Chemical Bonding II Molecular Compounds I II IV
Chemical Bonding II. Molecular Compounds I II IV

B. Lewis Structures z. Octet Rule y. Most atoms form bonds in order to obtain 8 valence ey. Full energy level stability ~ Noble Gases Ne
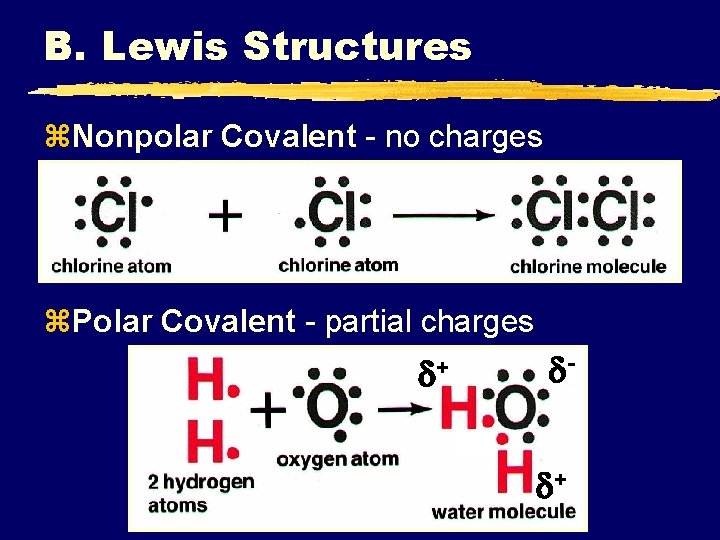
B. Lewis Structures z. Nonpolar Covalent - no charges z. Polar Covalent - partial charges + +

C. Molecular Nomenclature z. Prefix System (binary compounds) 1. How do we recognize these? Because they’re made up of 2 nonmetals? Where are nonmetals found? ? ? 2. Add prefixes to indicate # of atoms. Omit mono- prefix on first element. 3. Change the ending of the second element to -ide.
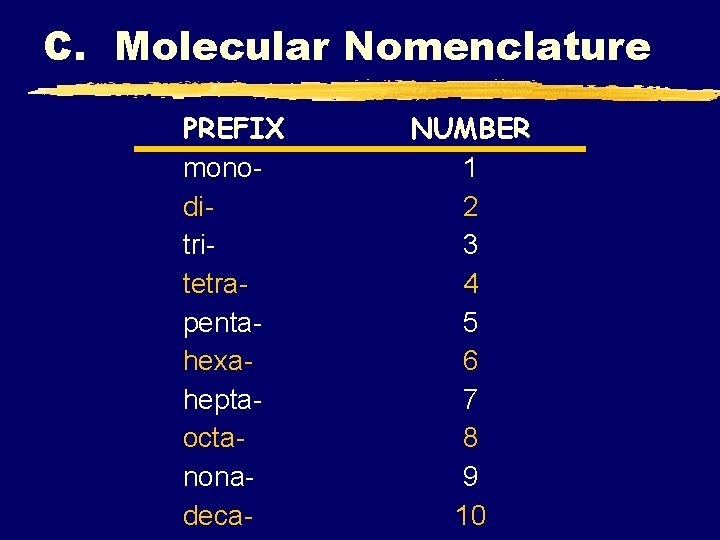
C. Molecular Nomenclature PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

Special Rules with Mono z If there is only one of the first element, then the prefix mono is not used. z If there is only one of the second element, then the prefix mono is necessary. z. Ex. z CO – Carbon Monoxide z CO 2 – Carbon Dioxide z C 2 O – Dicarbon Monoxide

Also…. z There is no reducing for covalent compounds. What you see is what you get. If you reduced, you would change the chemical formula and ratio of the elements in the bond…. Not good…. z C 2 H 4 - Dicarbon Tetrahydride z CH 2 - Carbon Dihydride z N 3 O 6 - Trinitrogen Hexaoxide z NO 2 - Nitrogen Dioxide

C. Molecular Nomenclature z. CCl 4 ycarbon tetrachloride z. N 2 O ydinitrogen monoxide z. SF 6 ysulfur hexafluoride

C. Molecular Nomenclature zarsenic trichloride y. As. Cl 3 zdinitrogen pentoxide y. N 2 O 5 ztetraphosphorus decoxide y. P 4 O 10
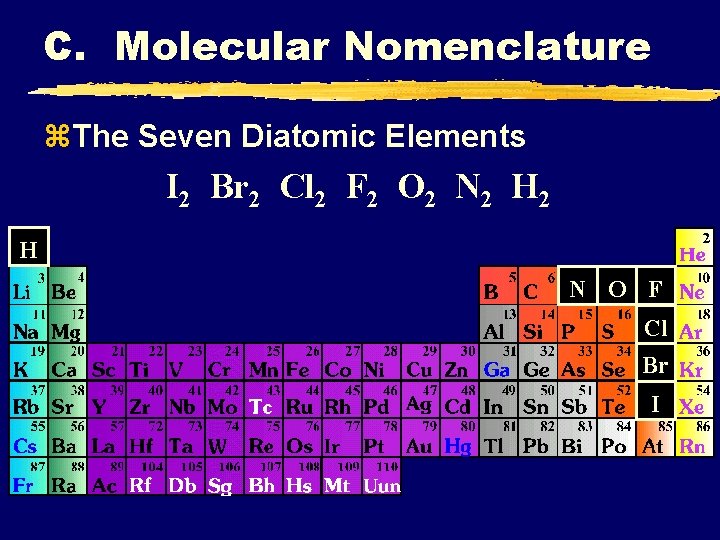
C. Molecular Nomenclature z. The Seven Diatomic Elements I 2 Br 2 Cl 2 F 2 O 2 N 2 H N O F Cl Br I
- Slides: 10