Chemical BONDING Chemical Bond A bond results from

Chemical BONDING

Chemical Bond • A bond results from the attraction of nuclei for electrons – All atoms trying to achieve a stable octet • IN OTHER WORDS – the p+ in one nucleus are attracted to the e- of another atom • Electronegativity

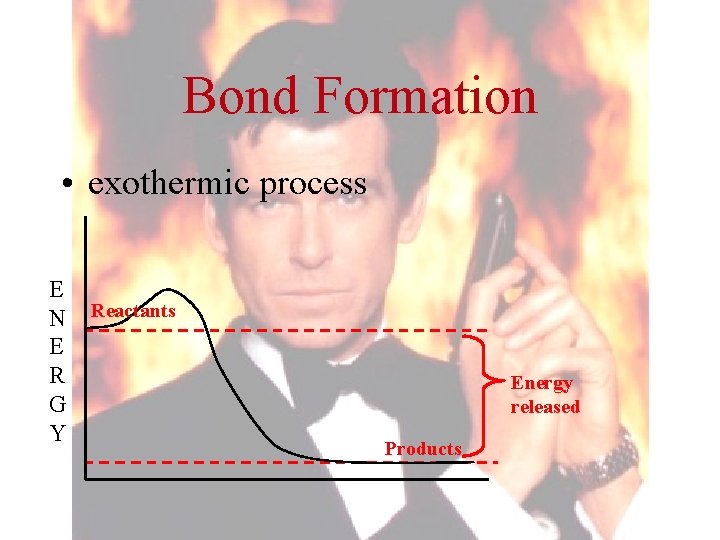
Bond Formation • exothermic process E N E R G Y Reactants Energy released Products
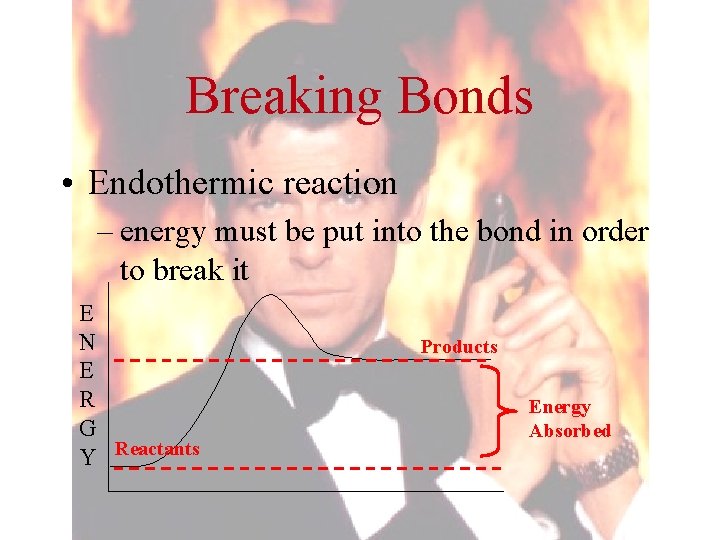
Breaking Bonds • Endothermic reaction – energy must be put into the bond in order to break it E N E R G Y Reactants Products Energy Absorbed

Bond Strength • Strong, STABLE bonds require lots of energy to be formed or broken • weak bonds require little E

Two Major Types of Bonding • Ionic Bonding – forms ionic compounds – transfer of e- • Covalent Bonding – forms molecules – sharing e-

One minor type of bonding • Metallic bonding – Occurs between like atoms of a metal in the free state – Valence e- are mobile (move freely among all metal atoms) – Positive ions in a sea of electrons • Metallic characteristics – High mp temps, ductile, malleable, shiny – Hard substances – Good conductors of heat and electricity as (s) and (l)

It’s the mobile electrons that enable me tals to conduct electricity!!!!!!

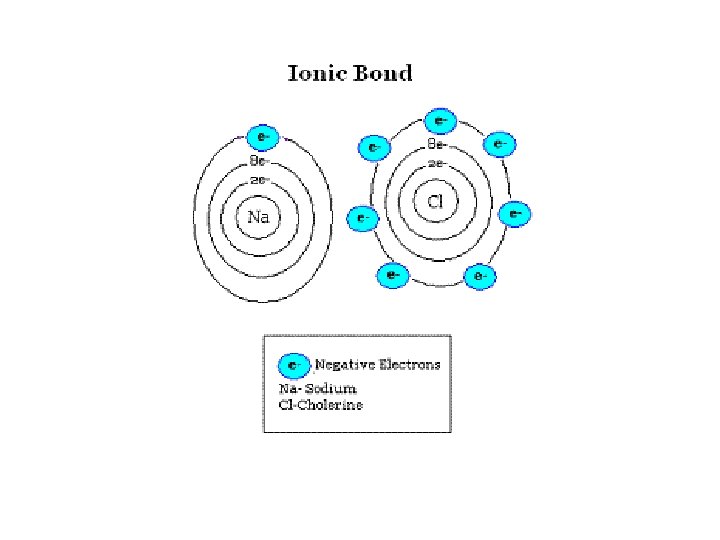
IONic Bonding • electrons are transferred between valence shells of atoms • ionic compounds are NOT MOLECULES made of ions • ionic compounds are called Salts or Crystals
![IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ] IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ]](http://slidetodoc.com/presentation_image_h2/076d164787f7ce56347cbfa725b4a925/image-11.jpg)
IONic bonding • Always formed between metals and non-metals + [METALS ] [NON-METALS ] Lost e- Gained e-

IONic Bonding • Electronegativity difference > 2. 0 – Look up e-neg of the atoms in the bond and subtract Na. Cl Ca. Cl 2 • Compounds with polyatomic ions Na. NO 3

Properties of Ionic Compounds SALTS Crystals • hard solid @ 22 o. C • high mp temperatures • nonconductors of electricity in solid phase • good conductors in liquid phase or dissolved in water (aq)

Covalent Bonding molecules • Pairs of e- are shared between non-metal atoms • electronegativity difference < 2. 0 • forms polyatomic ions

Properties of Molecular Substances Covalent bonding • Low m. p. temp and b. p. temps • relatively soft solids as compared to ionic compounds • nonconductors of electricity in any phase

Covalent, Ionic, metallic bonding? • NO 2 • • sodium • hydride • Hg • • H 2 S • • sulfate • NH 4+ Aluminum phosphate KH KCl HF • CO • Co Also study your characteristics!
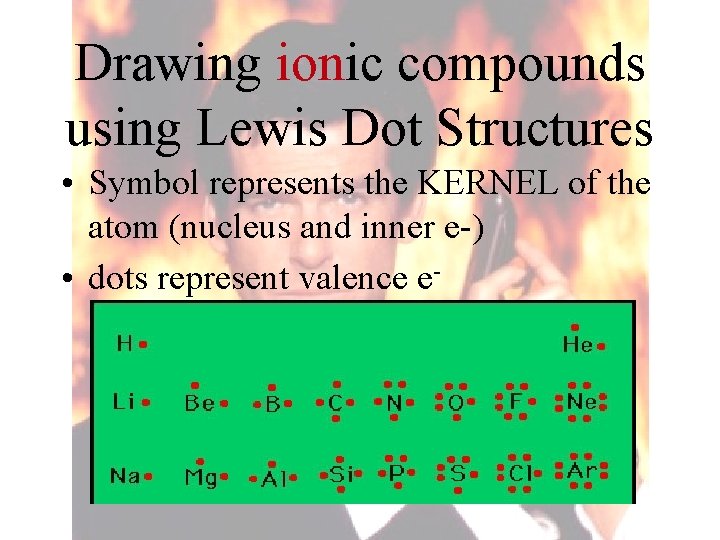
Drawing ionic compounds using Lewis Dot Structures • Symbol represents the KERNEL of the atom (nucleus and inner e-) • dots represent valence e-

Na. Cl • This is the finished Lewis Dot Structure How did we get here? + [Na] [ Cl ] -

• Step 1 after checking that it is IONIC – Determine which atom will be the +ion – Determine which atom will be the - ion • Step 2 – Write the symbol for the + ion first. • NO DOTS – Draw the e- dot diagram for the – ion • COMPLETE outer shell • Step 3 – Enclose both in brackets and show each charge

Draw the Lewis Diagrams • Li. F • Mg. O • Ca. Cl 2 • K 2 S
Drawing molecules using Lewis Dot Structures • Symbol represents the KERNEL of the atom (nucleus and inner e-) • dots represent valence e-

Always remember atoms are trying to complete their outer shell! The number of electrons the atoms needs is the total number of bonds they can make. Ex. … H? O? F? N? Cl? C? one two one three one four

Methane CH 4 • This is the finished Lewis dot structure How did we get here?

• Step 1 – count total valence e- involved • Step 2 – connect the central atom (usually the first in the formula) to the others with single bonds • Step 3 – complete valence shells of outer atoms • Step 4 – add any extra e- to central atom IF the central atom has 8 valence e- surrounding it. . YOU’RE DONE!

Sometimes. . . • You only have two atoms, so there is no central atom, but follow the same rules. • Check & Share to make sure all the atoms are “happy”. Cl 2 Br 2 H 2 O 2 N 2 HCl
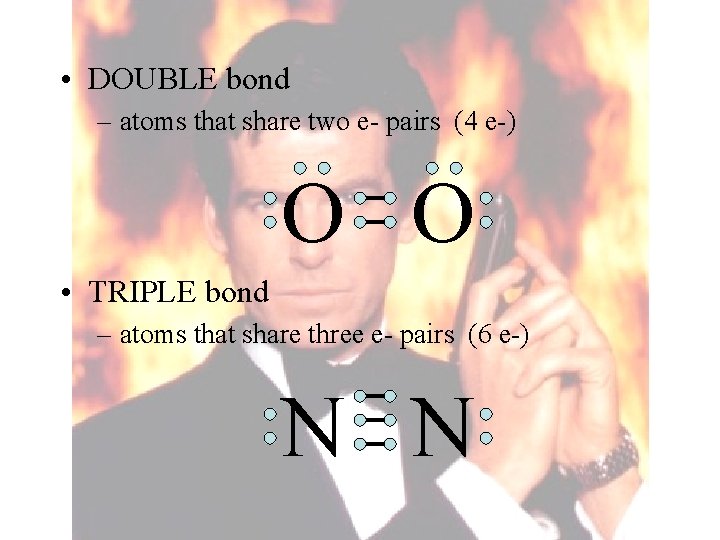
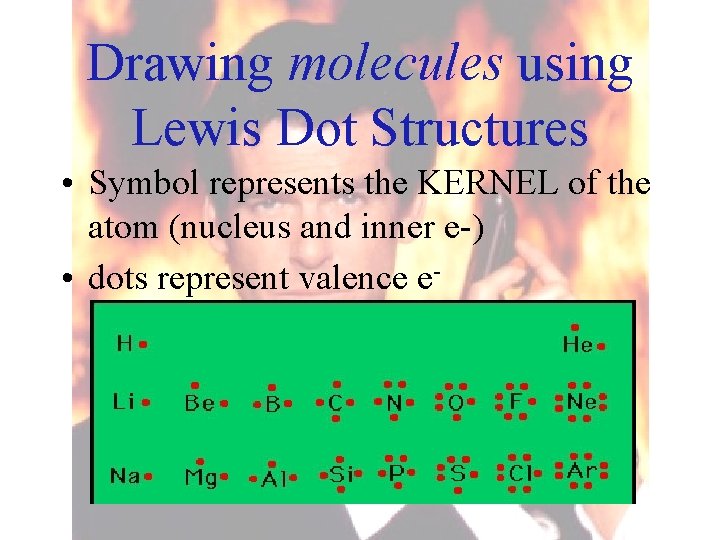
• DOUBLE bond – atoms that share two e- pairs (4 e-) O O • TRIPLE bond – atoms that share three e- pairs (6 e-) N N

Draw Lewis Dot Structures You may represent valence electrons from different atoms with the following symbols x, , CO 2 NH 3

Draw the Lewis Dot Diagram for polyatomic ions • Count all valence e- needed for covalent bonding • Add or subtract other electrons based on the charge REMEMBER! A positive charge means it LOST electrons!!!!!

Draw Polyatomics • Ammonium • Sulfate

Types of Covalent Bonds • NON-Polar bonds – Electrons shared evenly in the bond – E-neg difference is zero Between identical atoms Diatomic molecules

Types of Covalent Bonds Polar bond – Electrons unevenly shared – E-neg difference greater than zero but less than 2. 0 closer to 2. 0 more polar more “ionic character”

Place these molecules in order of increasing bond polarity which is least and which is most? • • • HCl CH 4 CO 2 NH 3 N 2 HF a. k. a. “ionic character”

non-polar MOLECULES • Sometimes the bonds within a molecule are polar and yet the molecule is non-polar because its shape is symmetrical. H Draw Lewis dot first and see if equal on all sides H C H H

Polar molecules (a. k. a. Dipoles) • Not equal on all sides – Polar bond between 2 atoms makes a polar molecule – asymmetrical shape of molecule
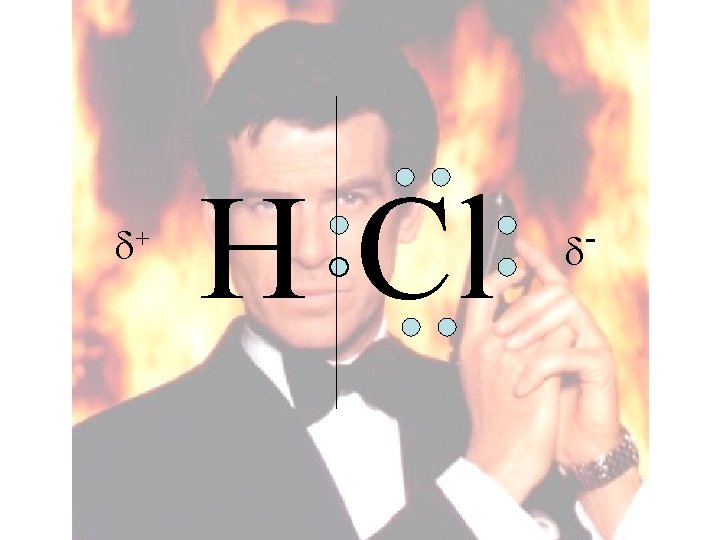
+ H Cl

Space filling model “Electron-Cloud” model + H Cl -

Water is asymmetrical + O - H + H

Water is a bent molecule H H O H H

W-A-T-E-R as bent as it can be! Water’s polar MOLECULE! The H is positive The O is not - not

Making sense of the polar non-polar thing BONDS Non-polar Polar Identical Different MOLECULES Non-polar Symmetrical Polar Asymmetrical

IONIC bonds …. Ionic bonds are so polar that the electrons are not shared but transferred between atoms forming ions!!!!!!

4 Shapes of molecules
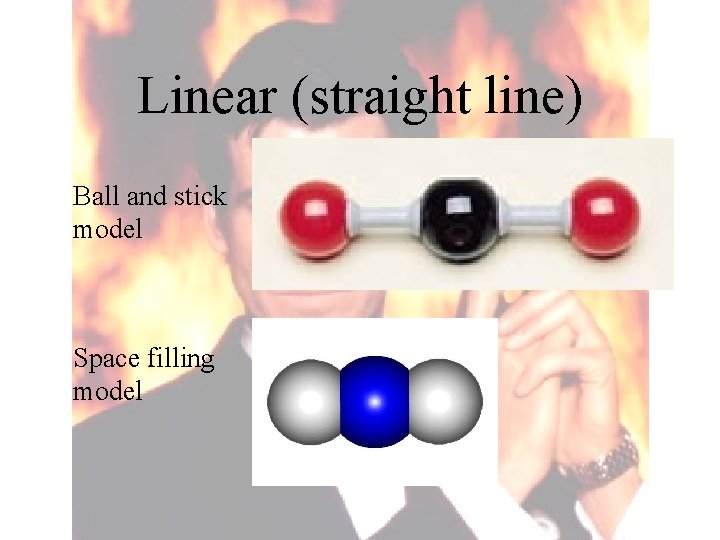
Linear (straight line) Ball and stick model Space filling model

Bent Ball and stick model Space filling model

Trigonal pyramid Ball and stick model Space filling model
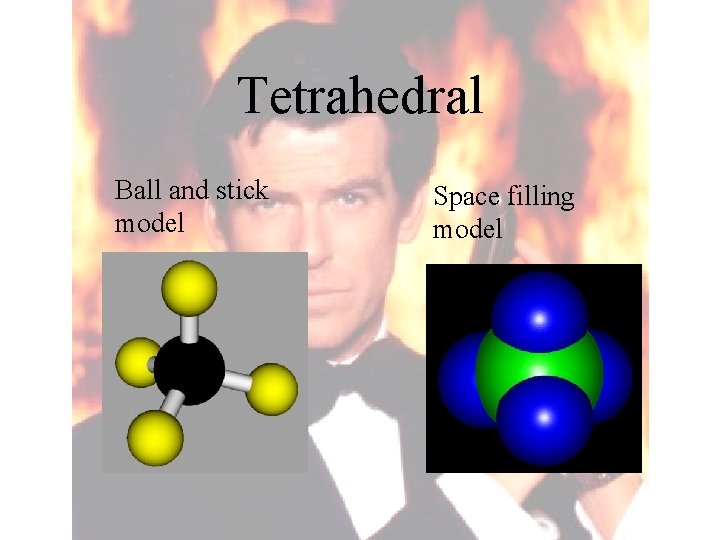
Tetrahedral Ball and stick model Space filling model
Intermolecular attractions • Attractions between molecules – van der Waals forces • Weak attractive forces between non -polar molecules – Hydrogen “bonding” • Strong attraction between special polar molecules

van der Waals • Non-polar molecules can exist in liquid and solid phases because van der Waals forces keep the molecules attracted to each other • Exist between CO 2, CH 4, CCl 4, CF 4, diatomics and monoatomics
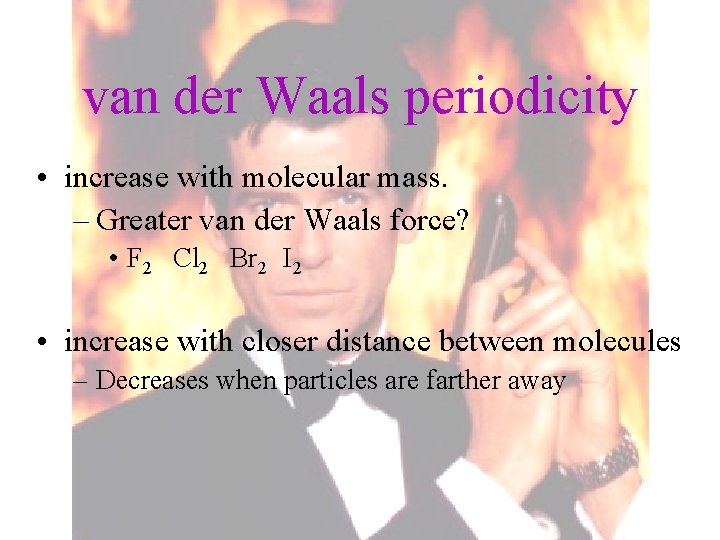
van der Waals periodicity • increase with molecular mass. – Greater van der Waals force? • F 2 Cl 2 Br 2 I 2 • increase with closer distance between molecules – Decreases when particles are farther away
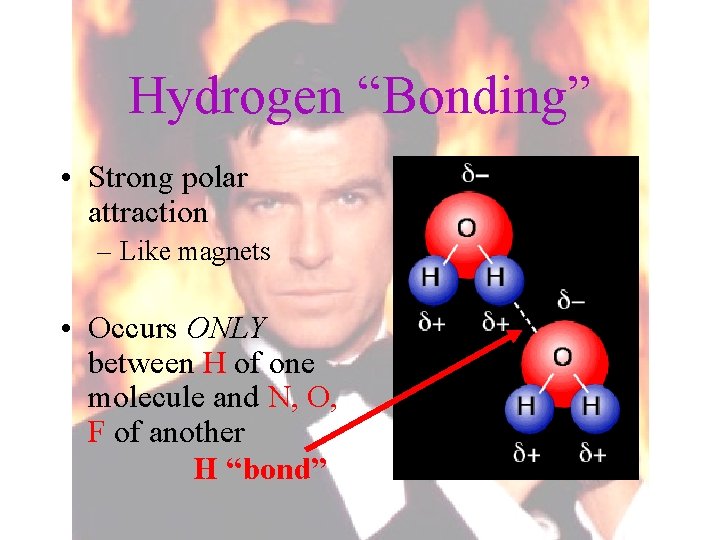
Hydrogen “Bonding” • Strong polar attraction – Like magnets • Occurs ONLY between H of one molecule and N, O, F of another H “bond”

Why does H “bonding” occur? • Nitrogen, Oxygen and Fluorine – small atoms with strong nuclear charges • powerful atoms – very high electronegativities

Intermolecular forces dictate chemical properties • Strong intermolecular forces cause high b. p. , m. p. and slow evaporation (low vapor pressure) of a substance.

Which substance has the highest boiling point? • HF • NH 3 • H 2 O Fluorine has the highest e-neg, SO HF will experience the • WHY? needs the most energy to weaken the i. m. f. and boil strongest H bonding and
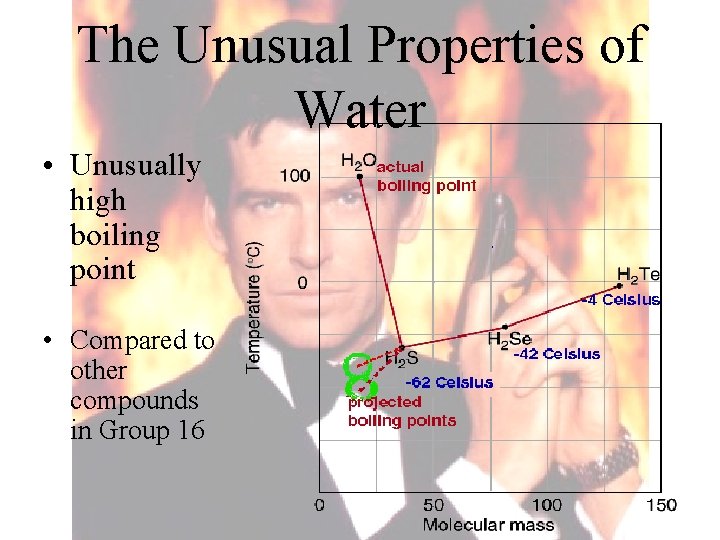
The Unusual Properties of Water • Unusually high boiling point • Compared to other compounds in Group 16

Density? ?

H 2 O(s) is less dense than H 2 O(l) • The hydrogen bonding in water(l) molecules is random. The molecules are closely packed. • The hydrogen bonding in water(s) molecules has a specific open lattice pattern. The molecules are farther apart.

The End
- Slides: 58