Chemical Bonding CHEMICAL BONDS A chemical bond is

Chemical Bonding

CHEMICAL BONDS • A chemical bond is the force that holds atoms together. • There are 3 types of bonds: • Ionic bond • Covalent bond • Metallic bond

Chemical bond • Chemical bonds can form : • A) By the attraction between opposite charged ions • B) By the sharing of electrons between atoms

Positive Ion Formation • A positive ion forms when an atom loses one or more valence electrons in order to attain a noble gas configuration (octet rule). • A positively charged ion is called a cation • Ex.

Metal Ions • Metals atoms are reactive because they lose valence electrons easily, have Low ionization energy and low electron affinity • The group 1 and 2 metals are the most reactive metals in the P. T. • Metals in group 1 form +1 ions • Metals in group 2 form +2 ions • Metals in group 13 form +3 ions • Transition metals: is difficult to predict

Negative Ion Formation • Nonmetals, located on the right side of the P. T. , easily gain electrons to attain a stable outer configuration(octet rule) and form an anion.

Nonmetal Ions • Elements in group 15 gain 3 electrons and form -3 ions • Elements in group 16 gain 2 electrons and form -2 ions • Elements in group 17 gain 1 electron and form -1 ions

Lewis dot diagrams for Ions

Practice • Write the symbol of the ion and the dot structure of the following : • a)calcium ion • b) germanium ion • c) phosphide • d) oxide

The Ionic Bond
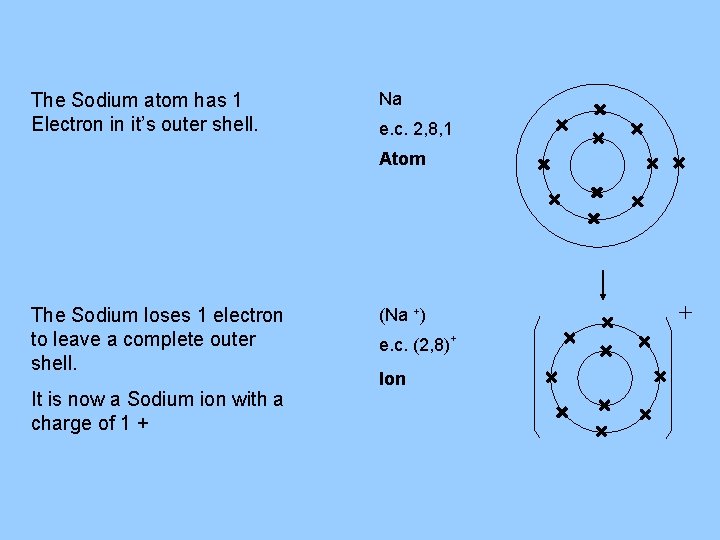
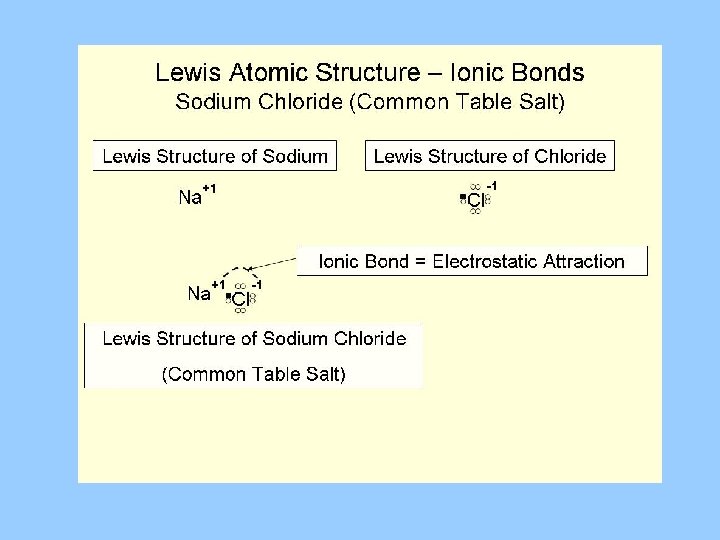
The Sodium atom has 1 Electron in it’s outer shell. Na e. c. 2, 8, 1 Atom The Sodium loses 1 electron to leave a complete outer shell. It is now a Sodium ion with a charge of 1 + (Na +) e. c. (2, 8)+ Ion +
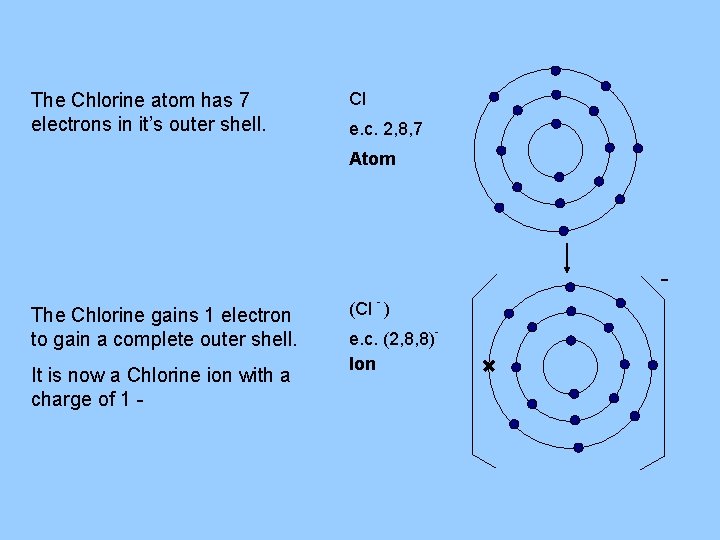
The Chlorine atom has 7 electrons in it’s outer shell. Cl e. c. 2, 8, 7 Atom The Chlorine gains 1 electron to gain a complete outer shell. It is now a Chlorine ion with a charge of 1 - (Cl - ) e. c. (2, 8, 8)Ion
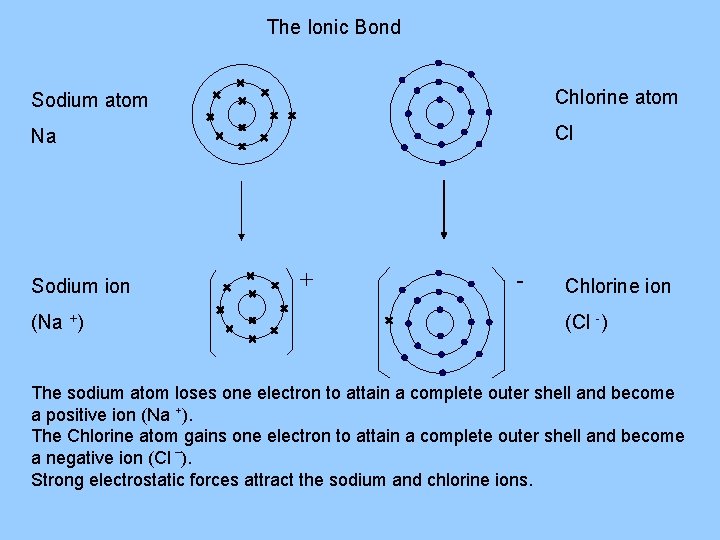
The Ionic Bond Sodium atom Chlorine atom Na Cl Sodium ion (Na +) + - Chlorine ion (Cl -) The sodium atom loses one electron to attain a complete outer shell and become a positive ion (Na +). The Chlorine atom gains one electron to attain a complete outer shell and become a negative ion (Cl –). Strong electrostatic forces attract the sodium and chlorine ions.
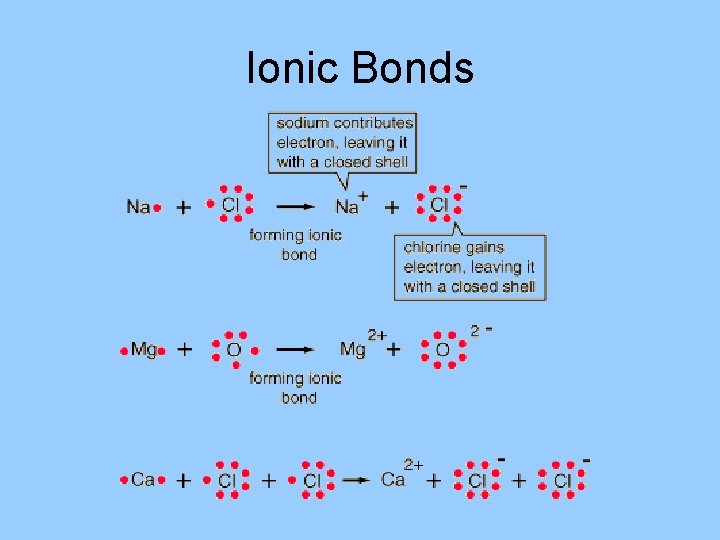
Ionic Bonds

Ion Cartoon

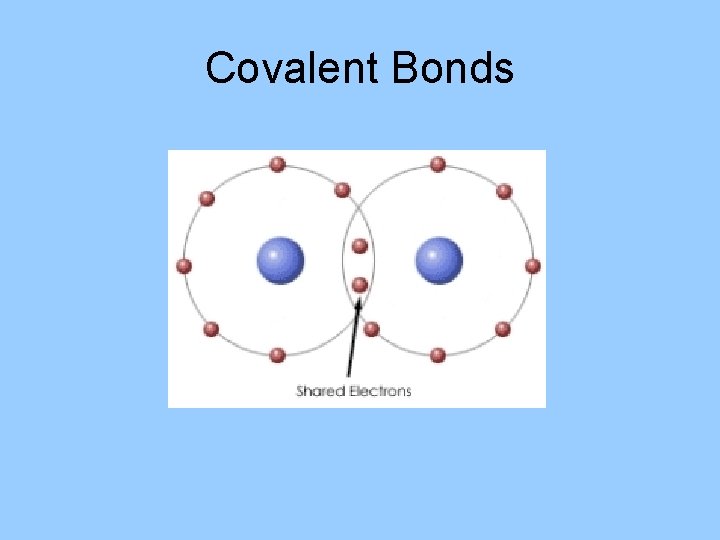
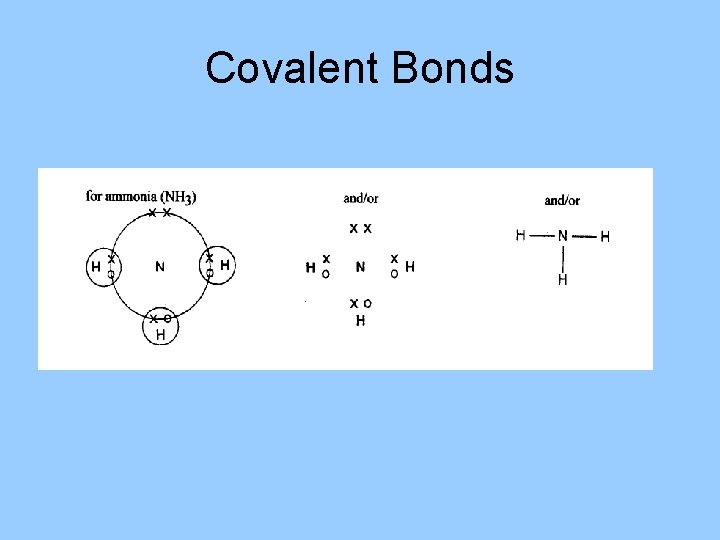
Covalent Bonds • Atoms in nonionic compounds share electrons. • A molecule is formed when 2 or more atoms bond covalently. • The majority of covalent bonds form between atoms of nonmetallic elements.
Covalent Bonds
Covalent Bonds

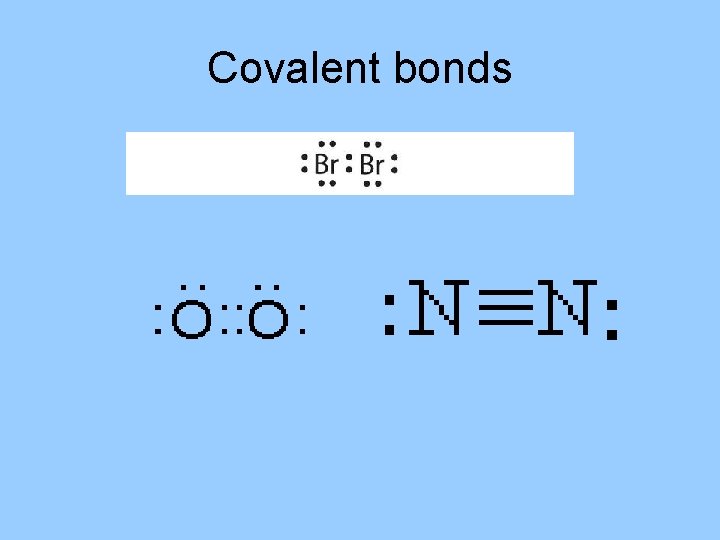
Covalent bonds can be single, double or triple • Single covalent bond: atoms share 1 pair of electrons • Double covalent bond: atoms share 2 pairs of electrons • Triple covalent bond: atoms share 3 pairs of electrons
Covalent bonds

Covalent bonds • The halogens, group 17, form single covalent bonds with atoms of other nonmetals. They can only form 1 bond since they have 7 valence electrons. • The chalcogens, group 16, have 6 valence electrons, can form a total of 2 bonds. These can be single or double. • The group 15 elements, have 5 valence electrons, can form a total of 3 bonds. These can be single, double or triple.

Practice • Draw the Lewis structures for each molecule: • a) PH 3 • b) H 2 S • c) HCl • d) CCl 4 • e) Si. H 4 • f) OBr 2

Electronegativity and Polarity • The type of bond formed is related to each atom´s attraction for electrons. • Electronegativity indicates the relative ability of an element´s atoms to attract electrons in a chemical bond. • Nonmetals have higher electronegativities than do metals. • Each element is assigned a value:

Electronegativity
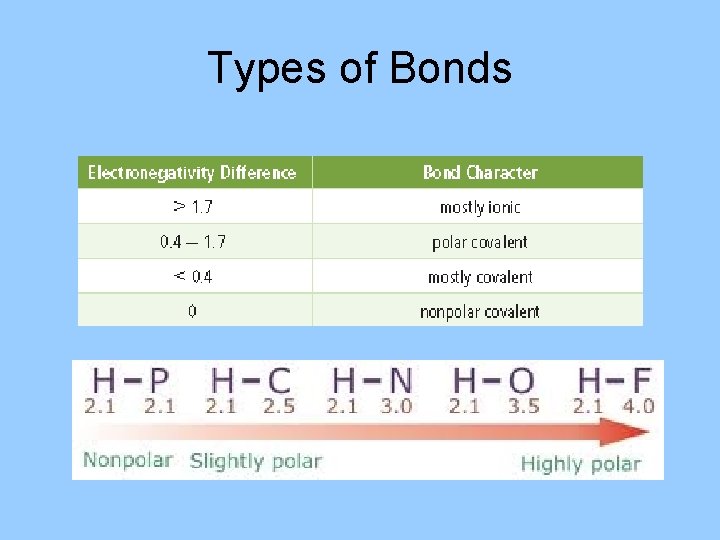
Types of Bonds

Types of bonds • Bond can be: polar covalent, nonpolar covalent and ionic. • Polar bond: a bond in which electrons are shared unequally. • Nonpolar bond: a bond in which electrons are shared equally. • Ionic bond: a bond in which electrons are transferred. • The character of a bond depends on how strongly each of the bonded atoms attracts electrons.

Practice • Use electronegativities to classify each of the following bonds as nonpolar covalent, polar covalent or ionic: • a) O-H • b) O-K • c) Cl- As • d) N -N

POLARITY OF MOLECULES • Nonpolar molecules: are molecules that have a symmetrical arrangement and the dipoles cancel each other. • Polar molecules: one end of the molecule is more negatively charged than the other end, have a dipole.
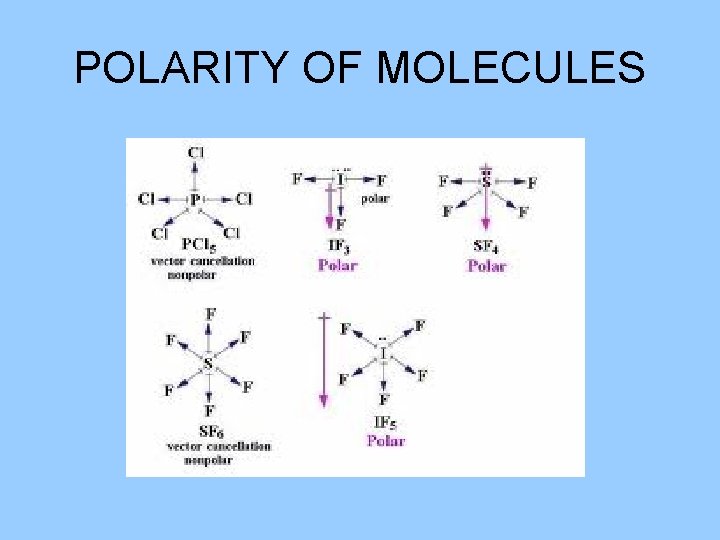
POLARITY OF MOLECULES

PRACTICE • Indicate if each of the following molecules is polar or nonpolar: • a) BF 3 • b) CH 3 F • c) CCl 4 • d) NF 3 • e) Br 2
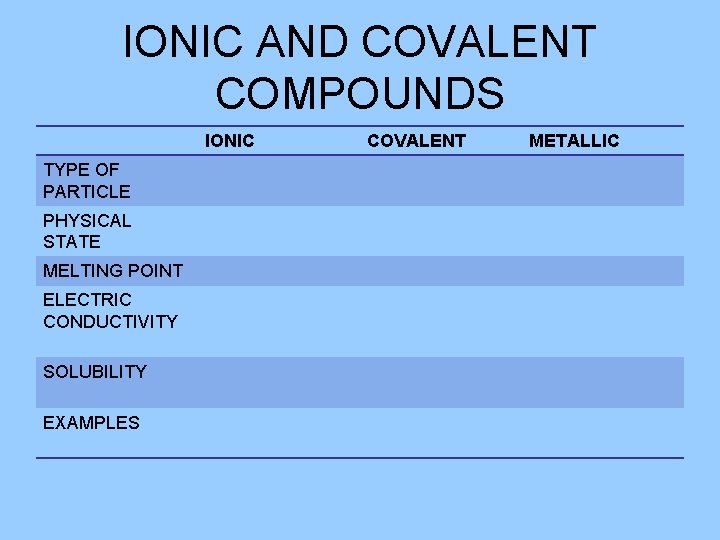
IONIC AND COVALENT COMPOUNDS IONIC TYPE OF PARTICLE PHYSICAL STATE MELTING POINT ELECTRIC CONDUCTIVITY SOLUBILITY EXAMPLES COVALENT METALLIC
- Slides: 32