Chemical Bonding Chapter 4 Section 1 A chemical

Chemical Bonding Chapter 4 Section 1

• A chemical bond is: a force of attraction between any two atoms in a compound. • Bonding between atoms occurs because it creates a more stable arrangement for the atoms.

Lewis Symbols – Dot Diagrams • Convenient way to show the valence electrons

Three types of bonding • Metallic bonding – results from the attraction between metal atoms and the surrounding sea of electrons • Ionic bonding – results from the electrical attraction between large numbers of cations and anions • Covalent bonding – results from the sharing of electron pairs between two atoms

Ionic Bonding • Many atoms transfer electrons and other atoms accept electrons, creating cations (positive metal ions) and anions (negative nonmetal ions). • The resulting ions are attracted to each other by electrostatic force.

Ionic bonding between Na and Cl


Covalent bonding • In many cases electrons do not completely transfer from one atom to another. • The electrons between atoms are shared.

Covalent bonding between H 2 • Hydrogen’s electron configuration is 1 s 1 • Because both H atoms need 1 more electron to become isoelectronic with He, it is unlikely that either will give up an electron. • They share the two electrons. H· + H· H: H

Types of Covalent Bonds • When electrons are shared equally the bond is called a NONPOLAR covalent bond. (i. e. H 2) • Sometimes the electrons between two atoms are NOT shared equally. The bond created is called a POLAR covalent bond. . . H· + ·Cl: H: Cl: . .

How to classify bond types • Electronegativity – measure of the ability of an atom to attract electrons in a chemical bond • Each element on the periodic table is assigned an electronegativity value (see page 87) that ranges from 0. 7 to 4. 0. • The difference in the electronegativity determines the bonding type (ionic, polar covalent, or nonpolar covalent).

If the electronegativity difference is: 1. 7 and higher ionic 0. 3 to 1. 7 polar covalent 0. 0 to 0. 3 nonpolar covalent

Practice Problems What type of bond will occur between iodine and the following elements: cesium, iron, and sulfur? The electronegativities of each are: Iodine 2. 5 Cesium 0. 7 Iron 1. 8 Sulfur 2. 5

Answers to problems Bonding between I and: Electronegativity difference Bond Type Cesium 2. 5 – 0. 7 = 1. 8 Ionic Iron 2. 5 – 1. 8 = 0. 7 Polar covalent Sulfur 2. 5 – 2. 5 = 0. 0 Nonpolar covalent

What if I get an electronegativity difference that is 0. 3 or 1. 7? • These cut-off numbers are guidelines. • It is a gradual change not stair-step.

Determine the type of bond between the following pairs. Bonding between Li & Cl S & O Ca & Br P & H Si & Cl S & Br Electronegativity difference Bond type
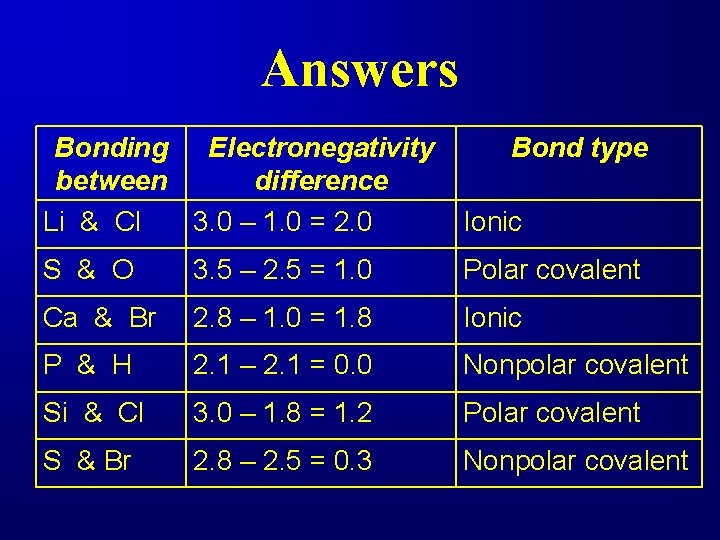
Answers Bonding Electronegativity between difference Li & Cl 3. 0 – 1. 0 = 2. 0 Bond type Ionic S & O 3. 5 – 2. 5 = 1. 0 Polar covalent Ca & Br 2. 8 – 1. 0 = 1. 8 Ionic P & H 2. 1 – 2. 1 = 0. 0 Nonpolar covalent Si & Cl 3. 0 – 1. 8 = 1. 2 Polar covalent S & Br 2. 8 – 2. 5 = 0. 3 Nonpolar covalent
- Slides: 17