Chemical Bonding Copyright 2014 Chemistry Corner Chemical Bond
Chemical Bonding Copyright © 2014 Chemistry Corner

Chemical Bond: attractive force between atoms or ions that binds them together as a unit atoms form bonds in order to… ü decrease potential energy (PE) ü increase stability Copyright © 2014 Chemistry Corner

• Covalent bonding — electrons are shared between atoms in a molecule or in polyatomic ions • Ionic bonding — positively and negatively charged ions are held together by electrostatic forces electrons are transferred Copyright © 2014 Chemistry Corner
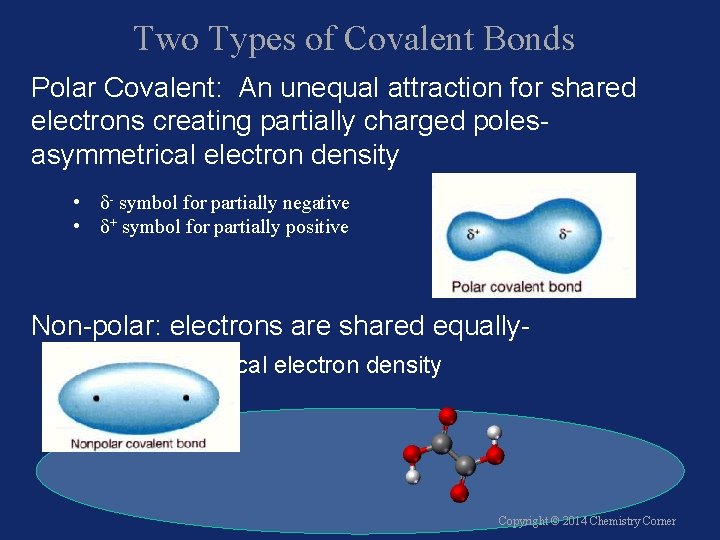
Two Types of Covalent Bonds Polar Covalent: An unequal attraction for shared electrons creating partially charged polesasymmetrical electron density • δ- symbol for partially negative • δ+ symbol for partially positive Non-polar: electrons are shared equallysymmetrical electron density Copyright © 2014 Chemistry Corner

Copyright © 2014 Chemistry Corner

Determining Type of Bond Using Electronegativity Difference Recall: Electronegativity is the measure of an atom’s ability to attract electrons! For Polar Covalent Bonds: higher electronegative atom = lower electronegative atom = + Copyright © 2014 Chemistry Corner

Determining Type of Bond Using Electronegativity Difference For Ionic Bonds: ü The higher electronegative atom forms an anion. (gains electrons) ü The lower electronegative atom forms a cation. (loses electrons) Copyright © 2014 Chemistry Corner

Determining Type of Bond Using Electronegativity Difference Copyright © 2014 Chemistry Corner
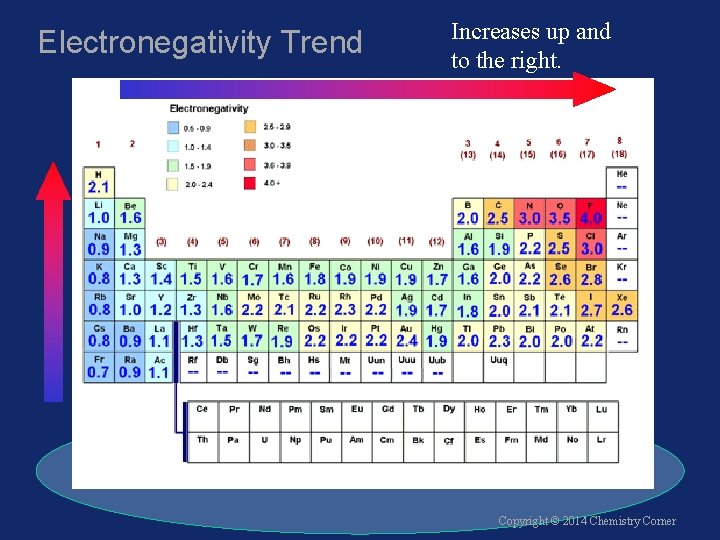
Electronegativity Trend Increases up and to the right. Copyright © 2014 Chemistry Corner
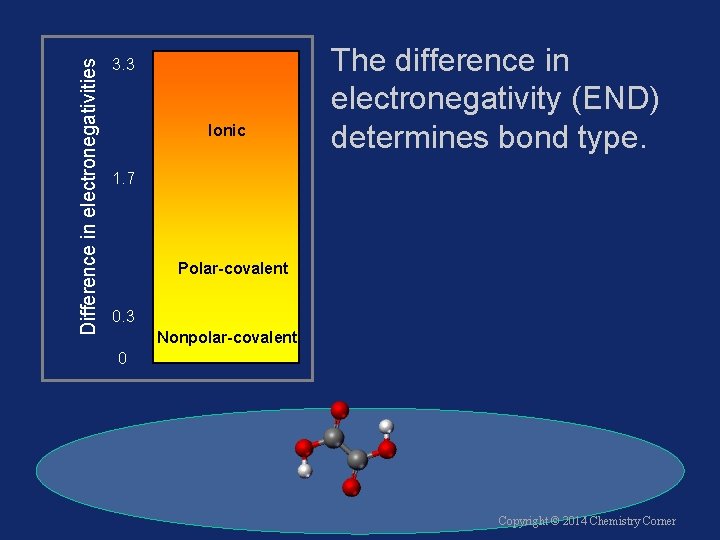
Difference in electronegativities 3. 3 Ionic The difference in electronegativity (END) determines bond type. 1. 7 Polar-covalent 0. 3 Nonpolar-covalent 0 Copyright © 2014 Chemistry Corner
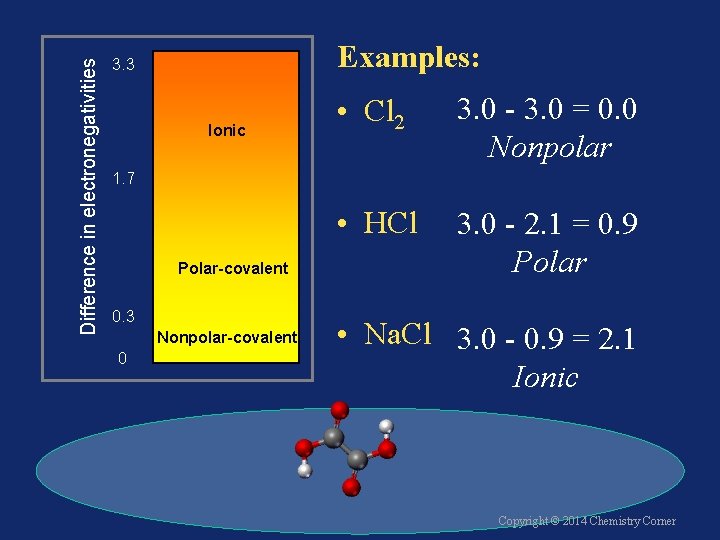
Difference in electronegativities Examples: 3. 3 Ionic • Cl 2 3. 0 - 3. 0 = 0. 0 Nonpolar • HCl 3. 0 - 2. 1 = 0. 9 Polar 1. 7 Polar-covalent 0. 3 Nonpolar-covalent 0 • Na. Cl 3. 0 - 0. 9 = 2. 1 Ionic Copyright © 2014 Chemistry Corner
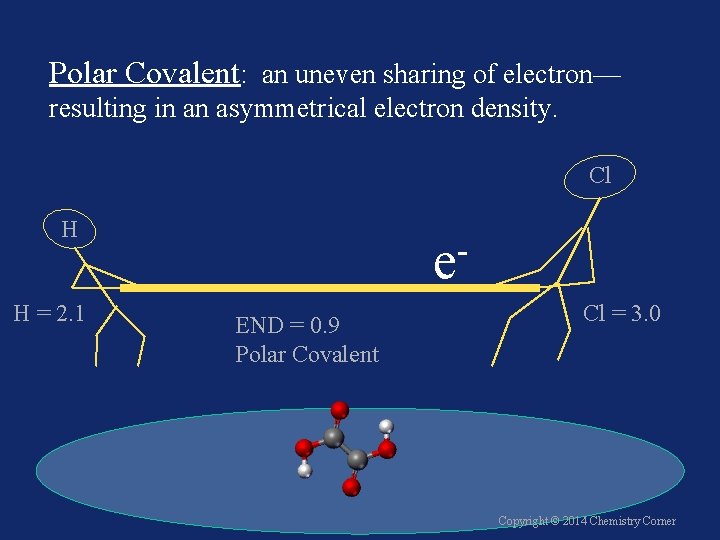
Polar Covalent: an uneven sharing of electron— resulting in an asymmetrical electron density. Cl H H = 2. 1 e END = 0. 9 Polar Covalent Cl = 3. 0 Copyright © 2014 Chemistry Corner

Non-Polar Covalent: an even sharing of electron— resulting in an symmetrical electron density. Cl Cl e Cl = 3. 0 END = 0. 0 Non-Polar Covalent Cl = 3. 0 Copyright © 2014 Chemistry Corner
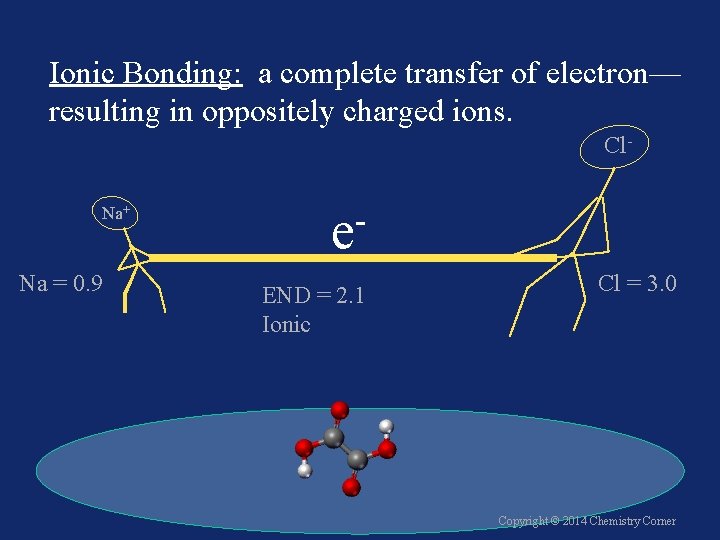
Ionic Bonding: a complete transfer of electron— resulting in oppositely charged ions. Cl. Na+ Na = 0. 9 e END = 2. 1 Ionic Cl = 3. 0 Copyright © 2014 Chemistry Corner
Practice Use electronegativity values to classify bonding between: Sulfur (S) and Hydrogen (H) Cesium (Cs) and Fluorine (F) Chlorine (Cl) and Phosphorus (P) Copyright © 2014 Chemistry Corner

Practice Solution Bonding END Bond type atom More negative S and H 2. 5 – 2. 1 = 0. 4 polar-covalent sulfur Cs and F 4. 0 – 0. 8 = 3. 2 ionic fluorine Cl and P 3. 0 – 2. 2 = 0. 8 polar-covalent chlorine O and O 3. 5 – 3. 5 = 0. 0 nonpolar-covalent NA Copyright © 2014 Chemistry Corner

How Does Bonding Work? e- transfer 1 s 2 s + Li 2 p Ionic Bond is then formed between F- & Li+ 2 s 1 s - F Copyright © 2014 Chemistry Corner
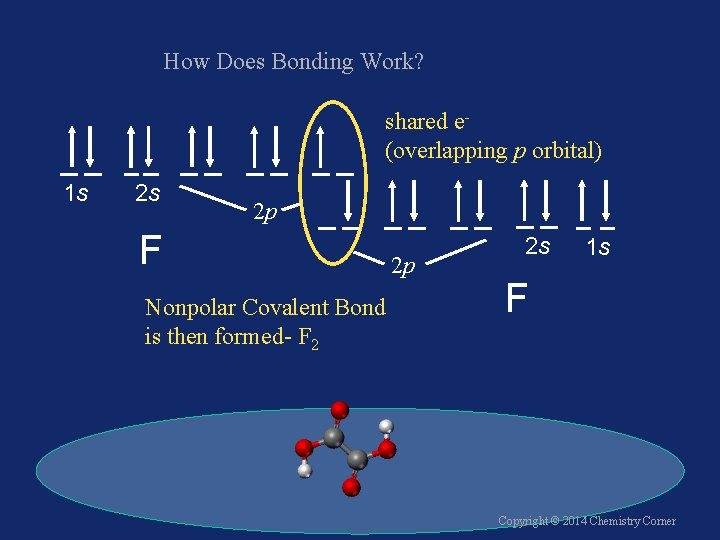
How Does Bonding Work? shared e(overlapping p orbital) 1 s 2 s 2 p F Nonpolar Covalent Bond is then formed- F 2 2 p 2 s 1 s F Copyright © 2014 Chemistry Corner
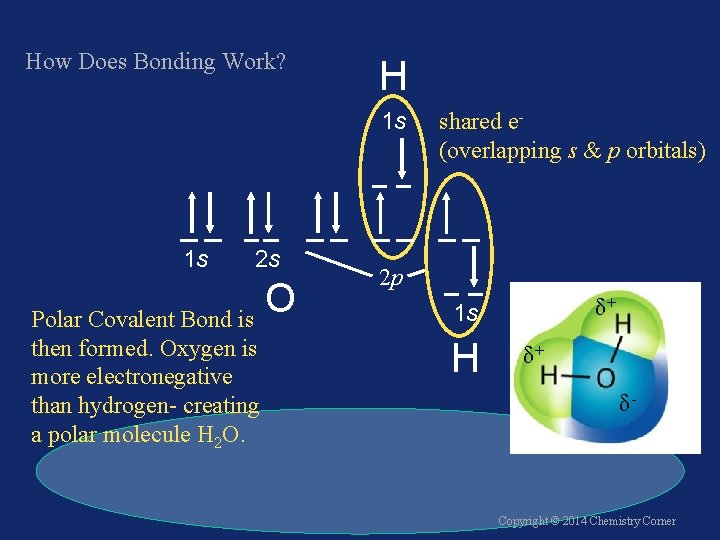
How Does Bonding Work? H 1 s 1 s 2 s O Polar Covalent Bond is then formed. Oxygen is more electronegative than hydrogen- creating a polar molecule H 2 O. shared e(overlapping s & p orbitals) 2 p δ+ 1 s H δ+ δ- Copyright © 2014 Chemistry Corner
Wrapping it Up! Nonpolar Covalent Bond üelectrons are shared equally üsymmetrical electron density üusually identical atoms Copyright © 2014 Chemistry Corner
Wrapping it Up! Polar Covalent Bond ü electrons are shared unequally ü asymmetrical electron density ü results in partial charges Copyright © 2014 Chemistry Corner
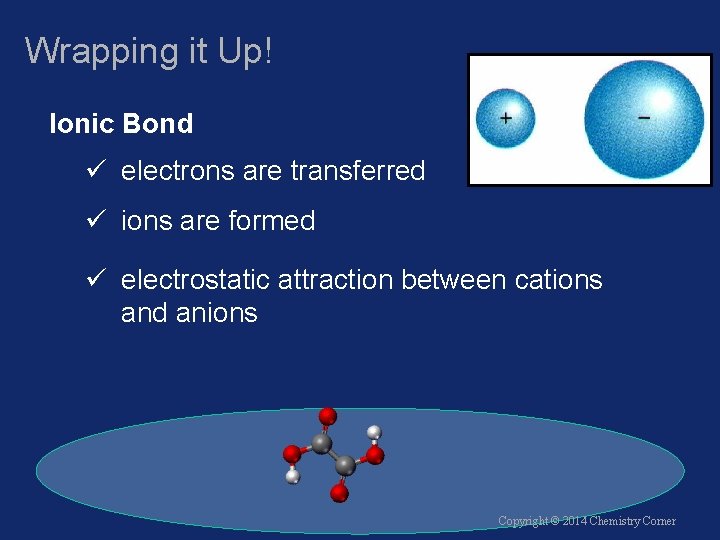
Wrapping it Up! Ionic Bond ü electrons are transferred ü ions are formed ü electrostatic attraction between cations and anions Copyright © 2014 Chemistry Corner
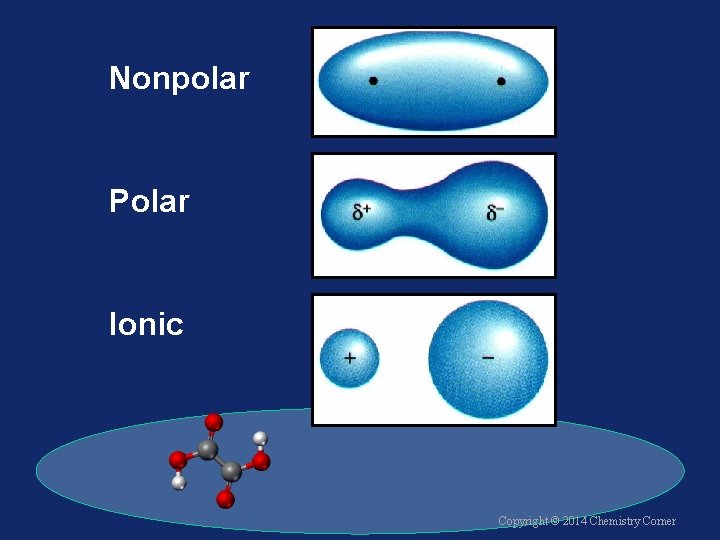
Nonpolar Polar Ionic Copyright © 2014 Chemistry Corner
More Wrapping it Up! Covalent – sharing of electrons (Non-metal + Non-metal) Did you notice this? Non-polar Polar Ionic – transfer of electrons (Metal + Non-metal) Did you notice this? Copyright © 2014 Chemistry Corner

EXIT TICKET: Why do atoms form bonds? Copyright © 2014 Chemistry Corner

Copyright © 2014 Chemistry Corner. All rights reserved. Purchase of this unit entitles the purchaser the right to reproduce the pages in limited quantities for classroom use only. Duplication for an entire school, an entire school system or commercial purposes is strictly forbidden without written permission from the publisher. Chemistry Corner, chemistrycorner. tn@gmail. com Empowering students for the road ahead Copyright © 2014 Chemistry Corner

Disclaimer This publication is provided with the understanding that the author and publisher are not engaged to render any type of psychological or legal or any other kind of professional advice. No warranties or guarantees are expressed or implied by the author's or publisher's choice to include any of the content in this publication. Neither the author nor the publisher shall be liable for any psychological, emotional, financial or commercial damages, including, but not limited to, special, incidental, consequential or other damages. Our views and rights are the same: You are responsible for your own choices, actions and results. Copyright Chemistry Corner This publication contains material protected under International and Federal Copyright Laws and Treaties. Any unauthorized reprint or use of this material is prohibited. The original purchaser is granted the right to photocopy, use or display this material for his/her educational use without limitation. No part of this publication may be reproduced or transmitted in any form or by any means (electronic or mechanical, including photocopying, recording, or by any information storage and retrieval system) in order to provide use to another person without express written permission from the author and publisher unless additional licenses or group licenses have been purchased. Copyright © 2014 Chemistry Corner
- Slides: 27