Chemical Bonding I Basic Concepts Copyright The Mc

Chemical Bonding I: Basic Concepts Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Chapter 9 • • 9. 1 Lewis dot symbols. 9. 2 The ionic bond. 9. 4 The covalent bond. 9. 5 Electronegativity. 9. 6 Writing Lewis structure. 9. 7 Formal charge and Lewis structure. 9. 8 The concept of resonance. 9. 9 Exceptions to the octet rule.
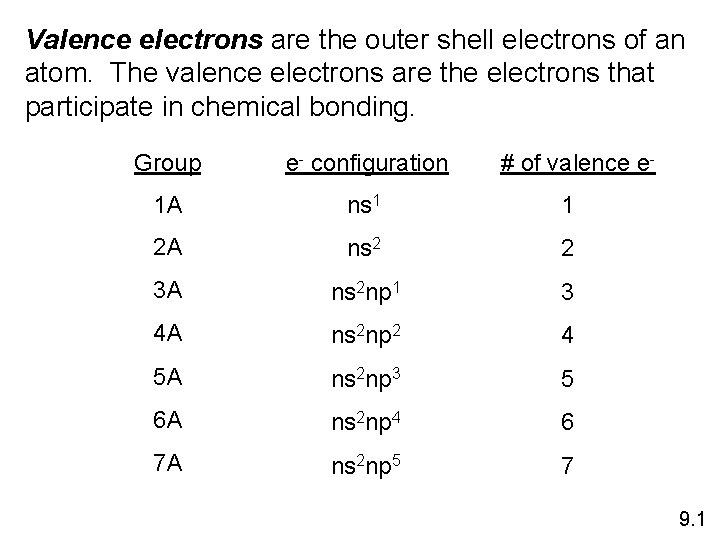
Valence electrons are the outer shell electrons of an atom. The valence electrons are the electrons that participate in chemical bonding. Group e- configuration # of valence e- 1 A ns 1 1 2 A ns 2 2 3 A ns 2 np 1 3 4 A ns 2 np 2 4 5 A ns 2 np 3 5 6 A ns 2 np 4 6 7 A ns 2 np 5 7 9. 1
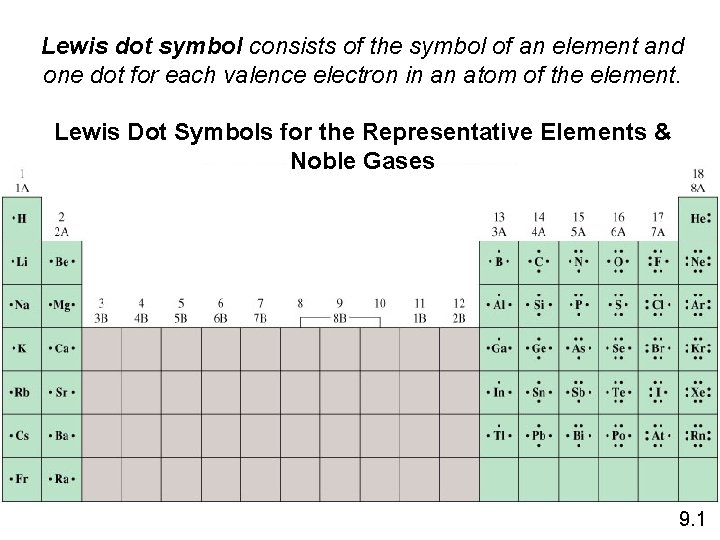
Lewis dot symbol consists of the symbol of an element and one dot for each valence electron in an atom of the element. Lewis Dot Symbols for the Representative Elements & Noble Gases 9. 1

The Ionic Bond is the electrostatic force that holds ions together in an ionic compound. Ionic compounds are compounds that contain only ionic bonds. Li + F 1 s 22 s 11 s 22 p 5 Li+ F [He] 1 s 1 s 2[2 Ne] 2 s 22 p 6 Li Li+ + e- e- + F F - Li+ + F - Li+ F - 9. 2

Chemistry In Action: Sodium Chloride Mining Salt Solar Evaporation for Salt

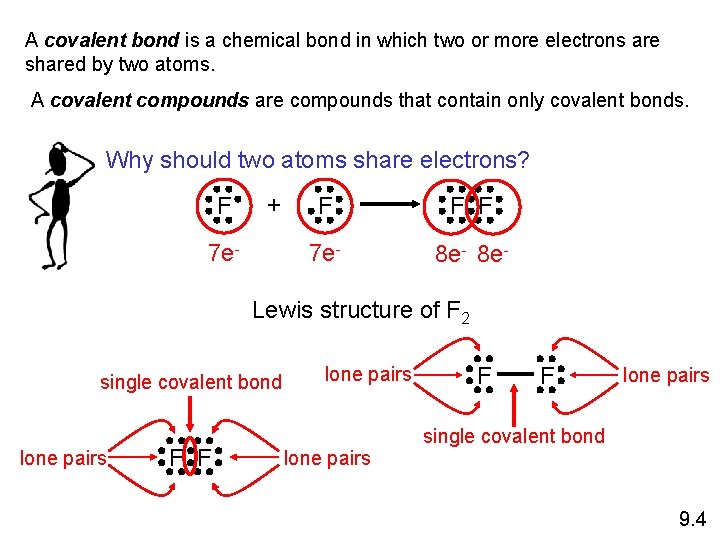
A covalent bond is a chemical bond in which two or more electrons are shared by two atoms. A covalent compounds are compounds that contain only covalent bonds. Why should two atoms share electrons? F + 7 e- F F F 7 e- 8 e- Lewis structure of F 2 single covalent bond lone pairs F F lone pairs single covalent bond 9. 4
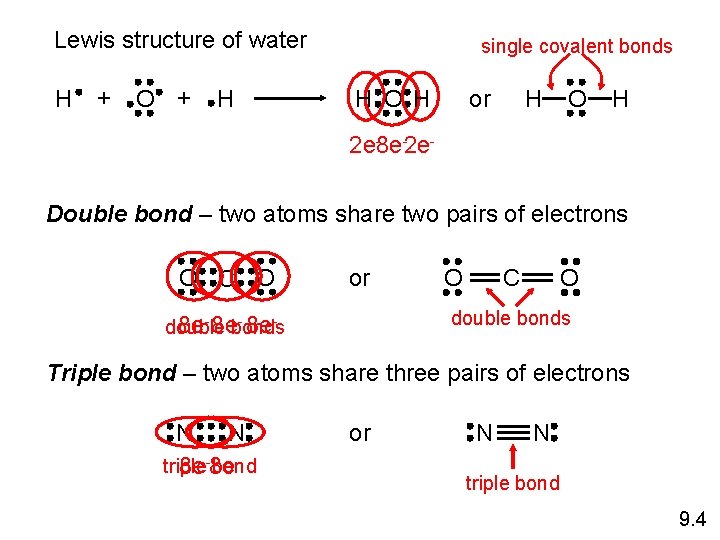
Lewis structure of water H + O + H single covalent bonds H O H or H O H 2 e-8 e-2 e. Double bond – two atoms share two pairs of electrons O C O or O O C double bonds 8 e- 8 edouble bonds Triple bond – two atoms share three pairs of electrons N N triple bond 8 e-8 e- or N N triple bond 9. 4
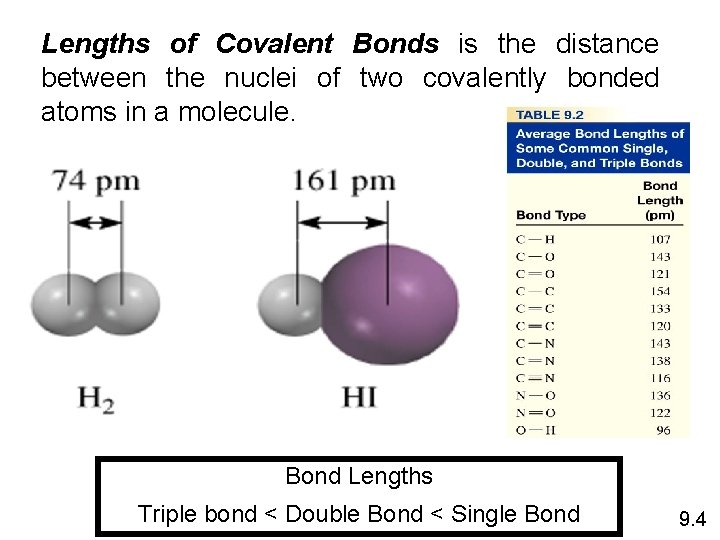
Lengths of Covalent Bonds is the distance between the nuclei of two covalently bonded atoms in a molecule. Bond Lengths Triple bond < Double Bond < Single Bond 9. 4
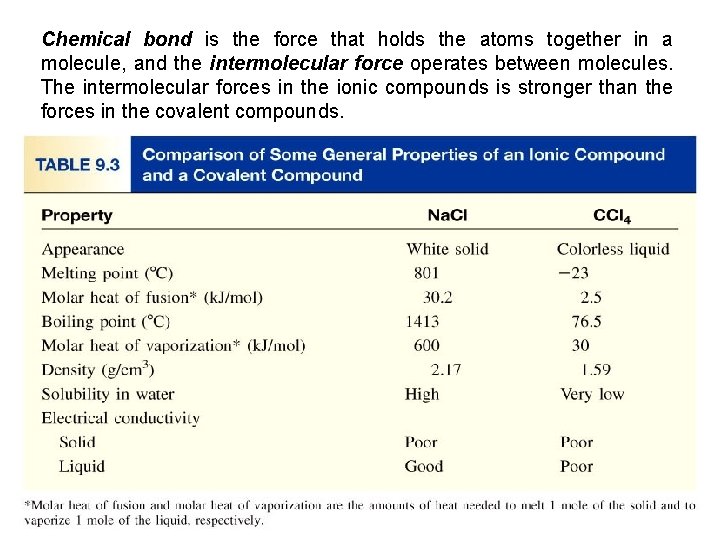
Chemical bond is the force that holds the atoms together in a molecule, and the intermolecular force operates between molecules. The intermolecular forces in the ionic compounds is stronger than the forces in the covalent compounds. 9. 4
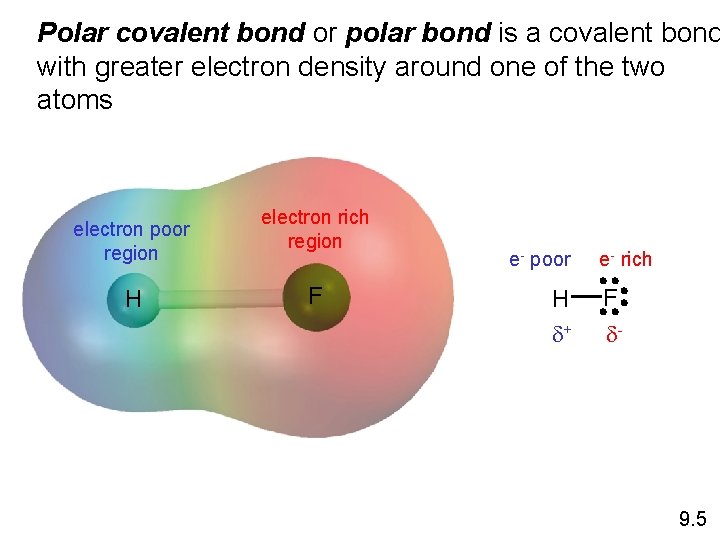
Polar covalent bond or polar bond is a covalent bond with greater electron density around one of the two atoms electron poor region H electron rich region F e- poor H d+ e- rich F d- 9. 5

Electronegativity is the ability of an atom to attract toward itself the electrons in a chemical bond. Electron Affinity - measurable, Cl is highest X (g) + e- X-(g) Electronegativity - relative, F is highest 9. 5
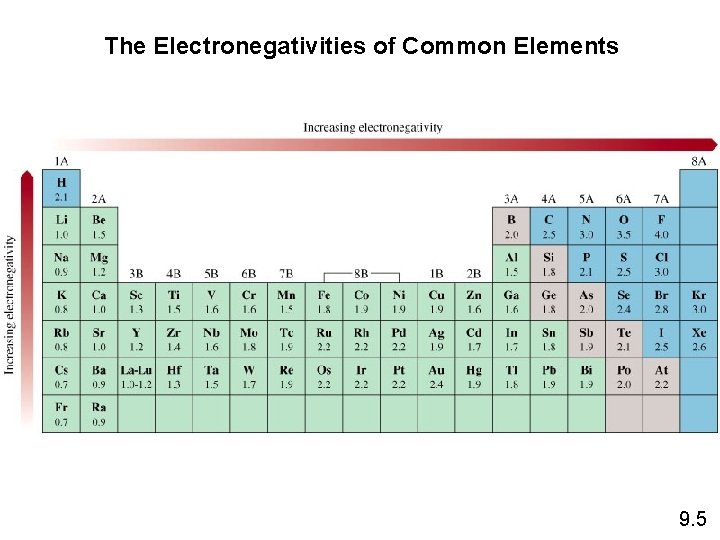
The Electronegativities of Common Elements 9. 5
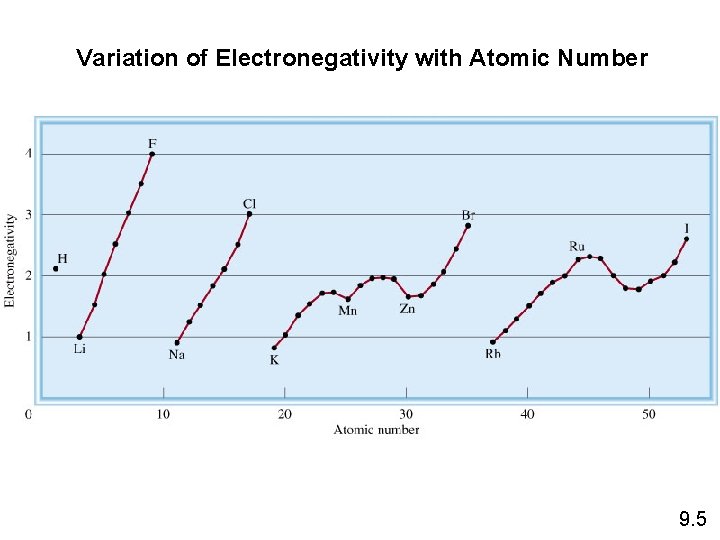
Variation of Electronegativity with Atomic Number 9. 5
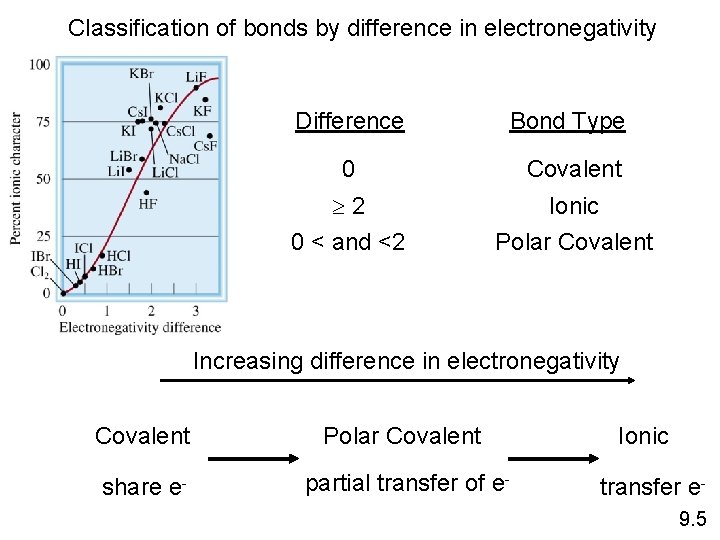
Classification of bonds by difference in electronegativity Difference Bond Type 0 Covalent 2 0 < and <2 Ionic Polar Covalent Increasing difference in electronegativity Covalent Polar Covalent share e- partial transfer of e- Ionic transfer e 9. 5

Classify the following bonds as ionic, polar covalent, or covalent: The bond in Cs. Cl; the bond in H 2 S; and the NN bond in H 2 NNH 2. Cs – 0. 7 Cl – 3. 0 – 0. 7 = 2. 3 Ionic H – 2. 1 S – 2. 5 – 2. 1 = 0. 4 Polar Covalent N – 3. 0 – 3. 0 = 0 Covalent 9. 5

Worked Example 9. 2

Writing Lewis Structures 1. Draw skeletal structure of compound showing what atoms are bonded to each other. Put least electronegative element in the center. 2. Count total number of valence e-. Add 1 for each negative charge. Subtract 1 for each positive charge. 3. Complete an octet for all atoms except hydrogen 4. If structure contains too many electrons, form double and triple bonds on central atom as needed. 9. 6
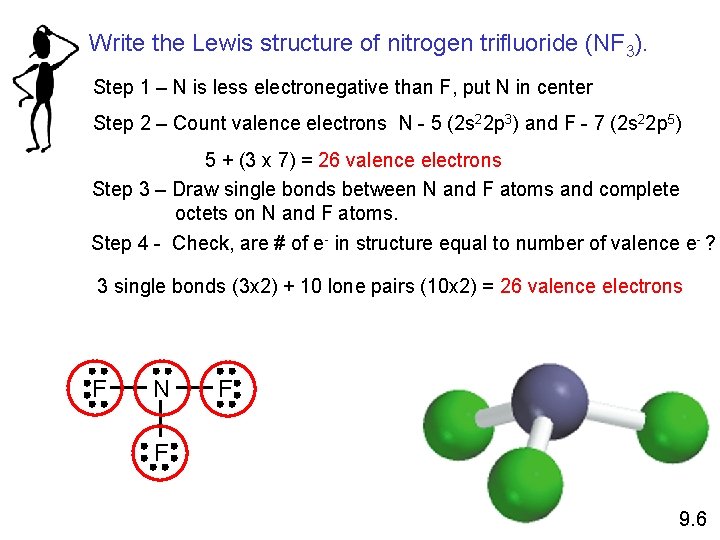
Write the Lewis structure of nitrogen trifluoride (NF 3). Step 1 – N is less electronegative than F, put N in center Step 2 – Count valence electrons N - 5 (2 s 22 p 3) and F - 7 (2 s 22 p 5) 5 + (3 x 7) = 26 valence electrons Step 3 – Draw single bonds between N and F atoms and complete octets on N and F atoms. Step 4 - Check, are # of e- in structure equal to number of valence e- ? 3 single bonds (3 x 2) + 10 lone pairs (10 x 2) = 26 valence electrons F N F F 9. 6

Write the Lewis structure of the carbonate ion (CO 32 -). Step 1 – C is less electronegative than O, put C in center Step 2 – Count valence electrons C - 4 (2 s 22 p 2) and O - 6 (2 s 22 p 4) -2 charge – 2 e- 4 + (3 x 6) + 2 = 24 valence electrons Step 3 – Draw single bonds between C and O atoms and complete octet on C and O atoms. Step 4 - Check, are # of e- in structure equal to number of valence e- ? 3 single bonds (3 x 2) + 10 lone pairs (10 x 2) = 26 valence electrons Step 5 - Too many electrons, form double bond and re-check # of e- O C O O 2 single bonds (2 x 2) = 4 1 double bond = 4 8 lone pairs (8 x 2) = 16 Total = 24 9. 6


Worked Example 9. 4

Worked Example 9. 5
- Slides: 24