depends on the interactions of all cations and anions. • A quantitative measure of the stability of any ionic solid is its lattice energy --- the energy required to completely separate one mole of a solid ionic compound into gaseous state.
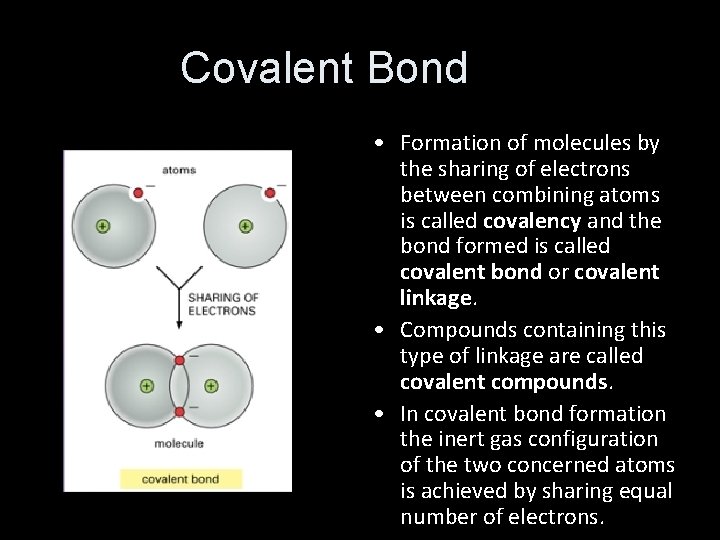
Covalent Bond • Formation of molecules by the sharing of electrons between combining atoms is called covalency and the bond formed is called covalent bond or covalent linkage. • Compounds containing this type of linkage are called covalent compounds. • In covalent bond formation the inert gas configuration of the two concerned atoms is achieved by sharing equal number of electrons.

Covalent Bond • The sharing of electrons to form chemical bond between two atoms is described by showing pairs of electrons between the bonded atoms. • If one pair of electrons is shared, the bond formed is called single bond, whereas sharing of two or three pairs of electrons leads to the formation of double or triple bonds respectively. • Due to sharing of electrons, both the bonding atoms acquire inert gas configuration.

Covalent Bond • Example 1: the formation of hydrogen molecule from 2 hydrogen atoms. • The covalent bond is represented by (–).
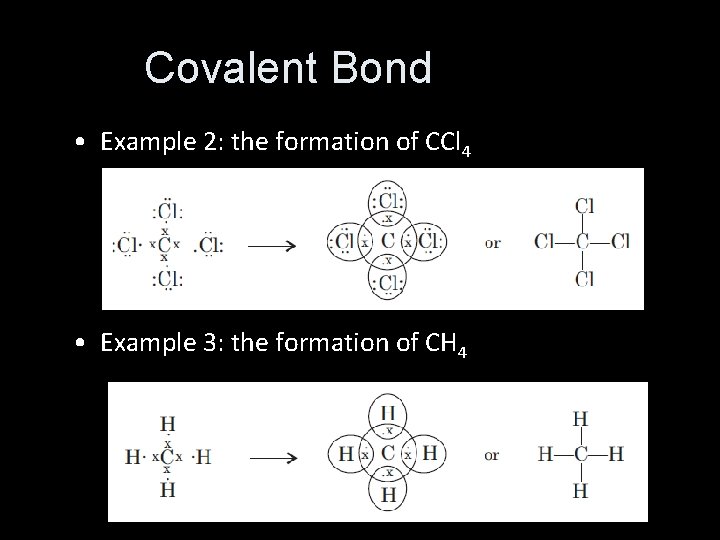
Covalent Bond • Example 2: the formation of CCl 4 • Example 3: the formation of CH 4
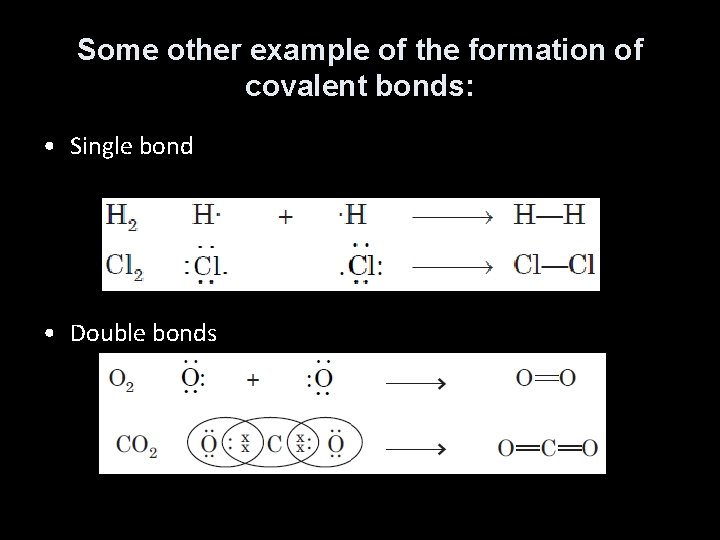
Some other example of the formation of covalent bonds: • Single bond • Double bonds
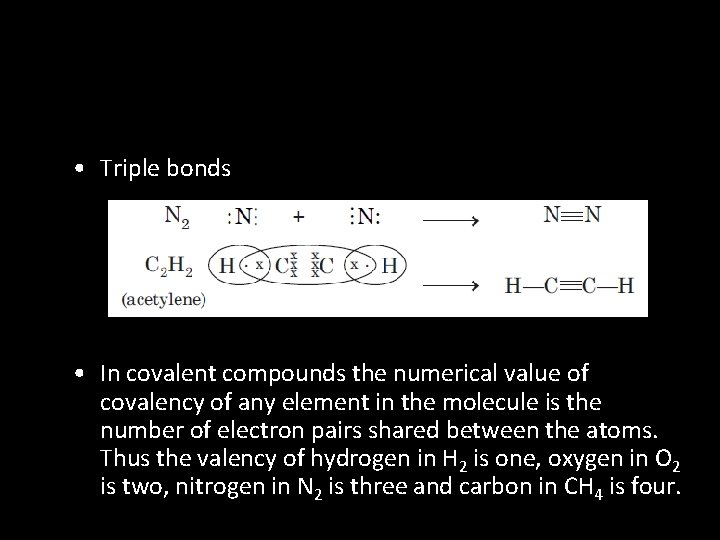
• Triple bonds • In covalent compounds the numerical value of covalency of any element in the molecule is the number of electron pairs shared between the atoms. Thus the valency of hydrogen in H 2 is one, oxygen in O 2 is two, nitrogen in N 2 is three and carbon in CH 4 is four.

• Two types of covalent bonds: § Polar covalent bonds When a bond is formed between unlike atoms, the bonding electrons will not be equally shared. Example: HCl The shared electrons will be shifted more towards the atom having higher electronegativity and this will result in the accumulation of a negative charge on it. The other atom will carry an equivalent positive charge.

• Example HCl: The chlorine atom acquires small amount of –ve charge because of its higher electronegativity and hydrogen has an equivalent +ve charge. The δ+ and δ – represents respectively the small +ve and –ve charge. Electronegativity: The ability of an atom to attract toward itself the electron in a chemical bond.

§ Non-polar covalent bonds. • When a bond is formed between atoms of the same element, the bonding electrons are equally shared on account of equal electronegativity of the atoms. • In case of such a bond, the centre of +ve charge coincides with the centre of –ve charge in the molecule. • For example, bonds involved in the formation of H 2, Cl 2, O 2, N 2 etc. are non-polar bonds.

• Covalent compounds possess following general characteristics: § Covalent compounds possess definite geometrical shapes. They exhibit isomerism because covalent bonds are rigid and possess directional characteristics. § Covalent compounds are mostly liquids and gases. The solid compounds are generally volatile. § They are highly soluble in organic solvents but slightly soluble in water. Some compounds like HCl and NH 3 readily dissolve in water because they react with water.

§ The melting and boiling points of covalent compounds are relatively low because the forces involved in covalent compounds are less strong than those involved in electrovalent or ionic compounds. § These compounds do not contain ions. Therefore, when dissolved, they do not conduct electricity. They even do not conduct electricity in the molten state.
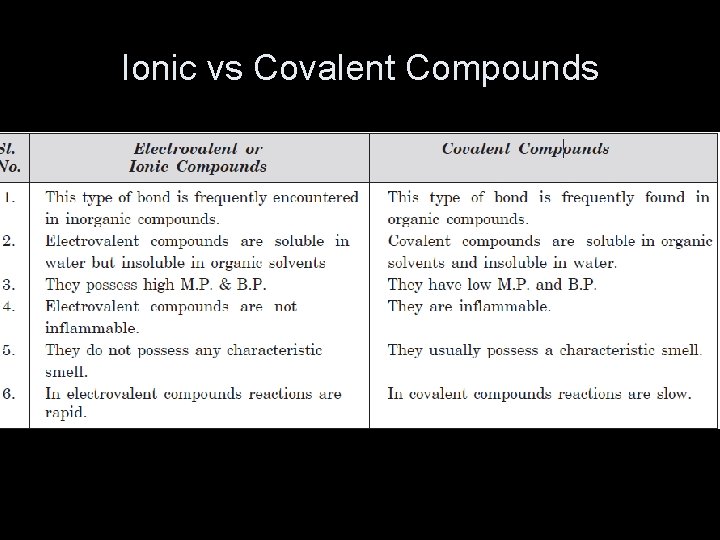
Ionic vs Covalent Compounds

Coordinate Bonds • When, an atom having a complete octet donates a pair of free valence electrons to another atom which is short of two electrons, the resulting bond is known as coordinate bond. Thus both the atoms acquire inert gas configurations. • The atom which donates a pair of electrons is called ‘donor’ and the other atom which accepts the electrons is called ‘acceptor’. The coordinate bond is similar to covalent bond except that both the shared electrons are donated by one atom.

• The formation of covalent and coordinate bond is illustrated below: • When one atom furnishes both electrons for the formation of a covalent bond as described above, the process is called ‘coordination’.

• Since one atom donates an electron pair and the other accepts, the molecule acquires polarity. These bonds, therefore, are also known as ‘semi polar bonds’ or ‘dative bond’. • Coordinate bond is represented by the symbol ( ). • Example:

• Following are the main characteristics of coordination compounds: § Coordinate compounds, like covalent compounds, exhibit space isomerism. This is due to directional characteristics possessed by coordinate linkage. § The B. P. and M. P. of these compounds have intermediate value between electrovalent and covalent compounds. § They are only slightly soluble in water and most of them are soluble in organic solvents.

Lewis Structure • The Lewis structure is a representation of a molecule that shows how the valence electrons are arranged among the atoms in the molecule.

Writing Lewis Structure • • Write the skeletal structure of the compound showing what atoms are bonded to what other atoms. Count the total number of valence electrons present, referring, if necessary to Lewis dot symbols. Draw a single covalent bond between the central atom and each of the surrounding atoms. Complete the octets of the atoms bonded to the central atom. If the octet rule is not met for the central atom, try double or triple bonds between the surrounding atoms and the central atom, using the lone pairs from the surrounding atoms.

• Example: Write the Lewis structure of nitric acid, HNO 3, in which the three O atoms are bonded to the central N atom and the ionizable H atom is bonded to one of the O atoms. • Answer: Step 1: The skeletal structure of HNO 3 is O N O H O

• Step 2: The outer-shell electron configurations of N, O, and H are N: 2 s 2 2 p 3 O: 2 s 2 2 p 4 H: 1 s 1 Thus, there are: 5 + (3 x 6) + 1 = 24, valence electrons to account for in HNO 3.
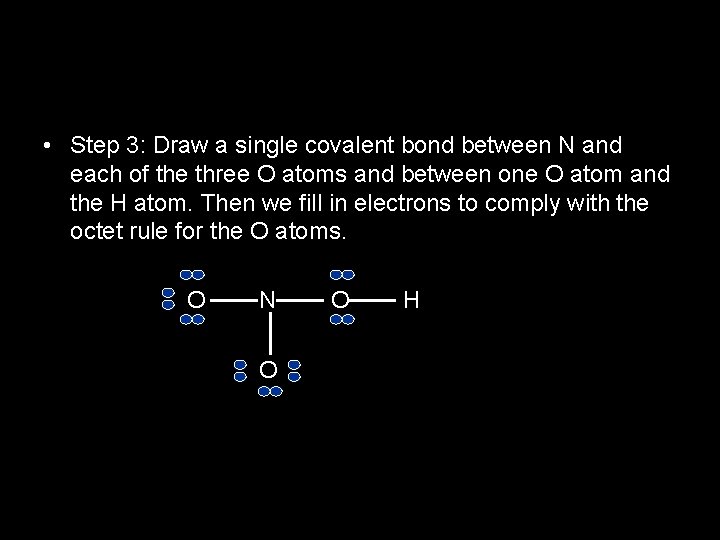
• Step 3: Draw a single covalent bond between N and each of the three O atoms and between one O atom and the H atom. Then we fill in electrons to comply with the octet rule for the O atoms. O N O O H
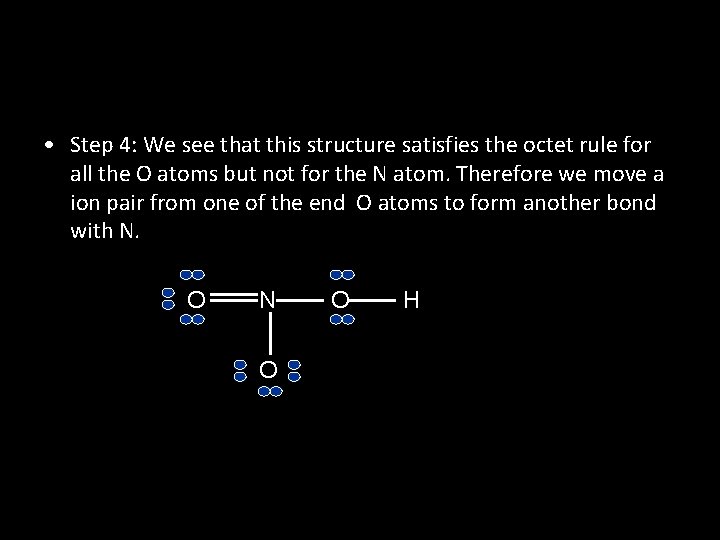
• Step 4: We see that this structure satisfies the octet rule for all the O atoms but not for the N atom. Therefore we move a ion pair from one of the end O atoms to form another bond with N. O N O O H

Formal Charge & Lewis Structure • Electrons are shared in a bond, we must divide the electrons in a bonding pair equally between the atoms forming the bond. The difference between the valence electrons in an isolated atom and the number of electrons assigned to that atom in a Lewis structure is called that atom’s formal charge. • The equation for calculating the formal charge on an atom in a molecule is given by: • FC = total number of valence electrons in the free atom – total number of nonbonding electrons - ½ (total number of bonding electrons)

• Example: Write formal charges of the carbonate ion! • Answer: The Lewis structure for carbonate ion 2 - O O C O

• The FC on the atoms can be calculated as follows: – The C atom : FC = 4 – 0 – ½ (8) = 0 – The O atom in C=O : FC = 6 – 4 – ½ (4) = 0 – The O atom in C-0 : FC = 6 – ½ (2) = -1 – Thus the Lewis formula for CO 32 - with FC is O O C O – Note that the sum of the FC is -2, the same as the charge on carbonate ion
- Slides: 45