Chemical Bonding Hydrogen atom based atomic orbitals a
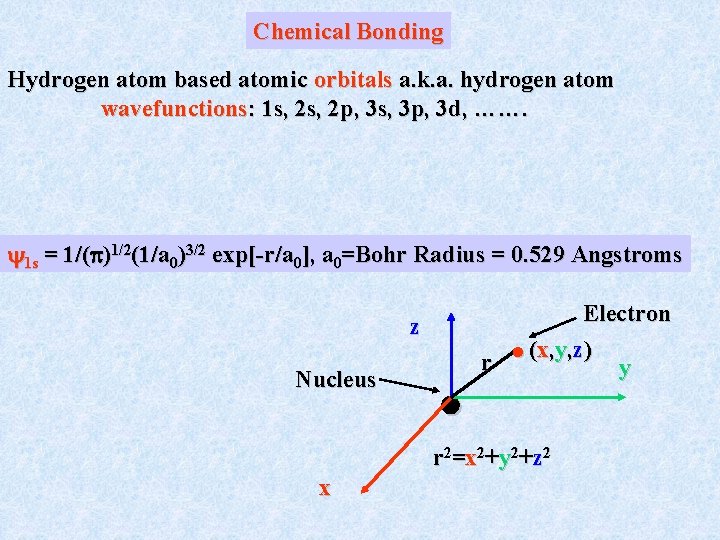
Chemical Bonding Hydrogen atom based atomic orbitals a. k. a. hydrogen atom wavefunctions: 1 s, 2 p, 3 s, 3 p, 3 d, ……. 1 s = 1/( )1/2(1/a 0)3/2 exp[-r/a 0], a 0=Bohr Radius = 0. 529 Angstroms z Nucleus Electron (x, y, z) r y r 2=x 2+y 2+z 2 x

Orbitals, Wavefunctions and Probabilities The orbital or wavefunction is just a mathematical function that can have a magnitude and sign (e. g. + 0. 1 or -0. 2) at a given point r in space. Probability of finding a 1 s electron at a particular point in space is often not as interesting as finding the electron in a thin shell between r and r+dr.
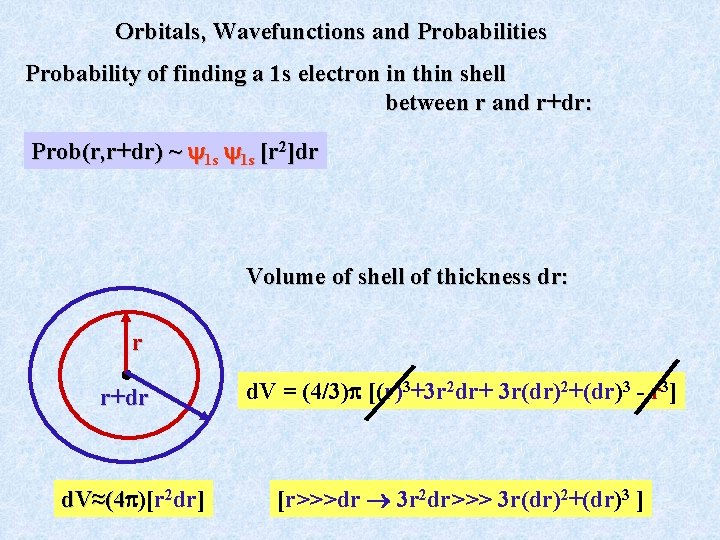
Orbitals, Wavefunctions and Probabilities Probability of finding a 1 s electron in thin shell between r and r+dr: Prob(r, r+dr) ~ 1 s 1 s [r 2]dr Volume of shell of thickness dr: r r+dr 2 dr] d. V≈(4 )[r ≈(4 d. V = (4/3) [(r)3+3 r 2 dr+ 3 r(dr)2+(dr)3 - r 3] [r>>>dr 3 r 2 dr>>> 3 r(dr)2+(dr)3 ]
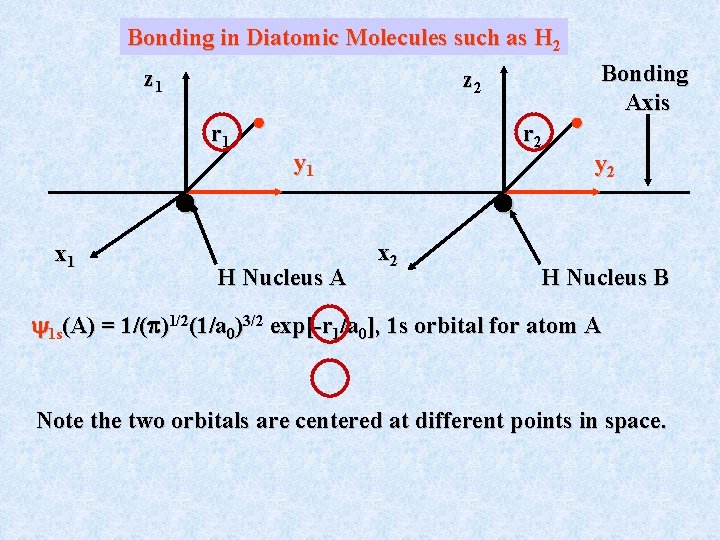
Bonding in Diatomic Molecules such as H 2 z 1 z 2 r 1 x 1 r 2 y 1 H Nucleus A x 2 Bonding Axis y 2 H Nucleus B 1 s(A) = 1/( )1/2(1/a 0)3/2 exp[-r 1/a 0], 1 s orbital for atom A Note the two orbitals are centered at different points in space.
![1 s = C 1[ 1 s(A) + 1 s(B)], Sigma 1 s 1 s = C 1[ 1 s(A) + 1 s(B)], Sigma 1 s](http://slidetodoc.com/presentation_image/ddf7c7baf6e9001cad564249e092c3d6/image-5.jpg)
1 s = C 1[ 1 s(A) + 1 s(B)], Sigma 1 s Bonding Molecular orbital. C 1 is a constant. Note that probabilities for finding electron at some position in space scale like [ 1 s]2 and [ 1 s*]2: [ 1 s*]2 = {C 2[ 1 s(A) - 1 s(B)]}2 = (C 2)2{[ 1 s(A)]2 + [ 1 s(B)]2 - 2[ 1 s(A)][ 1 s(B)]} “Non-interacting” part is result for large separation between nucleus A and B [ 1 s]2 = {C 1[ 1 s(A) + 1 s(B)]}2 = (C 1)2{[ 1 s(A)]2 + [ 1 s(B)]2 + 2[ 1 s(A)][ 1 s(B)]}

Notational Detail Oxtoby uses two different notations for orbitals in the 4 th and 5 th editions of the class text: 1 s in i the 4 th edition becomes g 1 s in the 5 th edition 1 s* in i the 4 th edition becomes u 1 s* in the 5 th edition The addition of g and u provides some extra identification of the orbitals and is the one encountered in the professional literature. g and u are from the German “gerade” and “ungerade”
![Wave Functions 1 s(A) H + 2 Electron Densities * ] [ 1 s Wave Functions 1 s(A) H + 2 Electron Densities * ] [ 1 s](http://slidetodoc.com/presentation_image/ddf7c7baf6e9001cad564249e092c3d6/image-7.jpg)
Wave Functions 1 s(A) H + 2 Electron Densities * ] [ 1 s * = C [ A - B ] 2 1 s 1 s + B A - 1 s ANTIBONDING - 1 s(B) A Pushes e- away from region between nuclei A and B B 1 s = C 1 [ 1 s + 1 s ] + + A B 1 s(A) + A B - 2[ 1 s(A)][ 1 s(B)] + 2[ 1 s(A)][ 1 s(B)] [ 1 s ] 2 BONDING Pushes e- between nuclei A and B 1 s(B) + 2 A 2 (n. i. ) ~ B 2 1 s 1 s NON-INTERACTING A 2 [( A ) + ( B ) ] B
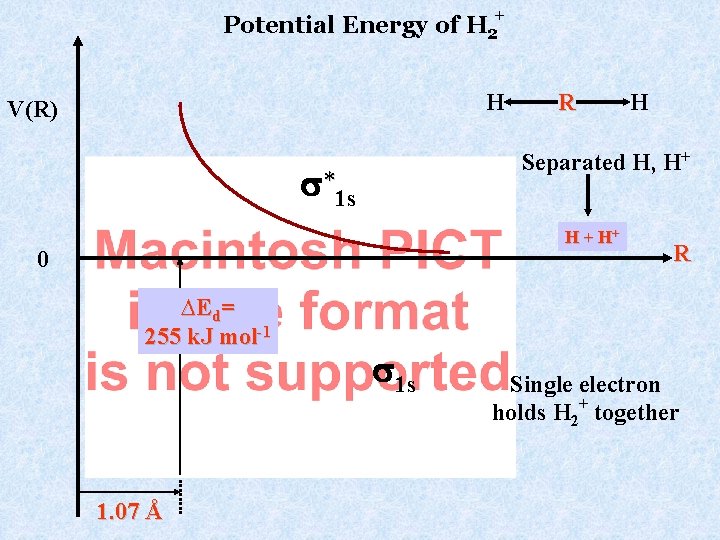
Potential Energy of H 2+ H V(R) R Separated H, H+ *1 s H + H+ 0 ∆Ed= 255 k. J mol-1 1. 07 Å H 1 s R Single electron holds H 2+ together
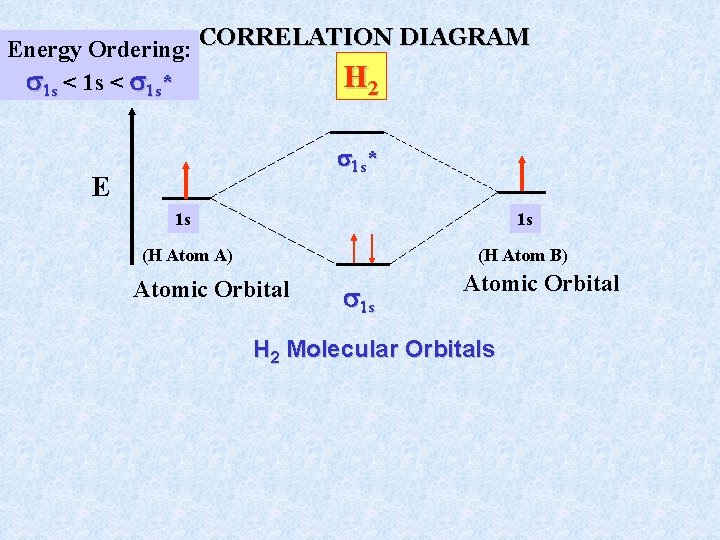
Energy Ordering: CORRELATION DIAGRAM H 2 1 s < 1 s* 1 s * E 1 s 1 s (H Atom A) (H Atom B) Atomic Orbital 1 s Atomic Orbital H 2 Molecular Orbitals
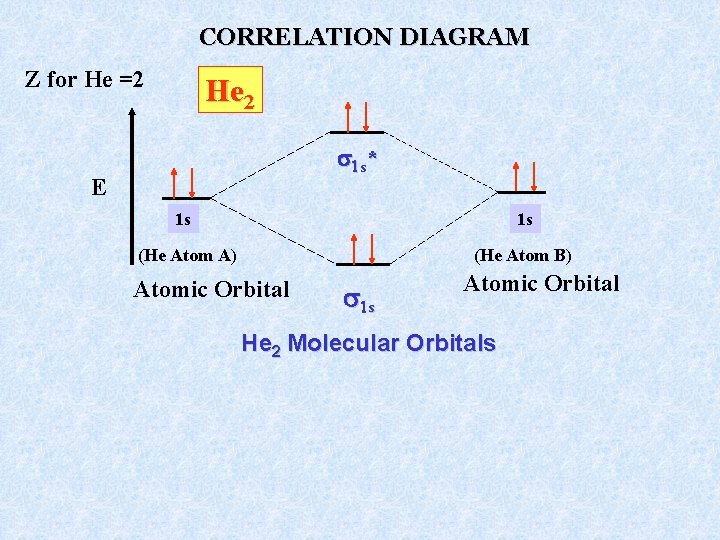
CORRELATION DIAGRAM Z for He =2 He 2 1 s * E 1 s 1 s (He Atom A) (He Atom B) Atomic Orbital 1 s Atomic Orbital He 2 Molecular Orbitals
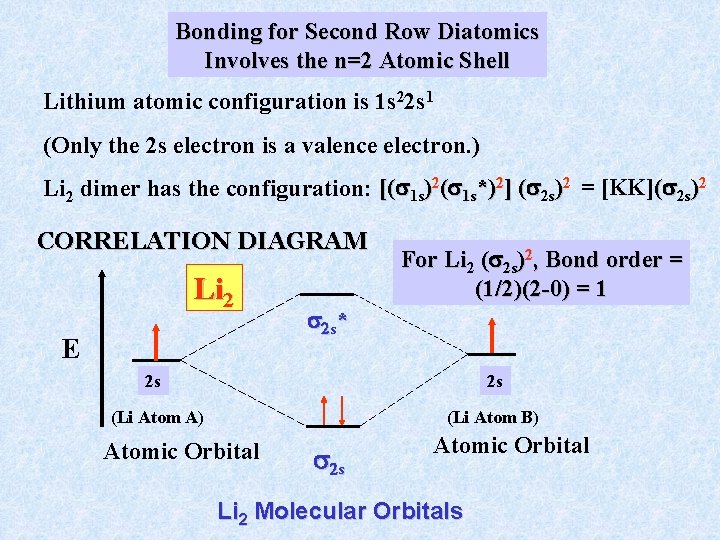
Bonding for Second Row Diatomics Involves the n=2 Atomic Shell Lithium atomic configuration is 1 s 22 s 1 (Only the 2 s electron is a valence electron. ) Li 2 dimer has the configuration: [( 1 s)2( 1 s*)2] ( 2 s)2 = [KK]( 2 s)2 CORRELATION DIAGRAM Li 2 E For Li 2 ( 2 s)2, Bond order = (1/2)(2 -0) = 1 2 s * 2 s 2 s (Li Atom A) (Li Atom B) Atomic Orbital 2 s Atomic Orbital Li 2 Molecular Orbitals

Bonding for Second Row Diatomics: the Role of 2 p Orbitals Once the 2 s, 2 s* molecular orbitals formed from the 2 s atomic orbitals on each atom are filled (4 electrons, Be 2), we must consider the role of the 2 p electrons (B 2 is first diatomic using 2 p electrons). There are 3 different sets of p orbitals (2 px, 2 py, and 2 pz), all mutually perpendicular. If we choose the molecular diatomic axis to be the z axis (this is arbitrary), we have a picture like this:
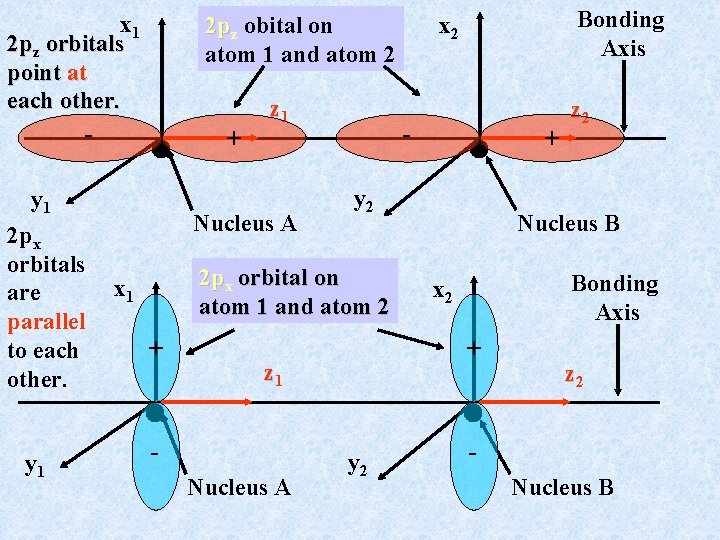
x 1 2 pz orbitals point at each other. - y 1 Nucleus A y 2 + z 2 Nucleus B Bonding Axis x 2 + 2 px orbital on atom 1 and atom 2 x 1 - + y 1 2 px orbitals are parallel to each other. + z 1 Bonding Axis x 2 2 pz obital on atom 1 and atom 2 z 1 Nucleus A y 2 z 2 Nucleus B
- Slides: 13