Chemical BONDING Chemical Bond A chemical bond results

Chemical BONDING

Chemical Bond • A chemical bond results as atoms try to –achieve a stable octet (or duet) in their outer shell just like the noble gases

Two Major Types of Bonding • Ionic Bonding SALTS Crystals – forms ionic compounds – transfer of e- between valence shells • Covalent Bonding – forms molecules – sharing e- between valence shells

One other type of bonding • Metallic bonding – Occurs between atoms of a metal or MIXTURE OF METALS (alloys) • Ex. Al: aluminum foil, Cu: copper wire, Steel – Valence e- are mobile (move freely among all metal atoms) • Metallic characteristics – High m. p. temps, ductile, malleable, shiny – Good conductors of heat and electricity
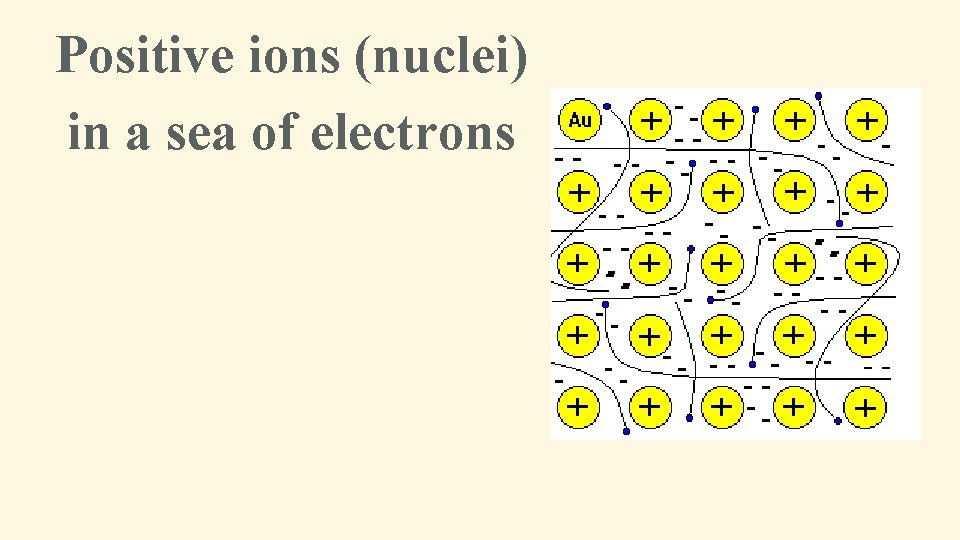
Positive ions (nuclei) in a sea of electrons
![• Ionic Bonds form between [METAL ] Lost e- + [NON-METAL Gains e- • Ionic Bonds form between [METAL ] Lost e- + [NON-METAL Gains e-](http://slidetodoc.com/presentation_image_h2/7b034b43794518846df50d6f8a448601/image-6.jpg)
• Ionic Bonds form between [METAL ] Lost e- + [NON-METAL Gains e- ]

also. . . • The bond to a POLYATOMIC ION Ø ex. KNO 3 Ø ex. NH 4 Cl

PROPERTIES OF IONIC COMPOUNDS

Form crystal lattice structures because of alternating anions(-) and cations (+) Hard, angular crystals at room temp ~20 o. C

• high melting point temperatures ex. Na. Cl 801 o. C (1, 474 o. F) Remember H 2 O melts at 0 o. C! Salts & Solution Ph. ET

Covalent Bonding • Pairs of e- are shared between non -metal atoms H 2 O NO 3 Na. Cl

• In covalent bonding – Electrons can be shared equally which makes the bond NON-POLAR

Or – Electrons can be shared Unequally which makes the bond POLAR

How electronegativity determines the types of bonds that form.
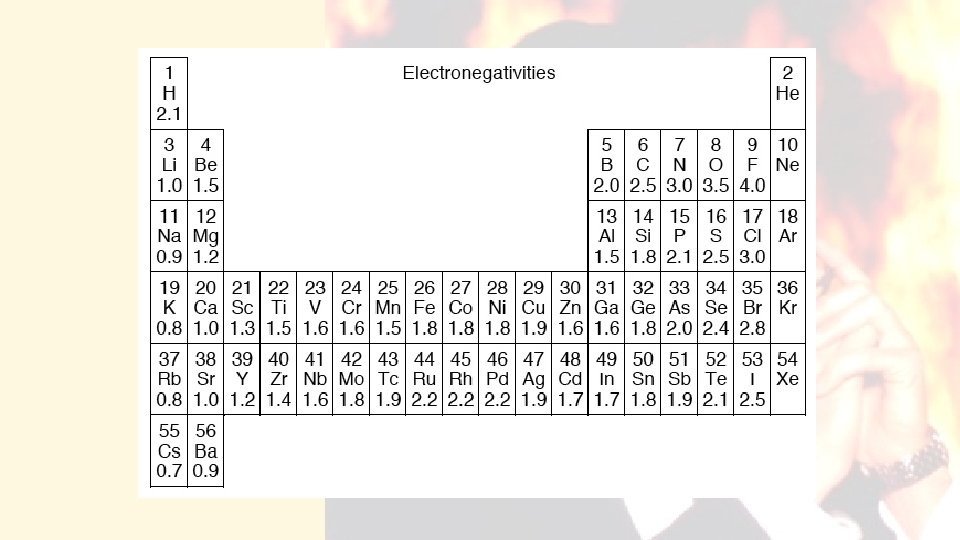

Electronegativity differences between atoms form a continuum of the types of bonds from nonpolar covalent to ionic. Dogs teaching chemistry

E-neg difference of up to & including 0. 4 = non-polar covalent (CH 4) between 0. 5 up to around 2. 0 polar covalent (H 2 O) Non-metals bonded together METAL - Non-metal

Properties of Molecular Substances Covalent bonding • Low m. p. temp and b. p. temps • relatively soft solids as compared to ionic compounds • Poor conductors & NONconductors of electricity in any phase

• Which are ionic and which are colavent? • Which covalents have non-polar bonds and which covalents have polar bonds? Na. H N 2 KI HCl HF Cs. F

The End
- Slides: 20