Chemical bonding Elements combine to form compounds The
Chemical bonding • Elements combine to form compounds • The forces of attraction which holds atoms together are known as chemical bonds
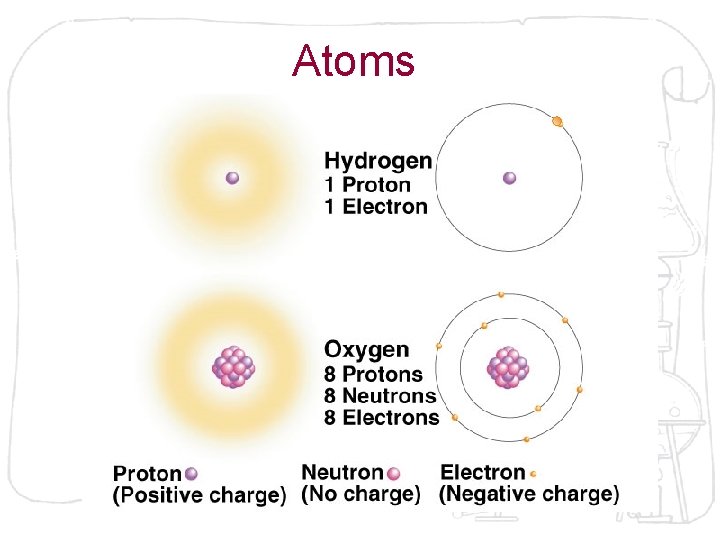
Atoms

Electrons and Atomic Behavior • Orbital refers to the area around a nucleus where an electron is most likely found. • Chemical behavior of an atom is determined by the number and arrangement of its orbitals. • Electrons are attracted to the positively charged nucleus, thus it takes energy to hold electrons in place.
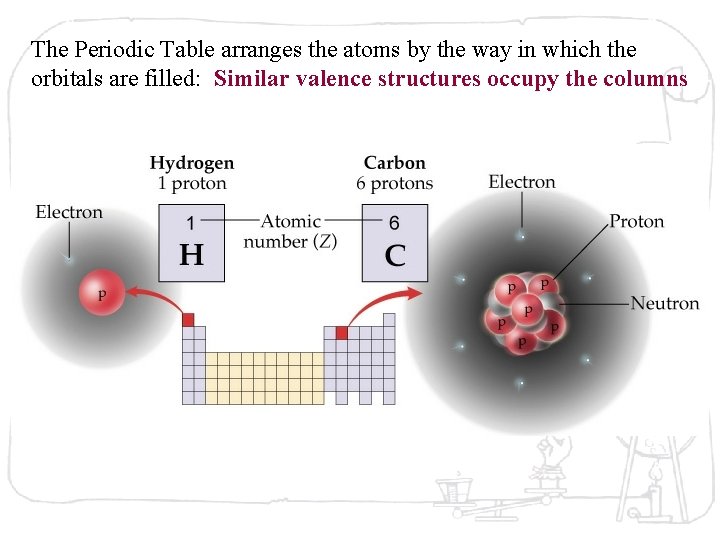
The Periodic Table arranges the atoms by the way in which the orbitals are filled: Similar valence structures occupy the columns
Periodic Table Noble gases
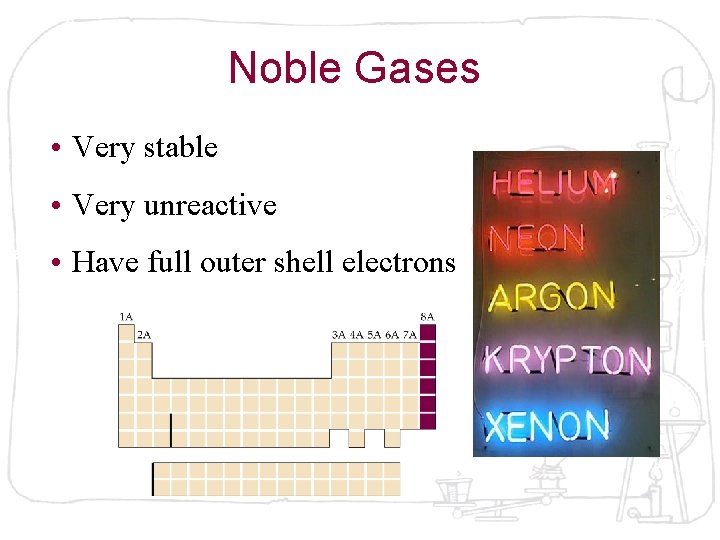
Noble Gases • Very stable • Very unreactive • Have full outer shell electrons

The Octet Rule Atoms tend to gain, lose, or share electrons until they have eight outer shell (valence) electrons. 8
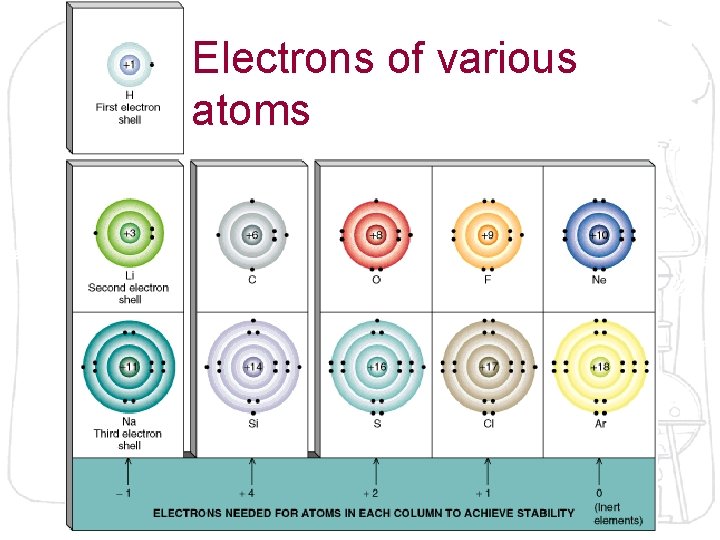
Electrons of various atoms
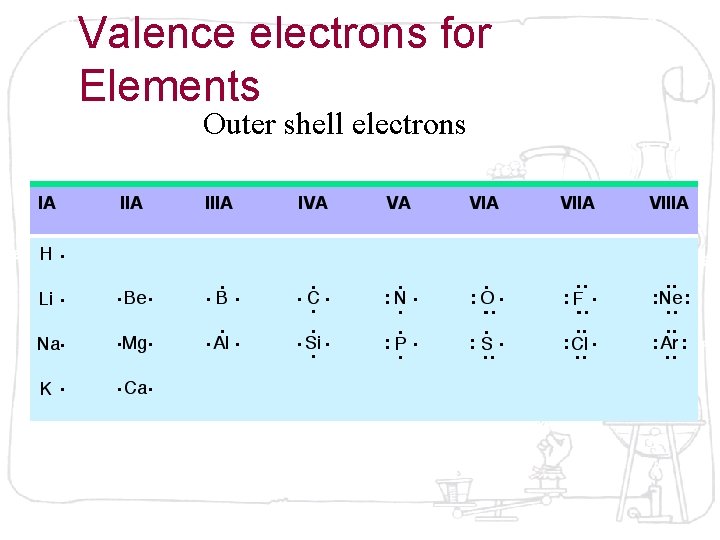
Valence electrons for Elements Outer shell electrons
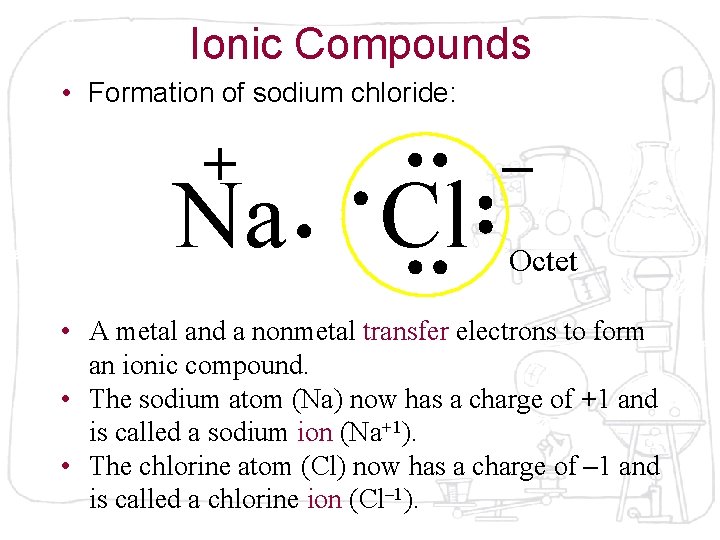
Ionic Compounds • Formation of sodium chloride: + Na Cl – Octet • A metal and a nonmetal transfer electrons to form an ionic compound. • The sodium atom (Na) now has a charge of +1 and is called a sodium ion (Na+1). • The chlorine atom (Cl) now has a charge of – 1 and is called a chlorine ion (Cl– 1).
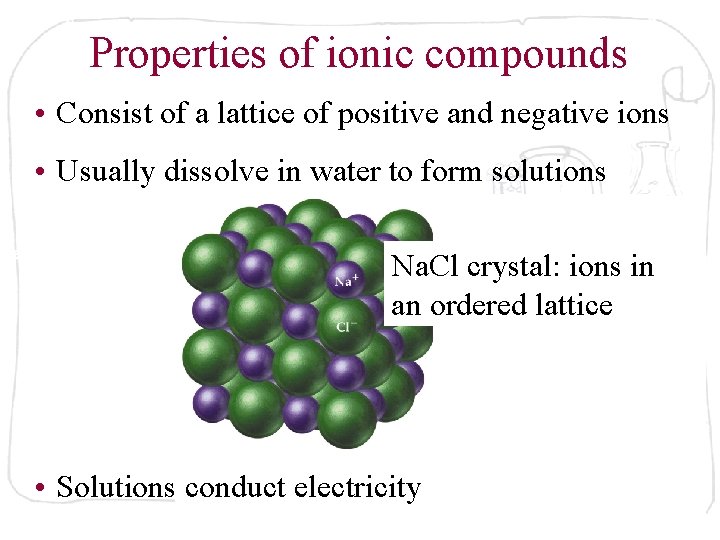
Properties of ionic compounds • Consist of a lattice of positive and negative ions • Usually dissolve in water to form solutions Na. Cl crystal: ions in an ordered lattice • Solutions conduct electricity
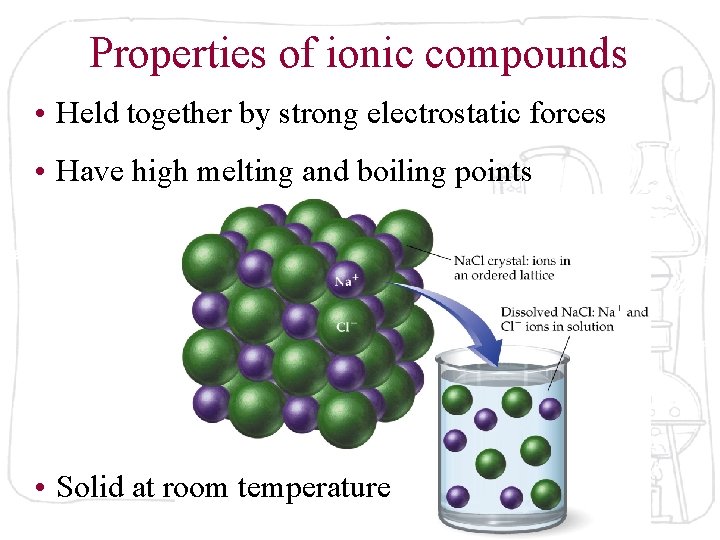
Properties of ionic compounds • Held together by strong electrostatic forces • Have high melting and boiling points • Solid at room temperature

Covalent Bonds

In covalent bonding, atoms still want to achieve a noble gas configuration (the octet rule). But rather than losing or gaining electrons, atoms now share an electron pair. The shared electron pair is called a bonding pair.

Cl 2 Chlorine forms a covalent bond with itself

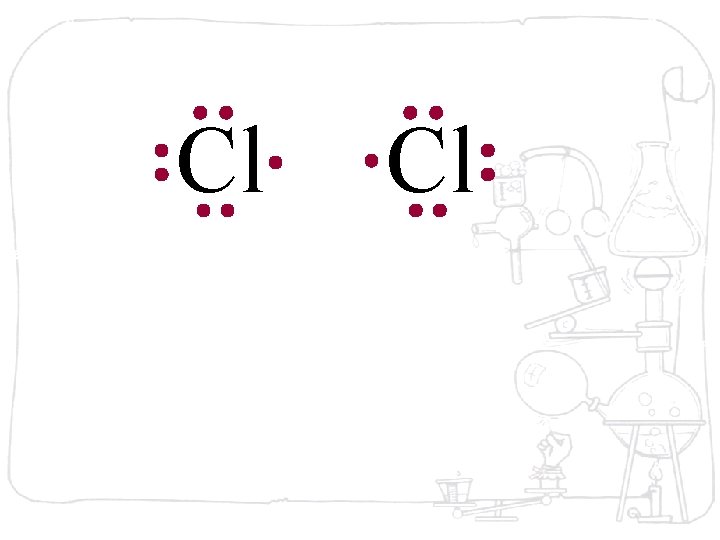
Cl Cl Each chlorine atom wants to gain one electron to achieve an octet

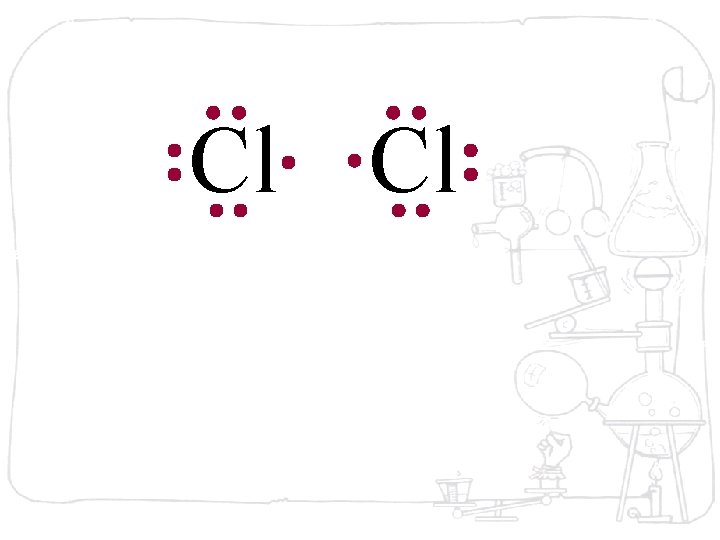
Cl Cl Neither atom will give up an electron – chlorine is highly electronegative. What’s the solution – what can they do to achieve an octet?
Cl Cl
Cl Cl
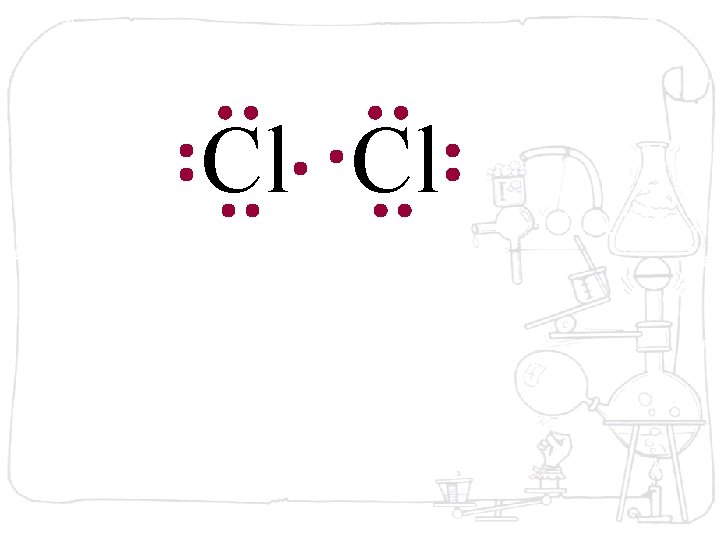
Cl Cl
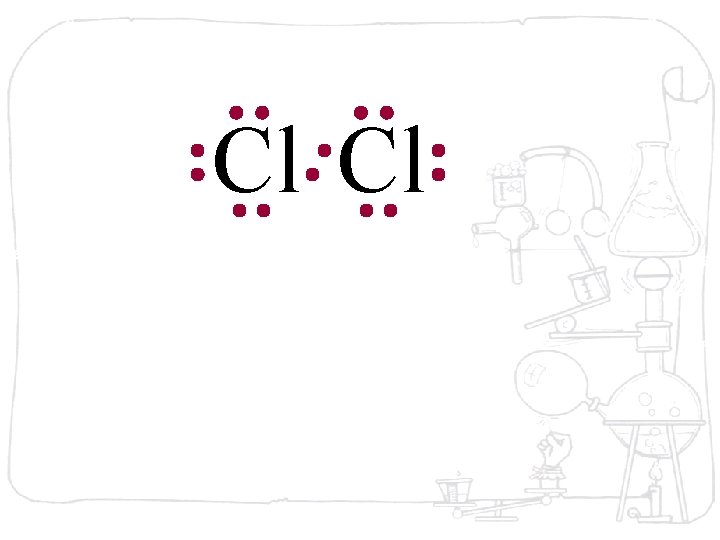
Cl Cl
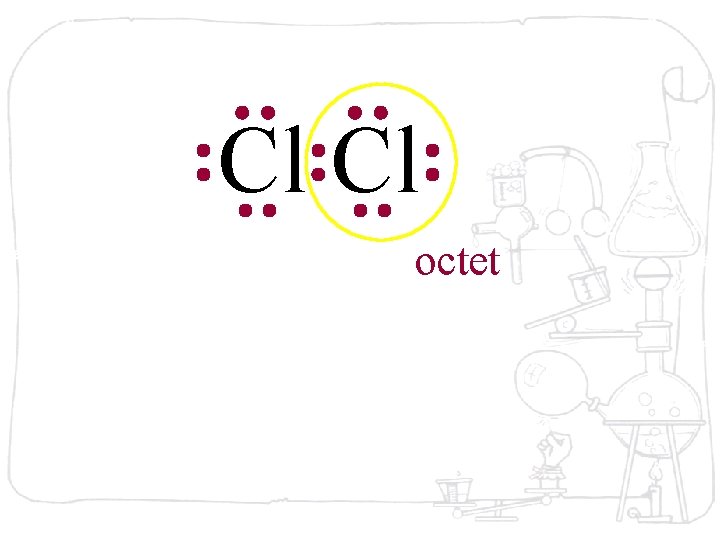
Cl Cl octet
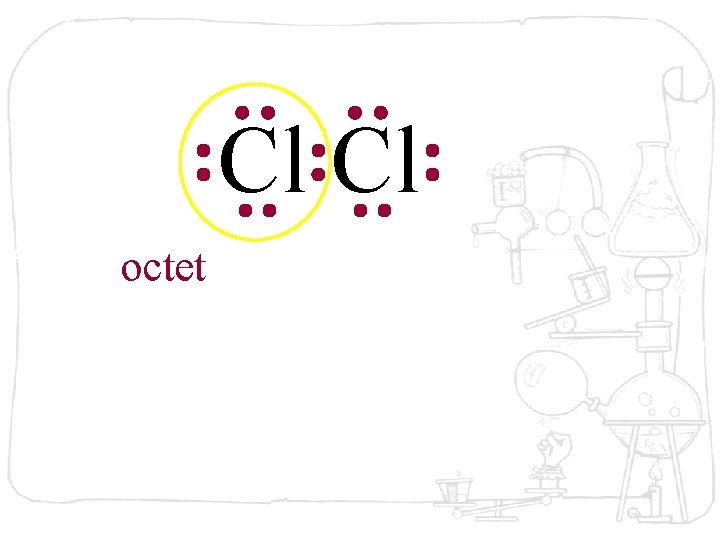
Cl Cl octet
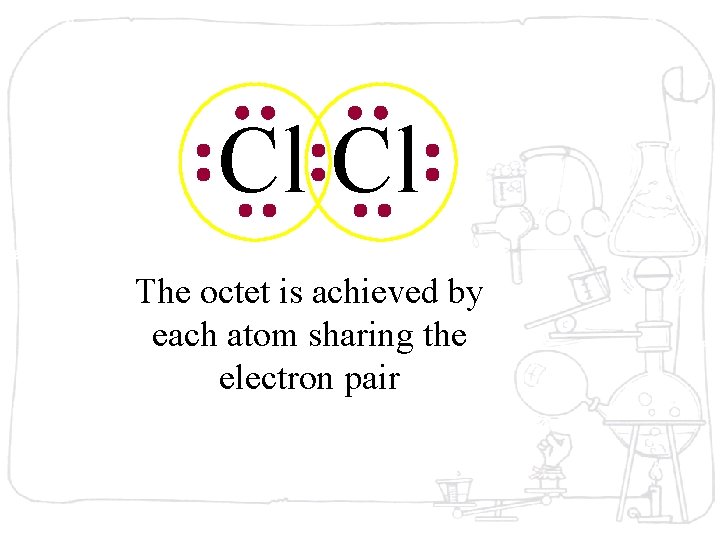
Cl Cl The octet is achieved by each atom sharing the electron pair
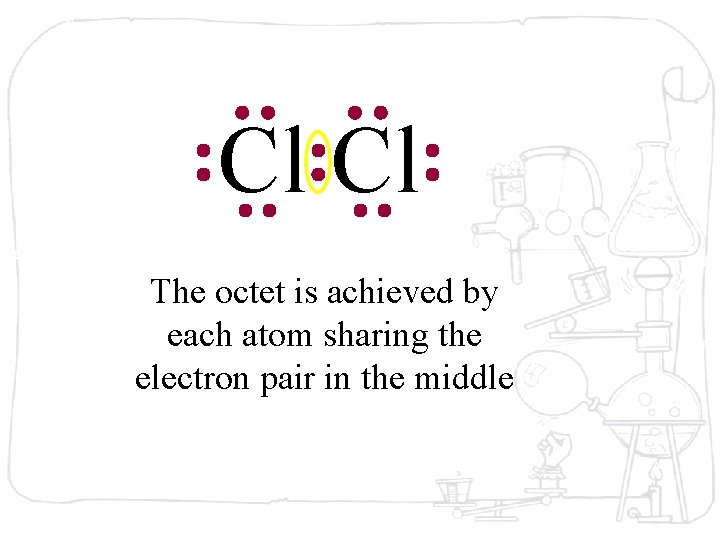
Cl Cl The octet is achieved by each atom sharing the electron pair in the middle
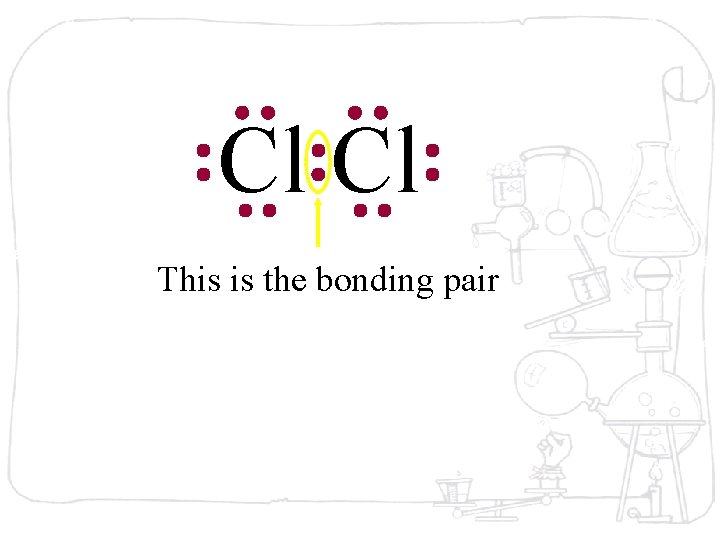
Cl Cl This is the bonding pair

Cl Cl It is called a Single bond

Cl Cl Single bonds are abbreviated with a dash
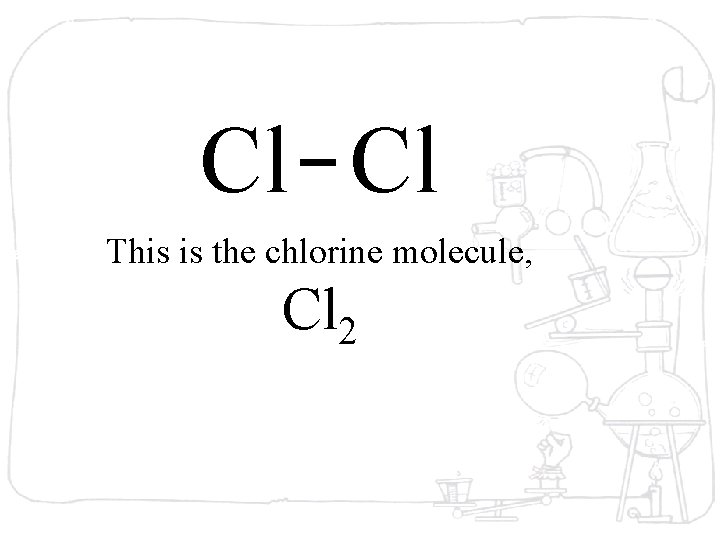
Cl Cl This is the chlorine molecule, Cl 2
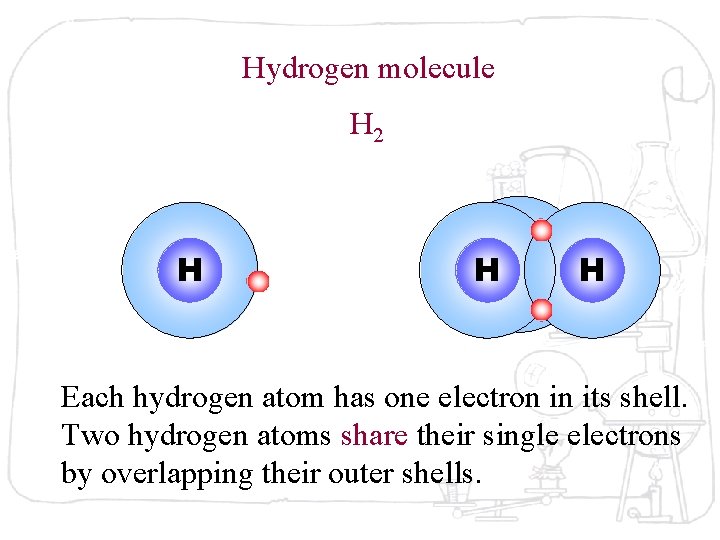
Hydrogen molecule H 2 H HH H Each hydrogen atom has one electron in its shell. Two hydrogen atoms share their single electrons by overlapping their outer shells.
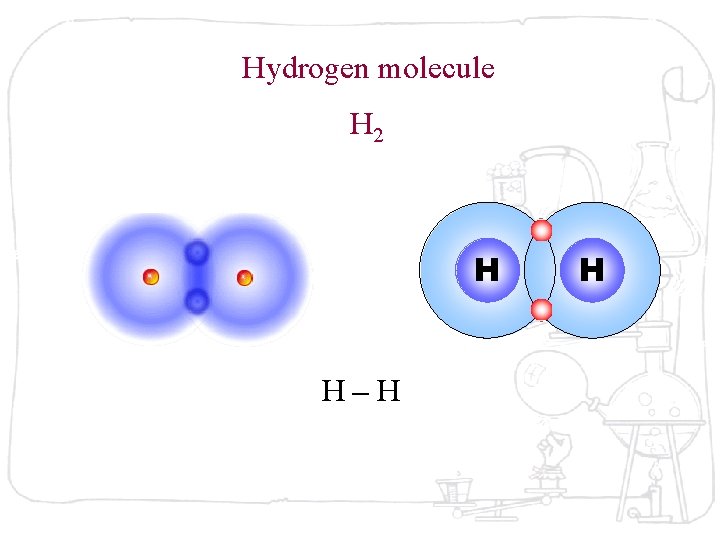
Hydrogen molecule H 2 H H–H H
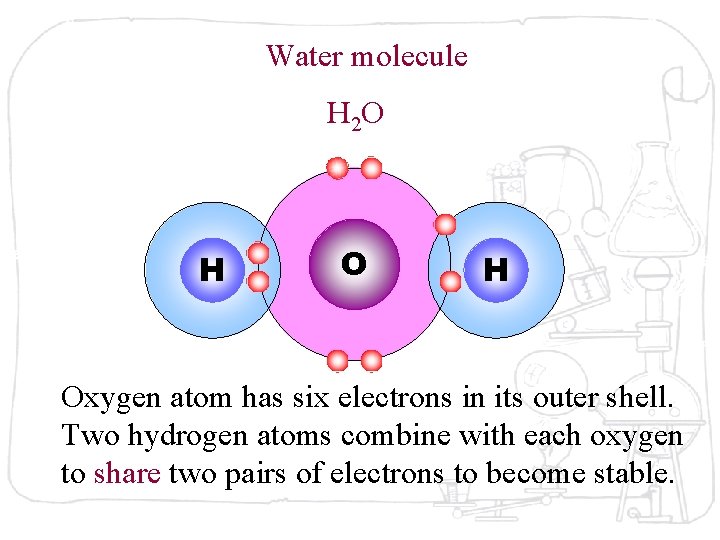
Water molecule H 2 O H Oxygen atom has six electrons in its outer shell. Two hydrogen atoms combine with each oxygen to share two pairs of electrons to become stable.
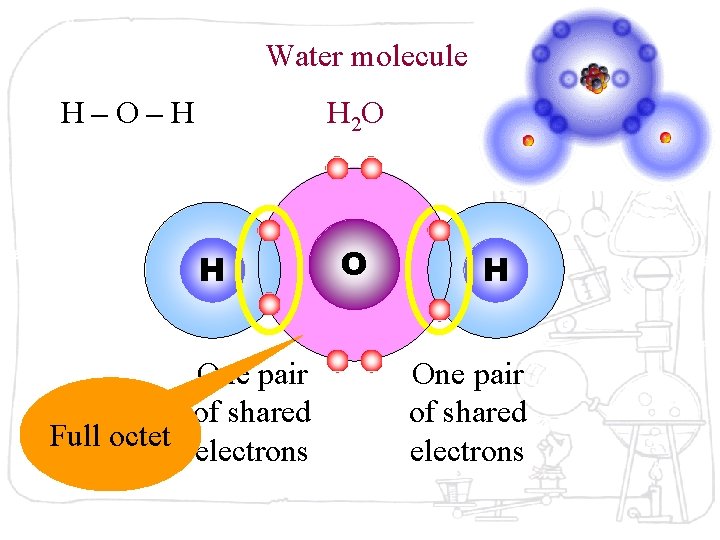
Water molecule H–O–H H 2 O H One pair of shared Full octet electrons O H One pair of shared electrons
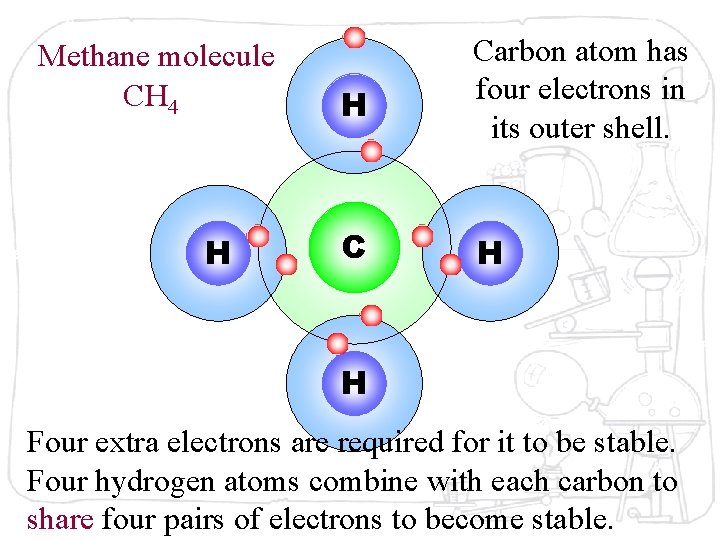
Methane molecule CH 4 H H Carbon atom has four electrons in its outer shell. C H H Four extra electrons are required for it to be stable. Four hydrogen atoms combine with each carbon to share four pairs of electrons to become stable.
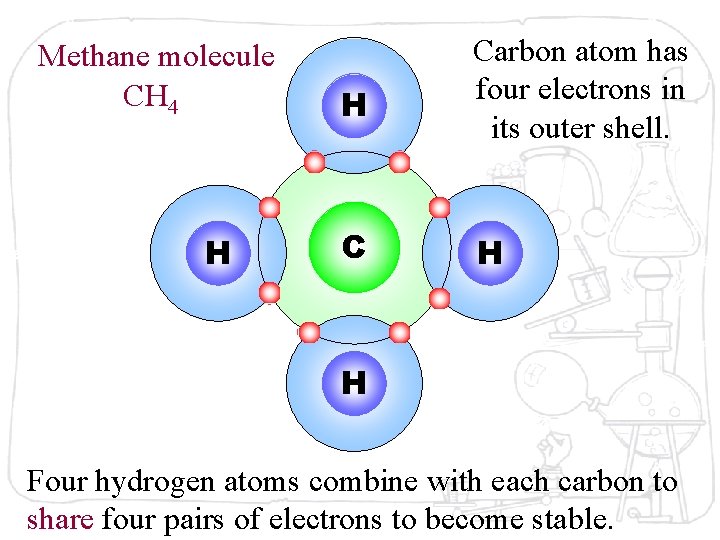
Methane molecule CH 4 H H Carbon atom has four electrons in its outer shell. C H H Four hydrogen atoms combine with each carbon to share four pairs of electrons to become stable.
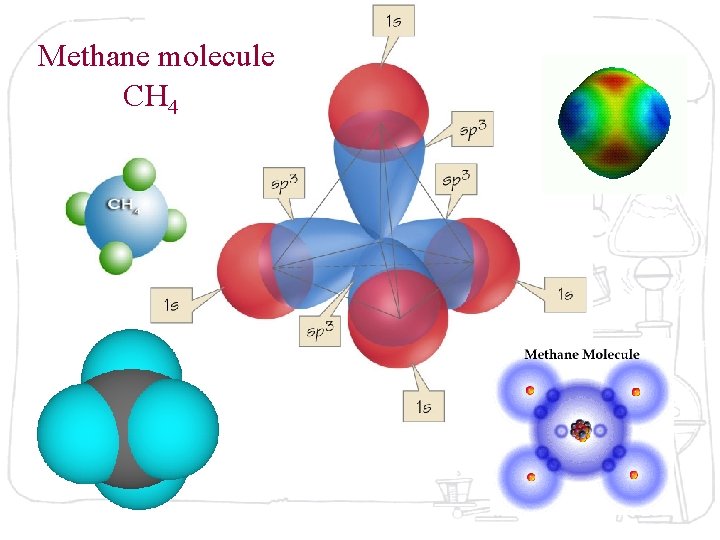
Methane molecule CH 4
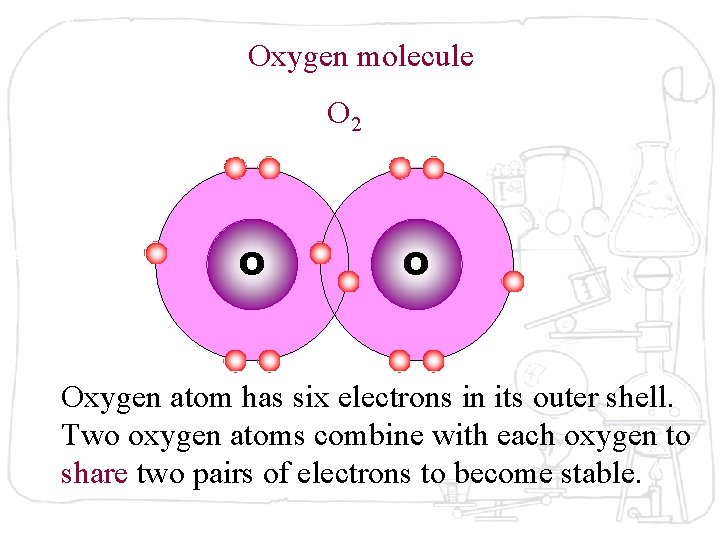
Oxygen molecule O 2 O O Oxygen atom has six electrons in its outer shell. Two oxygen atoms combine with each oxygen to share two pairs of electrons to become stable.
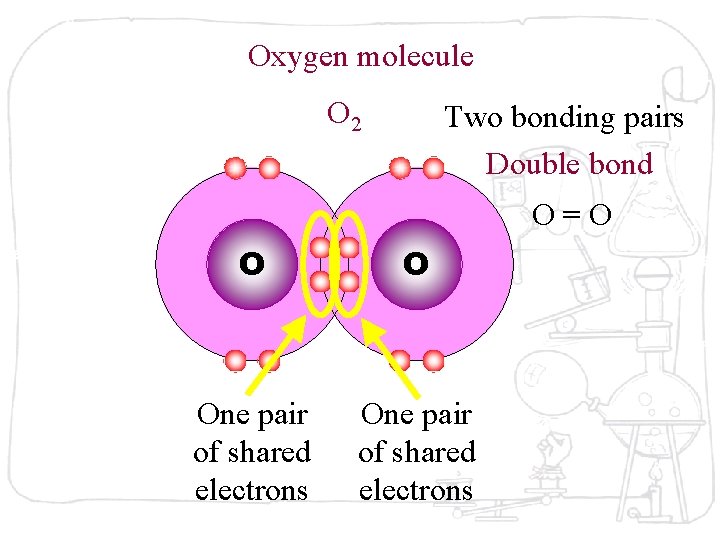
Oxygen molecule O 2 Two bonding pairs Double bond O=O O O One pair of shared electrons
- Slides: 38