Chemical BONDING Chemical Bond A bond results from

Chemical BONDING

Chemical Bond • A bond results from the attraction of nuclei for electrons – All atoms trying to achieve a stable octet • IN OTHER WORDS – the p+ in one nucleus are attracted to the e- of another atom • Electronegativity

Two Major Types of Bonding • Ionic Bonding – transfer of e- • Covalent Bonding – sharing e-

One minor type of bonding • Metallic bonding – Occurs between like atoms of a metal in the free state – Valence e- are mobile (move freely among all metal atoms) – Positive ions in a sea of electrons • Metallic characteristics – High mp temps, ductile, malleable, shiny – Hard substances – Good conductors of heat and electricity

It’s the mobile electrons that enable me tals to conduct electricity!!!!!!

IONic Bonding • electrons are transferred between valence shells of atoms • ionic compounds are called Salts or Crystals
![IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ] IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ]](http://slidetodoc.com/presentation_image_h2/7bdbf7fc961d155e9177e8809a54ebff/image-7.jpg)
IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ] Lost e- Gained e-
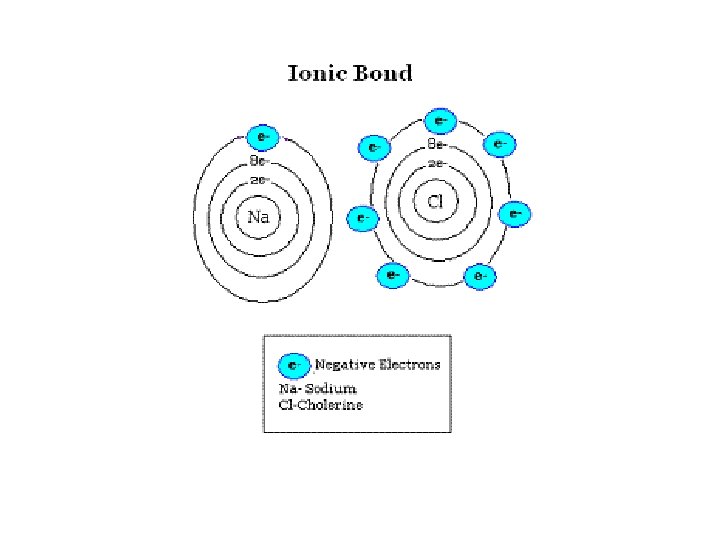

Properties of Ionic Compounds SALTS Crystals • hard solid @ 22 o. C • high mp temperatures • good conductors in liquid phase or dissolved in water (aq)

Covalent Bonding • Pairs of e- are shared between non-metal atoms

Properties of Molecular Substances Covalent bonding • Low m. p. temp and b. p. temps • relatively soft solids as compared to ionic compounds • nonconductors of electricity in any phase

Covalent, Ionic, metallic bonding? • NO 2 • • sodium • hydride • Hg • • H 2 S • • sulfate • NH 4+ Aluminum phosphate KH KCl HF • CO Also study your characteristics!
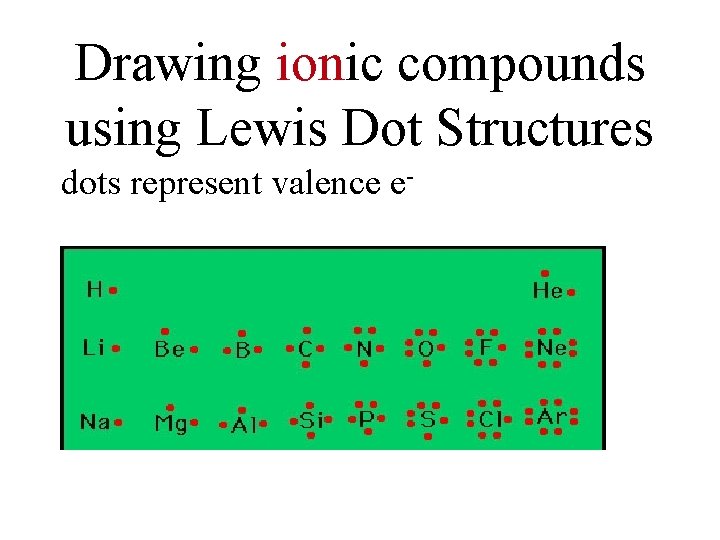
Drawing ionic compounds using Lewis Dot Structures dots represent valence e-

Na. Cl • This is the finished Lewis Dot Structure How did we get here? + [Na] [ Cl ] -

• Step 1 after checking that it is IONIC – Determine which atom will be the +ion – Determine which atom will be the - ion • Step 2 – Write the symbol for the + ion first. • NO DOTS – Draw the e- dot diagram for the – ion • COMPLETE outer shell • Step 3 – Enclose both in brackets and show each charge

Draw the Lewis Diagrams • Li. F • Mg. O • Ca. Cl 2 • K 2 S
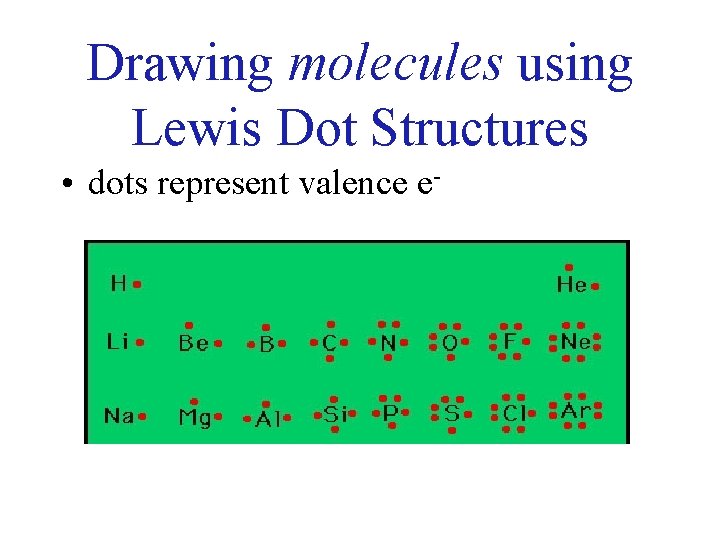
Drawing molecules using Lewis Dot Structures • dots represent valence e-

Always remember atoms are trying to complete their outer shell! The number of electrons the atoms needs is the total number of bonds they can make. Ex. … H? O? F? N? Cl? C? one two one three one four
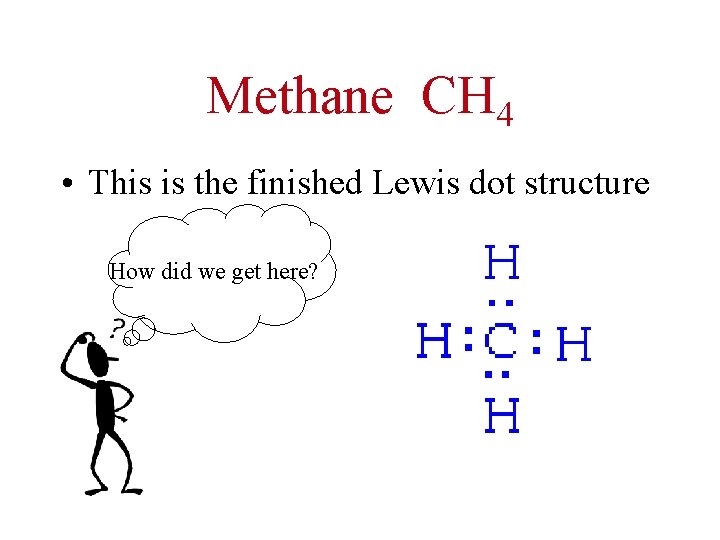
Methane CH 4 • This is the finished Lewis dot structure How did we get here?

• Step 1 – count total valence e- involved • Step 2 – connect the central atom (usually the first in the formula) to the others with single bonds • Step 3 – complete valence shells of outer atoms IF the central atom has 8 valence e- surrounding it. . YOU’RE DONE!

Sometimes. . . • You only have two atoms, so there is no central atom, but follow the same rules. • Check & Share to make sure all the atoms are “happy”. Cl 2 Br 2 H 2 O 2 N 2 HCl
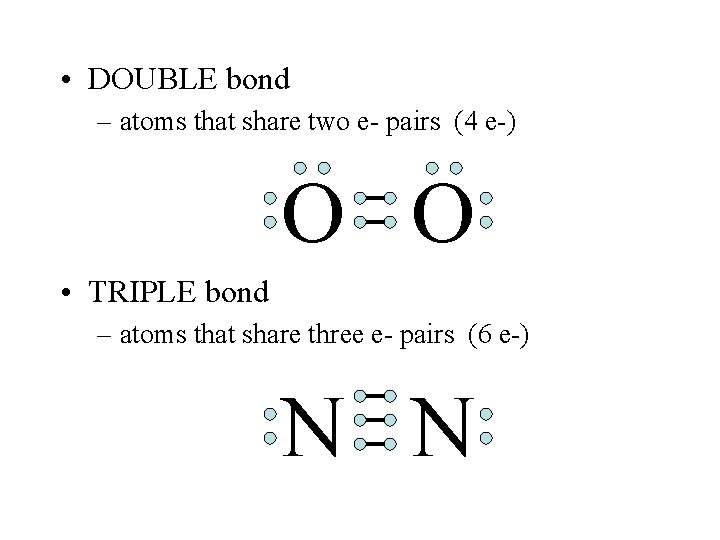
• DOUBLE bond – atoms that share two e- pairs (4 e-) O O • TRIPLE bond – atoms that share three e- pairs (6 e-) N N

Draw Lewis Dot Structures You may represent valence electrons from different atoms with the following symbols x, , CO 2 NH 3

Types of Covalent Bonds • NON-Polar bonds – Electrons shared evenly in the bond Between identical atoms Diatomic molecules

Types of Covalent Bonds Polar bond – Electrons unevenly shared

non-polar MOLECULES • Sometimes the bonds within a molecule are polar and yet the molecule is non-polar because its shape is symmetrical. H Draw Lewis dot first and see if equal on all sides H C H H

Polar molecules (a. k. a. Dipoles) • Not equal on all sides – Polar bond between 2 atoms makes a polar molecule – asymmetrical shape of molecule
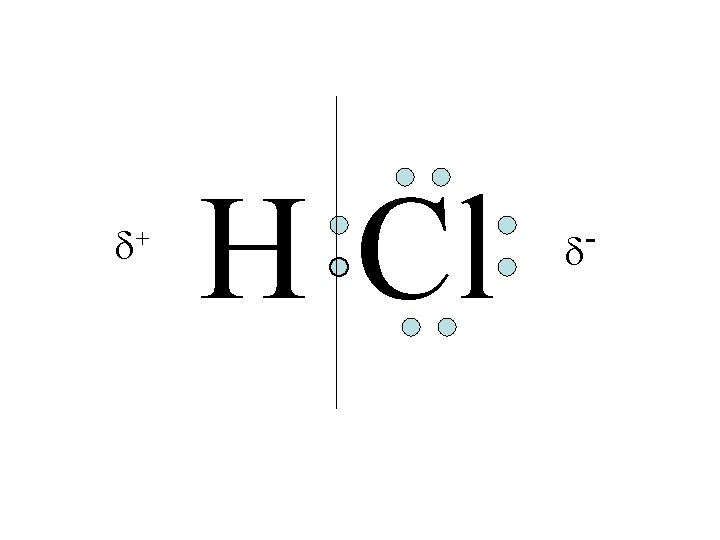
+ H Cl

Water is a bent molecule H H O H H
- Slides: 29