CHEMICAL BONDING II MOLECULAR ORBITAL THEORY DO NOW

CHEMICAL BONDING II MOLECULAR ORBITAL THEORY

DO NOW ØPick up handout. ØGet out homework handout.

HOMEWORK ANSWERS #1 a. Br. F 5 VE = 42; 6 e- pairs: 5 bonded pairs and 1 lone pair; shape: square pyramid b. C 2 H 2 VE = 10; 2 e- pairs: 2 bonded pairs; shape: linear c. BF 3 VE = 24; 3 e- pairs: three bonded pairs; shape: trigonal planar d. SO 2 VE = 18; 3 e- pairs: two bonded pairs and one lone pair; shape: bent e. SCl 2 VE = 20; 4 e- pairs: two bonded pairs and 2 lone pairs; shape: bent

HOMEWORK ANSWERS #2 A. BF 3 three e- pairs sp 2 hybridization B. CCl 4 four e- pairs sp 3 hybridization C. As. F 5 five e- pairs dsp 3 hybridization D. Cl. O 3 -1 four e- pairs sp 3 hybridization
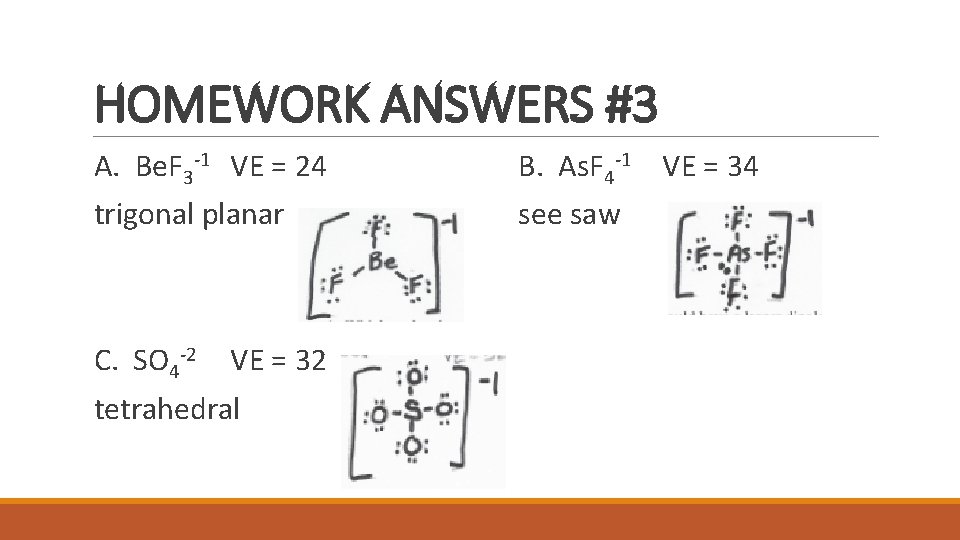
HOMEWORK ANSWERS #3 A. Be. F 3 -1 VE = 24 B. As. F 4 -1 VE = 34 trigonal planar see saw C. SO 4 -2 VE = 32 tetrahedral

HOMEWORK ANSWERS #4 - 6 4. HBr has the larger dipole moment. Br has a higher electronegativity that I so there is a greater difference between H-Br than H-I. 5. Molecules with no dipole moment: BCl 3 (trrgonal planar with three bonded pairs) and Be. Cl 2 (linear with two bonded pairs). 6. For a water molecule: c. the bonds are polar (eneg difference = 1. 4) and the molecule is polar (bent).

MOLECULAR ORBITAL MODEL Problems with LE Model 1. Electrons are not always localized as in the VSEPR theory; therefore resonance must be added and explained as best possible. 2. Molecules containing unpaired electrons are not easily dealt with using the localized model. 3. Magnetism is easily described for molecules using the MO theory. (Oxygen is paramagnetic which is unexplained by the localized electron model. ) 4. Bond energies are not easily related using the localized model.

MOLECULAR ORBITAL MODEL Be sure you know these terms. It will be tough to fill out the diagrams without them. TERMS TO KNOW: Bonding molecular orbital - an orbital lower in energy than the atomic orbitals from which it is composed (favors formation of molecule) represented by a B. Antibonding molecular orbital - an orbital higher in energy than the atomic orbitals from which it is composed (favors separated atoms) represented by a A or an *. Bond order - the difference between the number of bonding electrons and the number of antibonding electrons divided by two. Indicates bond strength.

MOLECULAR ORBITAL MODEL TERMS TO KNOW: Homonuclear diatomic molecules - those composed of two identical atoms. Heteronuclear diatomic molecules - those composed of two different atoms Paramagnetism - causes the substance to be drawn into a magnetic field; associated with unpaired electrons. Diamagnetic - causes the substance to be repelled by the magnetic field; associated with paired electrons.
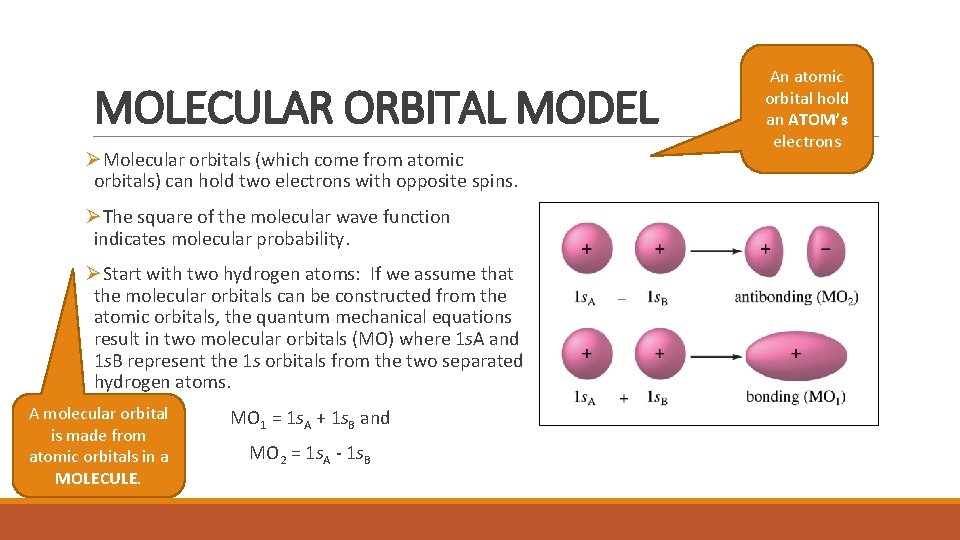
MOLECULAR ORBITAL MODEL ØMolecular orbitals (which come from atomic orbitals) can hold two electrons with opposite spins. ØThe square of the molecular wave function indicates molecular probability. ØStart with two hydrogen atoms: If we assume that the molecular orbitals can be constructed from the atomic orbitals, the quantum mechanical equations result in two molecular orbitals (MO) where 1 s. A and 1 s. B represent the 1 s orbitals from the two separated hydrogen atoms. A molecular orbital is made from atomic orbitals in a MOLECULE. MO 1 = 1 s. A + 1 s. B and MO 2 = 1 s. A - 1 s. B An atomic orbital hold an ATOM’s electrons

MOLECULAR ORBITAL MODEL The two hydrogen electrons The three orbital properties of interest are size, shape, and energy. AT THE RIGHT: - Each H entered on the outside with its lone 1 s electron. - As they approach each other, their two atomic orbitals blend to form two molecular orbitals. - One MO is of high energy and one MO is of low energy. - The electrons will choose the LOW energy route. - The electrons occupy the lower energy level and thus a bond is formed. Lower energy bonding orbital High energy antibonding orbital.
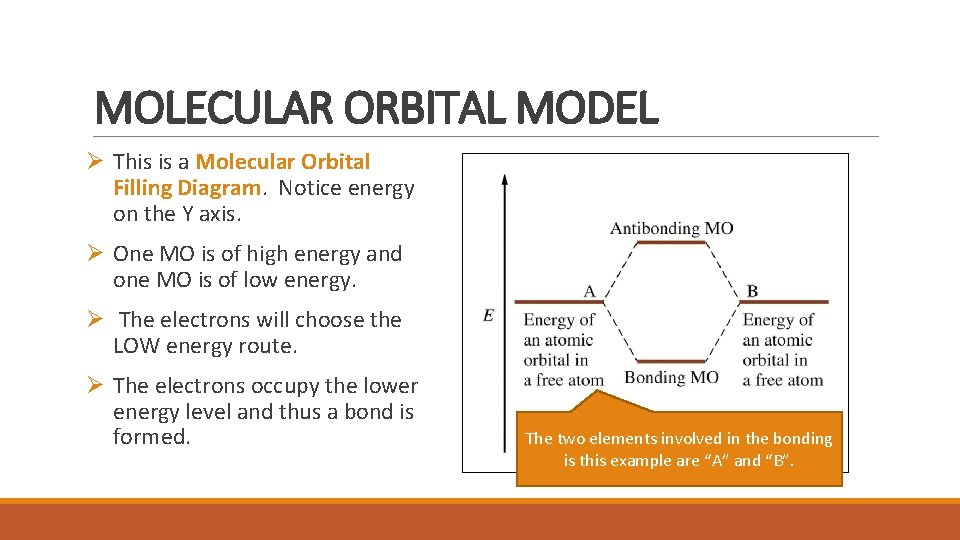
MOLECULAR ORBITAL MODEL Ø This is a Molecular Orbital Filling Diagram. Notice energy on the Y axis. Ø One MO is of high energy and one MO is of low energy. Ø The electrons will choose the LOW energy route. Ø The electrons occupy the lower energy level and thus a bond is formed. The two elements involved in the bonding is this example are “A” and “B”.

MOLECULAR ORBITAL MODEL Diatomic Helium, He 2 ØSince 4 electrons are involved, the first 2 (↑↓) get to be lazy and go to the low energy state. ØThe other 2 electrons(↑↓) must occupy the higher energy state and thus cancel out the bond. ØTherefore He 2 does not exist. One helium atom’s two electrons enter on the left, the other helium’s two electrons on the right.

MOLECULAR ORBITAL MODEL Read through these in your notes: 1. The electron probability of both molecular models is centered along a line passing through the two nuclei. For MO 1, the greatest probability is between the two nuclei (like constructive interference) and for MO 2, it is on either side of the nuclei (like destructive interference). This is the sigma (σ) bond - will be called the sigma (σ) molecular orbitals. 2. In the molecule, only the molecular orbitals exist. The 1 s atomic orbital no longer exists. 3. MO 1 is lower in energy than the 1 s orbital of a free hydrogen atom. MO 2 is higher in energy than the 1 s atomic orbital. The formation of a molecule is favored.

MOLECULAR ORBITAL MODEL 4. Electrons lower their energies by being attracted by both nuclei. 5. Labels on the molecular orbitals indicate their symmetry (shape), the parent atomic orbital, and whether they are bonding or antibonding. Bonding H 2 orbital - MO 1 = σ1 s Antibonding H 2 orbital - MO 2 = σ1 s* 6. Molecular electron configurations can be written just like atomic electron configurations. H 2 = σ1 s 2. 7. Each molecular orbital can hold two electrons with opposite spins. 8. Orbitals are conserved. The number of molecular orbitals is always the same and the atomic orbitals used to make them.

MOLECULAR ORBITAL MODEL Now you are ready! Watch this explanation. Watch: Introduction to Molecular Orbital Theory

MOLECULAR ORBITAL PRACTICE Get out the “Practice” sheet. We are going to fill in the energy diagram for F 2. Info on the next slide.
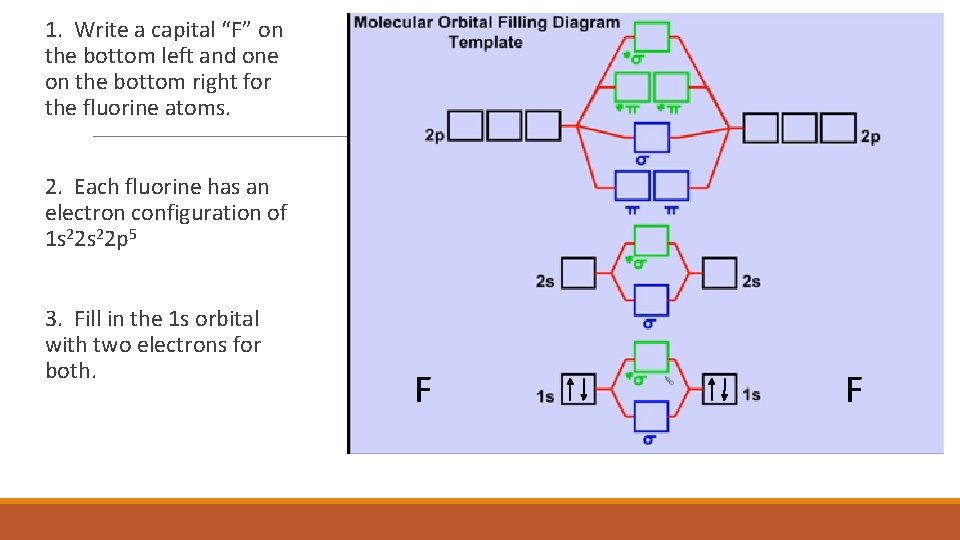
1. Write a capital “F” on the bottom left and one on the bottom right for the fluorine atoms. 2. Each fluorine has an electron configuration of 1 s 22 p 5 3. Fill in the 1 s orbital with two electrons for both. F F
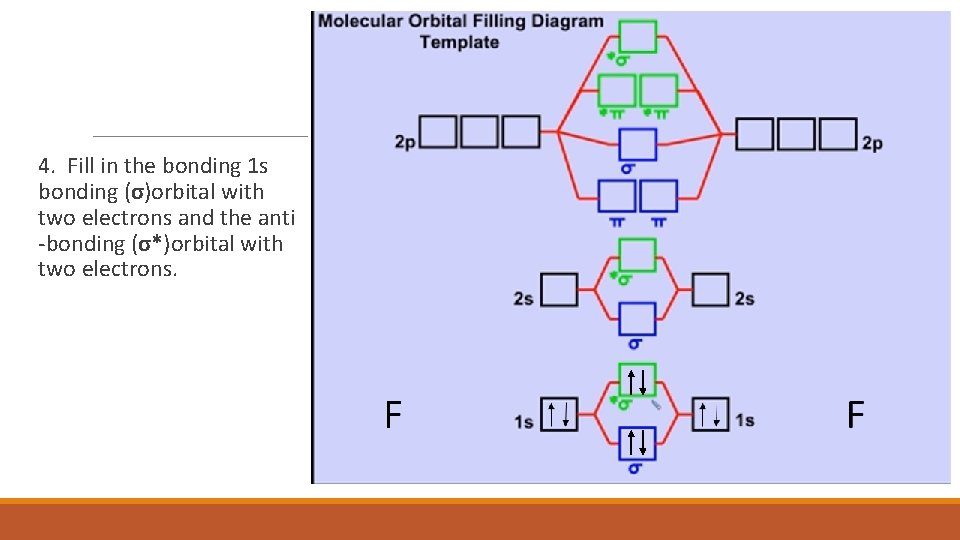
4. Fill in the bonding 1 s bonding (σ)orbital with two electrons and the anti -bonding (σ*)orbital with two electrons.
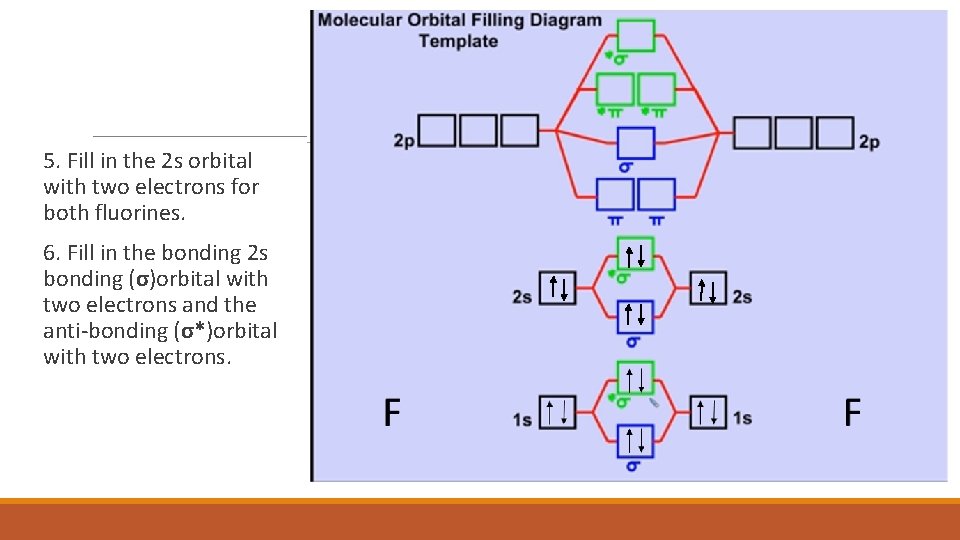
5. Fill in the 2 s orbital with two electrons for both fluorines. 6. Fill in the bonding 2 s bonding (σ)orbital with two electrons and the anti-bonding (σ*)orbital with two electrons.
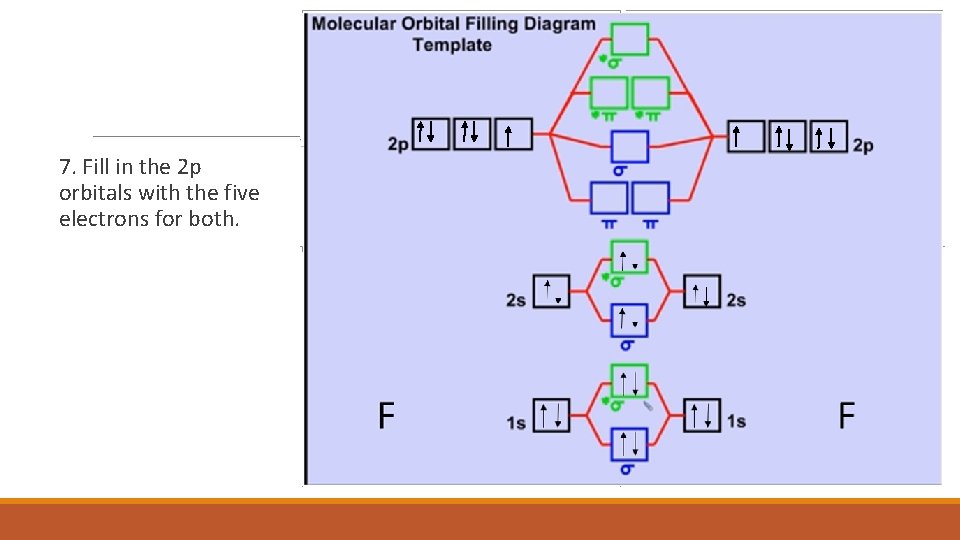
7. Fill in the 2 p orbitals with the five electrons for both.
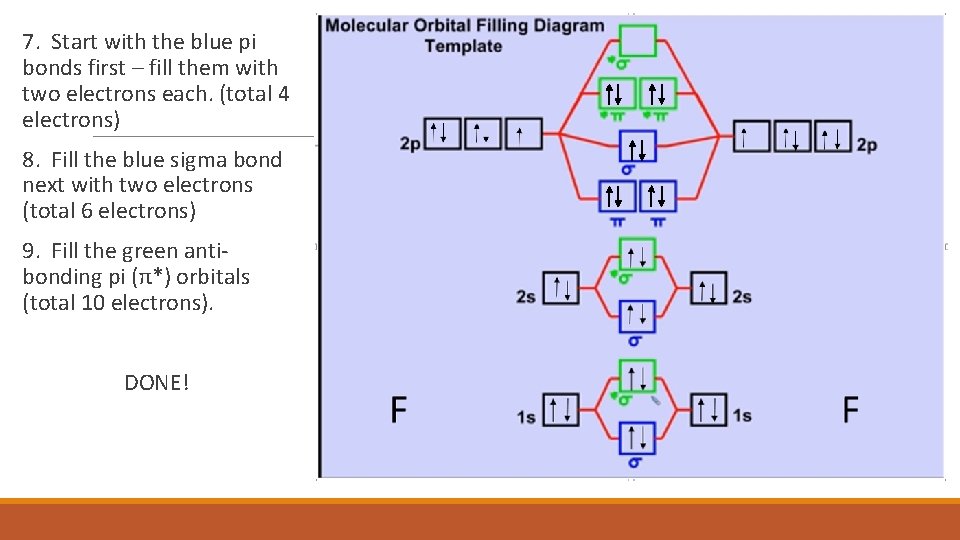
7. Start with the blue pi bonds first – fill them with two electrons each. (total 4 electrons) 8. Fill the blue sigma bond next with two electrons (total 6 electrons) 9. Fill the green antibonding pi (π*) orbitals (total 10 electrons). DONE!

MOLECULAR ORBITAL - PRACTICE Ø That is where you are going to stop with the handout. We will fill in the rest of it on Monday. Ø Your homework is to do the diagram (A only) on the MO Theory handout for the compounds given – both sides.
- Slides: 23