Chemical Bonding Chapter 3 262022 FSS 11034 Chemistry

Chemical Bonding : Chapter 3 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi. 1
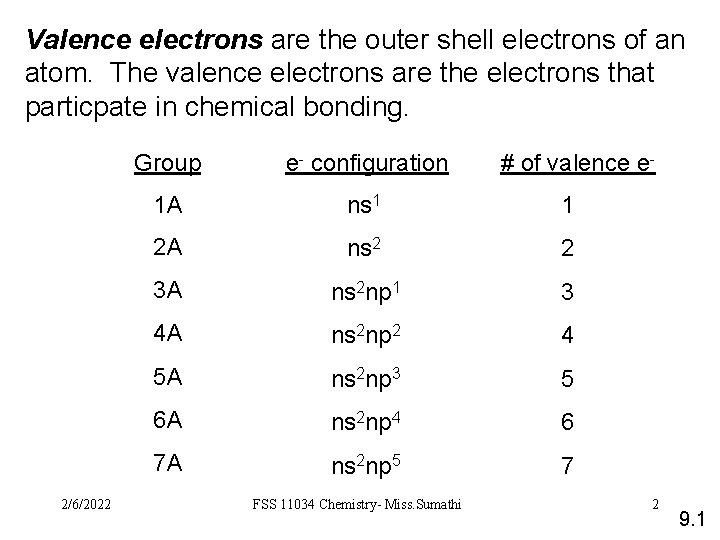
Valence electrons are the outer shell electrons of an atom. The valence electrons are the electrons that particpate in chemical bonding. 2/6/2022 Group e- configuration 1 A ns 1 1 2 A ns 2 2 3 A ns 2 np 1 3 4 A ns 2 np 2 4 5 A ns 2 np 3 5 6 A ns 2 np 4 6 7 A ns 2 np 5 7 FSS 11034 Chemistry- Miss. Sumathi # of valence e- 2 9. 1
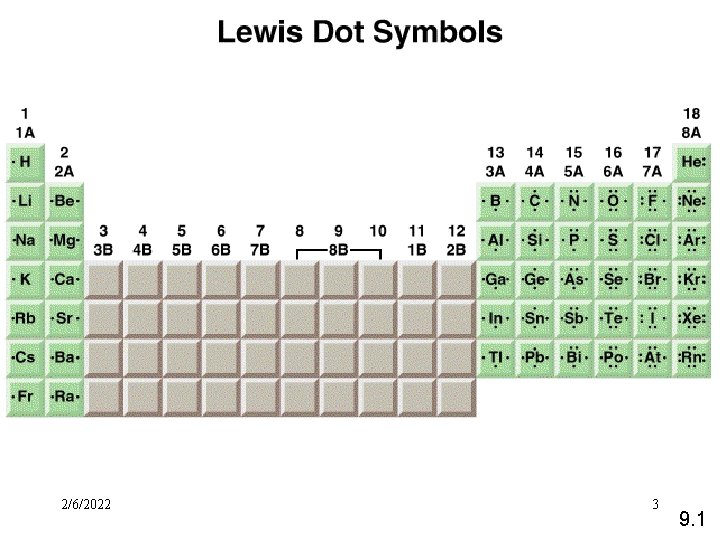
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 3 9. 1

2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 4

• In the formation of electrovalent bond / ionic bond: An ionic bond formed by electrostatic forces. Ionic bond formed by ions with 2 different charges and through a electron transfer. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 6

• Metal elements will donate electrons while non-metal will receive electrons to achieve octet. • This happen because metal more electropositive while non-metal more electronegative. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 7
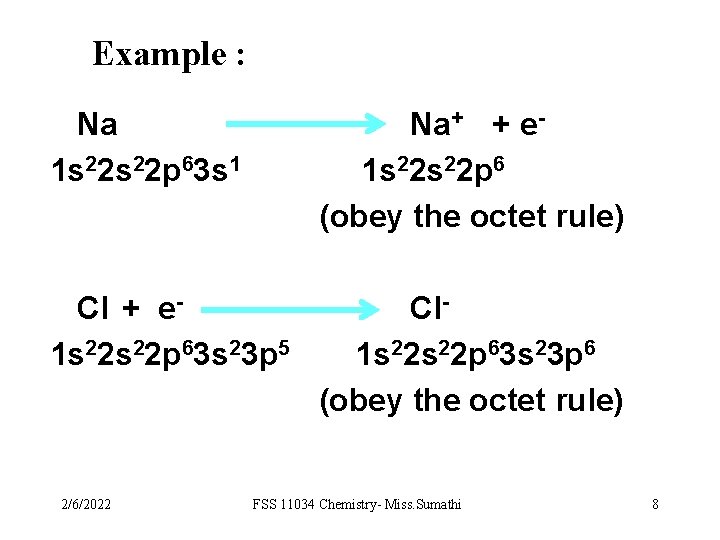
Example : Na 1 s 22 p 63 s 1 Na+ + e 1 s 22 p 6 (obey the octet rule) Cl + e 1 s 22 p 63 s 23 p 5 Cl 1 s 22 p 63 s 23 p 6 (obey the octet rule) 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 8
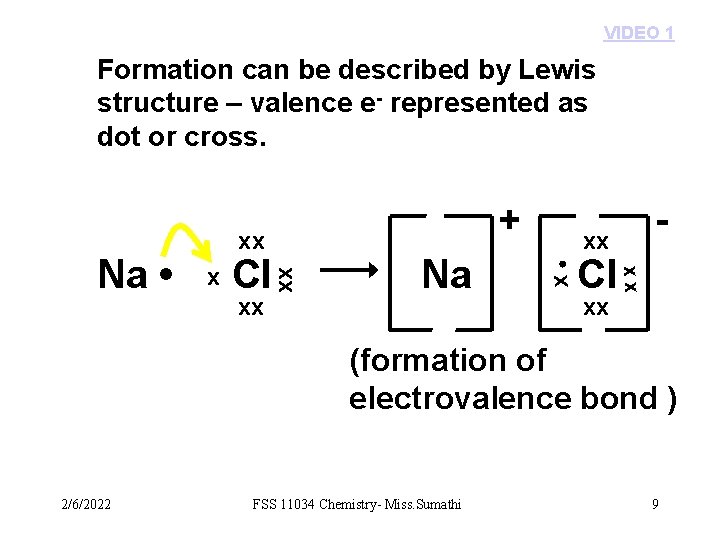
VIDEO 1 Formation can be described by Lewis structure – valence e- represented as dot or cross. xx xx Na Cl xx xx Cl - xx • x x xx Na • + (formation of electrovalence bond ) 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 9
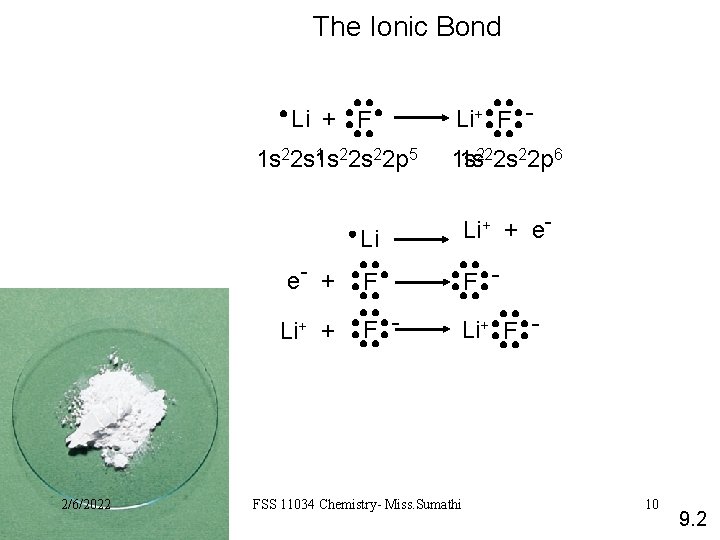
The Ionic Bond Li + F 1 22 s 22 p 5 1 s 22 s 1 s e- + Li+ + 2/6/2022 Li+ F 1 s 1 s 222 s 22 p 6 Li Li+ + e- F F - Li+ F - FSS 11034 Chemistry- Miss. Sumathi 10 9. 2

Exercise: a) Calcium bromide, Ca. Br 2 b) Plumbum oxide, Pb. O 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 11

Covalent Bond 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 12

Formed by sharing 1 or more pairs of valence electrons between the bonded atoms, involving only-non metal atoms. H. + 2/6/2022 . H H. . H or H ─ H FSS 11034 Chemistry- Miss. Sumathi 13

- Atoms other than H tends to form bonds until it is surrounded by 8 valence electrons. - Lone pairs are pairs of valence electrons that is not involved in covalent bond formation. - Can form different types of covalent bonds Ø single bond, 2 atoms are held by 1 pair of e. Ø double bond, 2 atoms shared 2 pairs of e. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 14
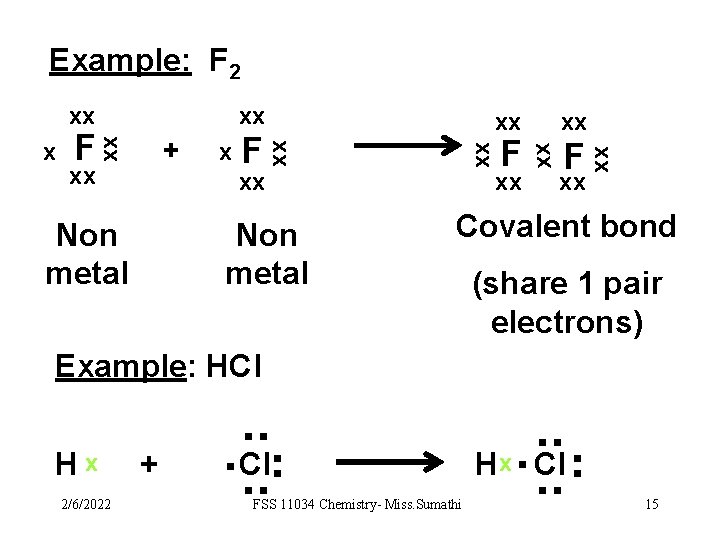
Example: F 2 xx xx x F xx Non metal F xx xx + xx xx F xx xx xx Covalent bond (share 1 pair electrons) Example: HCl FSS 11034 Chemistry- Miss. Sumathi . . Hx. Cl. . 2/6/2022 + . . Hx . . . Cl. . 15
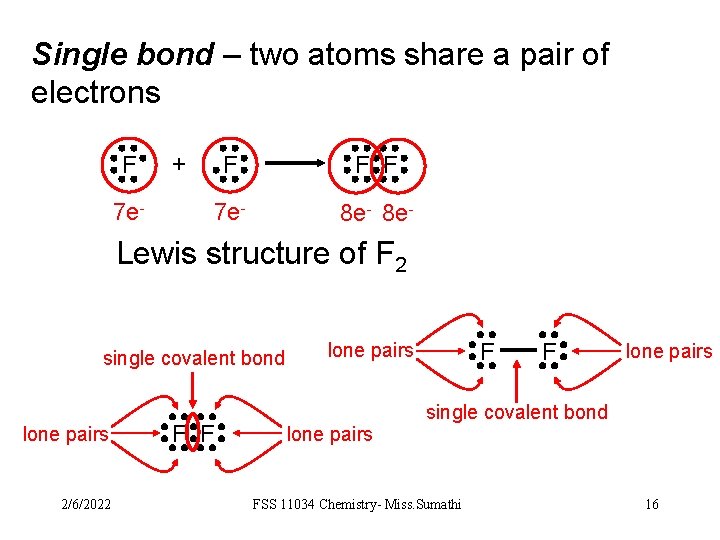
Single bond – two atoms share a pair of electrons F 7 e- + F F F 7 e- 8 e- Lewis structure of F 2 single covalent bond lone pairs 2/6/2022 F F F lone pairs single covalent bond FSS 11034 Chemistry- Miss. Sumathi 16
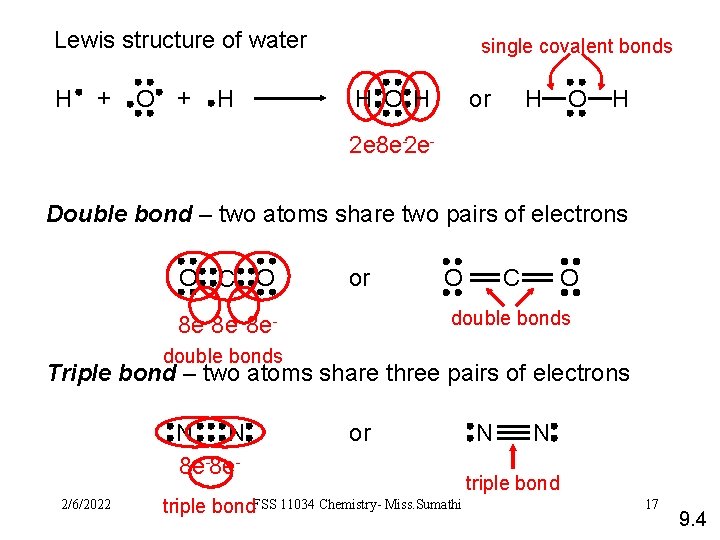
Lewis structure of water H + O + H single covalent bonds H O H or H O H 2 e-8 e-2 e. Double bond – two atoms share two pairs of electrons O C O or O O C double bonds 8 e- 8 edouble bonds Triple bond – two atoms share three pairs of electrons N N 8 e-8 e 2/6/2022 or triple bond. FSS 11034 Chemistry- Miss. Sumathi N N triple bond 17 9. 4

- Covalent bond formed when only 1 atom can provide both electrons of the shared pairs. - Also known as coordinate bond. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 18
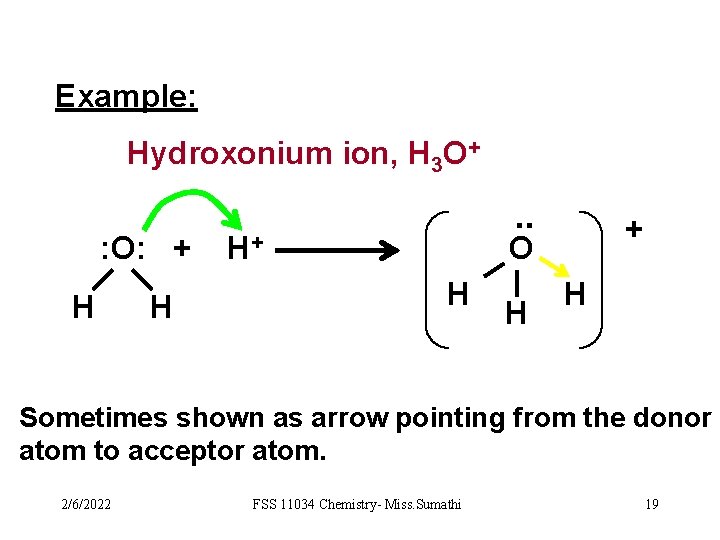
Example: Hydroxonium ion, H 3 O+ : O: + H H . . H+ + O H H H Sometimes shown as arrow pointing from the donor atom to acceptor atom. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 19

PHYSICAL PROPERTIES IONIC BOND COVALENT BOND • High melting and boiling points • A large amount of energy is required to break the bond, because the ions are held together by strong electrostatic forces of attraction. • Low melting and boiling points The melting and boiling points of covalent compounds are generally low. This is because of the fact that the forces between the molecules are weak and thus are easily overcome at low temperatures. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 20

Ionic Bond Covalent bond • Electrical conductor • In solution or in their molten states, ions become free to move and good conductors of electricity in these states. However, these do not conduct in their solid state because of strong electrostatic forces between the ions, that does not make them mobile. • Non-conductors • Since covalent compounds do not give ions in solution, these are poor • conductors of electricity in the fused or dissolved state. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 21

Ionic Bond Covalent bond • Solubility • Ionic compounds are • Covalent compounds are generally soluble in water generally insoluble or less and other polar solvents soluble in water and in other having high dielectric polar solvents. They are constants. This is due to however, soluble in nonlarger electrostatic polar solvents such as interactions between polar benzene, carbon solvents and ions. However tetrachloride etc. ionic compounds are insoluble in non-polar 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 22 solvents.

Lewis Structure 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 23

Writing Lewis Structures 1. Draw skeletal structure of compound showing what atoms are bonded to each other. Put the least electronegative element in the center. 2. Count total number of valence e-. Add 1 for each negative charge. Subtract 1 for each positive charge. 3. Complete an octet for all terminal atoms except hydrogen 4. If structure contains too many electrons, form double and triple bonds on central atom as needed. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 24
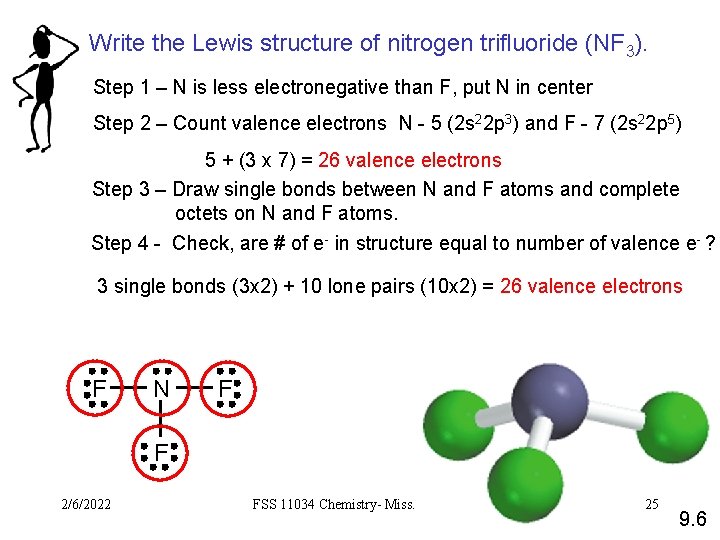
Write the Lewis structure of nitrogen trifluoride (NF 3). Step 1 – N is less electronegative than F, put N in center Step 2 – Count valence electrons N - 5 (2 s 22 p 3) and F - 7 (2 s 22 p 5) 5 + (3 x 7) = 26 valence electrons Step 3 – Draw single bonds between N and F atoms and complete octets on N and F atoms. Step 4 - Check, are # of e- in structure equal to number of valence e- ? 3 single bonds (3 x 2) + 10 lone pairs (10 x 2) = 26 valence electrons F N F F 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 25 9. 6

Species Step 1 Total no. of valence electron: 1 N atom = 1 x 5 = 5 3 F atoms = 3 x 1 = 3 8 2 H is less electronegative than F. Skeletal structure: F N F F 3 Draw a single bond. Complete the octets of the terminal atoms. . . F─N─F F NF 3 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 26

Write the Lewis structure of the carbonate ion (CO 32 -). Step 1 – C is less electronegative than O, put C in center Step 2 – Count valence electrons C - 4 (2 s 22 p 2) and O - 6 (2 s 22 p 4) -2 charge – 2 e 4 + (3 x 6) + 2 = 24 valence electrons Step 3 – Draw single bonds between C and O atoms and complete octet on C and O atoms. Step 4 - Check, are # of e- in structure equal to number of valence e- ? 3 single bonds (3 x 2) + 10 lone pairs (10 x 2) = 26 valence electrons Step 5 - Too many electrons, form double bond and re-check # of e- O C 2/6/2022 O O 2 single bonds (2 x 2) = 4 1 double bond = 4 8 lone pairs (8 x 2) = 16 Total = 24 FSS 11034 Chemistry- Miss. Sumathi 27 9. 6

Try it out • What is the Lewis structure for SF 4? • What is the Lewis structure for CCl 4 (a substance once used as a dry cleaning solvent until it was found to be very toxic) • Draw Lewis structure that obey the octet rule for the ions NO 3 -, SO 322/6/2022 FSS 11034 Chemistry- Miss. Sumathi 28

Intermolecular Forces and Liquids and Solids 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 29

• Intermolecular forces are attractive forces between molecules. • generally much weaker than covalent bonds. • The strength of intermolecular forces determine the physical properties of the substances 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 30

• Attractive forces between neutral molecules are 1. Van der Waals forces 2. Hydrogen Bond 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 31

Van der Waals forces • 3 types of Van der Waals forces a. dipole-dipole forces b. induce dipole-dipole forces c. London dispersion forces • The intermolecular forces increase in strength according to the following; London < dipole-dipole < H-bonding dispersion 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 32

(a) Dipole-Dipole Forces VIDEO 1 • A dipole-dipole forces exists between neutral polar molecules. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 33
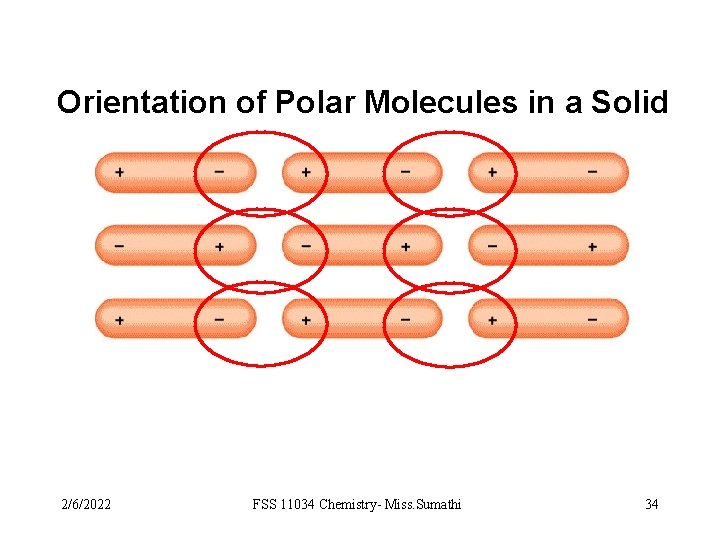
Orientation of Polar Molecules in a Solid 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 34

• The strength of dipole-dipole forces depends on the dipole moment of the molecule. • A more polar molecule, have stronger dipole -dipole forces. • Example: the dipole-dipole forces in H-Cl are stronger than in H-Br 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 35

b) London Dispersion Forces VIDEO 2 • Exist in all atoms and molecules. • Non-polar molecules would not seem to have any basis for attractive interactions • At any instant one side of the molecule has higher electron density while the other side has lower electron density. • At this instant, the atoms or molecules become a small instantaneous dipole. Then, induce a dipole on the neighboring atom and molecule. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 36
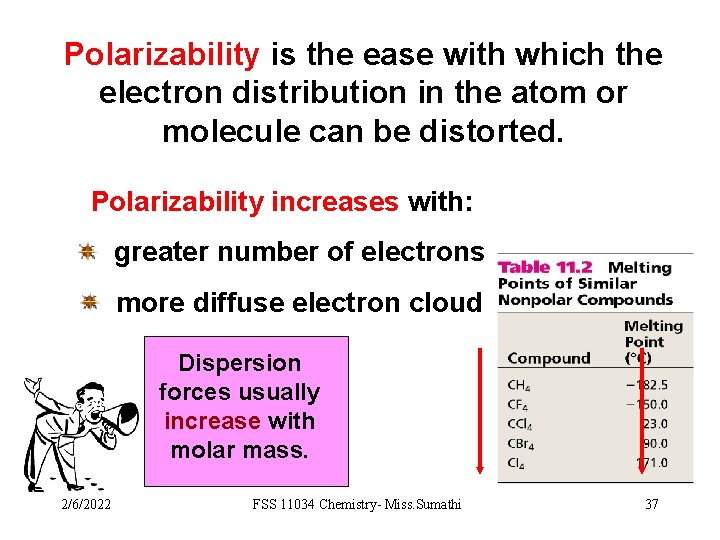
Polarizability is the ease with which the electron distribution in the atom or molecule can be distorted. Polarizability increases with: greater number of electrons more diffuse electron cloud Dispersion forces usually increase with molar mass. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 37

Factors which influence van der Waals forces i. Molecular size ii. Molecular shape iii. Molecular Polarity 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 38

i. Molecular Size VIDEO 3 § go down a group, number of electrons increases, atomic radius increases. §The more electrons, the more distance over which they can move, the bigger the possible temporary dipoles and therefore the bigger the dispersion forces. §the bigger molecules are "stickier". 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 39

VIDEO 4 ii. Molecular shape • Long thin molecules can develop bigger temporary dipoles due to electron movement than short fat ones containing the same numbers of electrons. • Long thin molecules can also lie closer together - these attractions are at their most effective if the molecules are really close. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 40
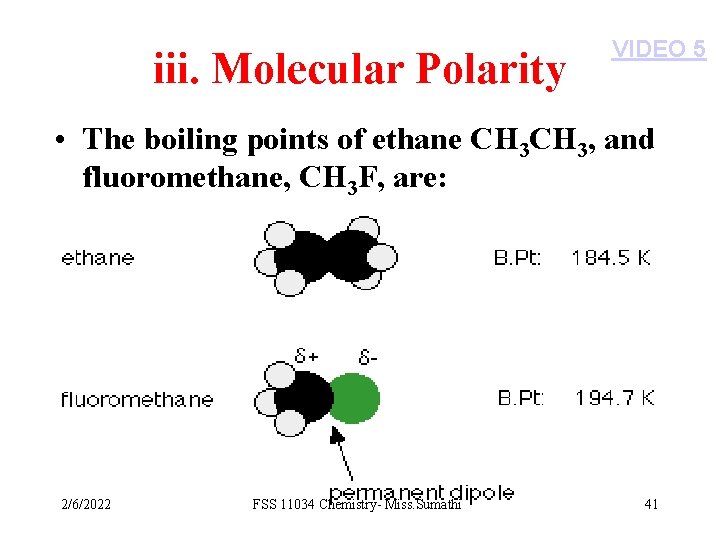
iii. Molecular Polarity VIDEO 5 • The boiling points of ethane CH 3, and fluoromethane, CH 3 F, are: 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 41

What type(s) of intermolecular forces exist between each of the following molecules? • HBr is a polar molecule: dipole-dipole forces. There also dispersion forces between HBr molecules. • SO 2 is a polar molecule: dipole-dipole forces. There also dispersion forces between SO 2 molecules. S O O • CH 4 is nonpolar: dispersion forces. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 42

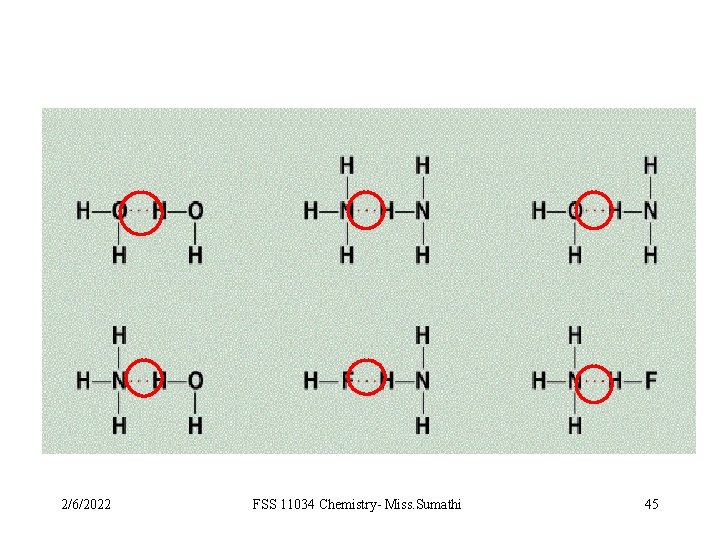
VIDEO 1 c)Hydrogen bonding - attraction force between the positivelycharged H atom of a molecule and the negatively-charged electronegative atom of another molecule. Example: N-H, O-H, or F-H bond 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 43
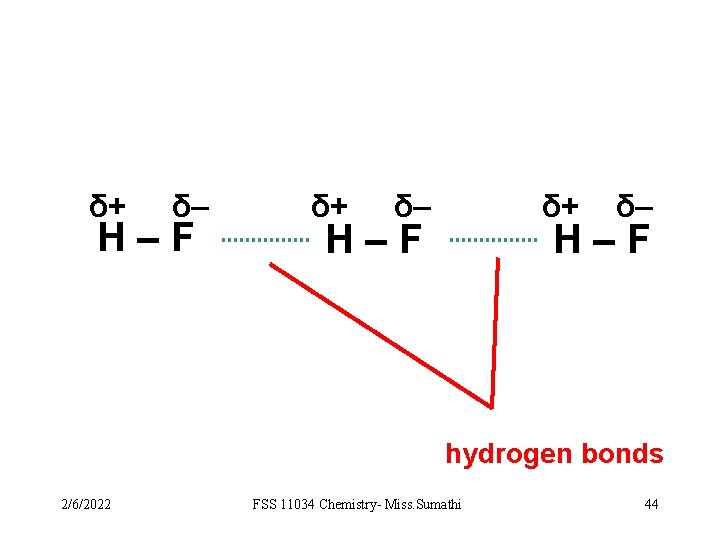
δ+ δ– H–F δ+ δ– δ+ H–F δ– H–F hydrogen bonds 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 44
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 45

Effects of Hydrogen Bond on Physical Properties i) the boiling points ii) the solubility of simple covalent molecules such as NH 3, CH 3 OH in water iii) The density of water and ice 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 46
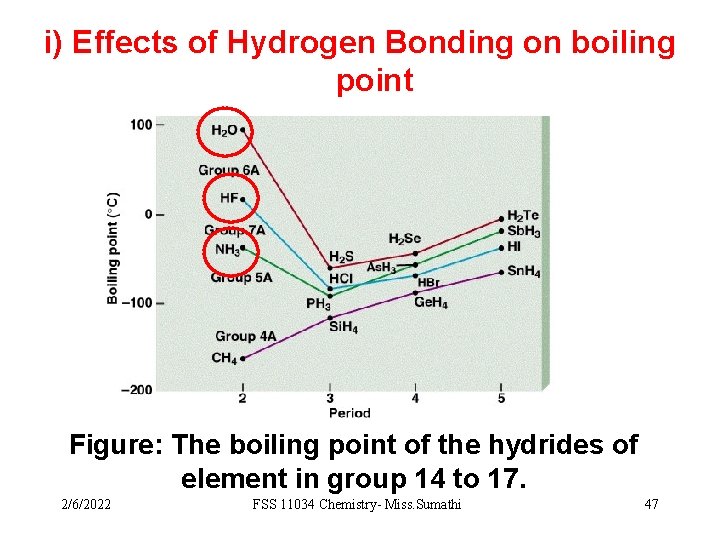
i) Effects of Hydrogen Bonding on boiling point Figure: The boiling point of the hydrides of element in group 14 to 17. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 47

From the figure: 1. The boiling points of hydrides of group 14 elements (CH 4, Si. H 4, Ge. H 4 and Sn. H 4) displays normal behavior. CH 4 < Si. H 4 < Ge. H 4 < Sn. H 4 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 48

q The type of intermolecular forces exist between the molecules are only Van der Waals q Hydrogen bonds are not present. q The boiling point increase regularly when the relative molecular mass increases. because the van der Waals forces increase as the molecular size increases. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 49

2. For hydrides of group 15, 16 & 17 elements (except NH 3, H 2 O & HF) hydrogen bonds are not present. q The increase in the boiling points for the hydrides of each periodic group is due to the increase in van der waals forces as the molecular size increases. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 50

3. The strength of H-bond is proportional to the polarity of the bond. Thus, H-F > H 2 O > NH 3 However, boiling point increases in such order H 2 O > HF > NH 3 q Even though fluorine is more electronegative than oxygen, H 2 O has higher boiling point than HF because there are 4 H-bonds per H 2 O molecule compared to only 2 per HF molecule. q HF has higher boiling point than NH 3 because of stronger H-bond of HF. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 51

ii) Effects of H-Bonding on the solubility of simple covalent compounds - Water is a good solvent for liquids and gases consisting of small polar molecules, that can form hydrogen bonds with water molecules. - For example, ammonia is soluble in water because ammonia molecules can form hydrogen bonds with water molecules. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 52
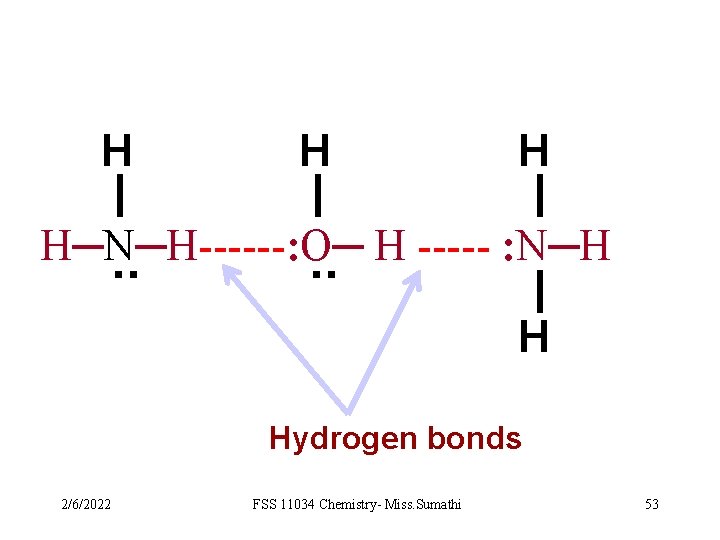
H H H H─N─H------: O─ H ----: N─H. . H Hydrogen bonds 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 53

q Most organic compounds are insoluble in water. q Organic compounds that are soluble in water include: - amines, for example ethylamine C 2 H 5 NH 2. - alcohols, for example ethanol, C 2 H 5 OH. - Carboxylic acid, such as ethanoic acid, CH 3 CH 2 OH. q Amines are soluble in water because the -NH 2 group in amines can form H-bonds with the water molecules. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 54
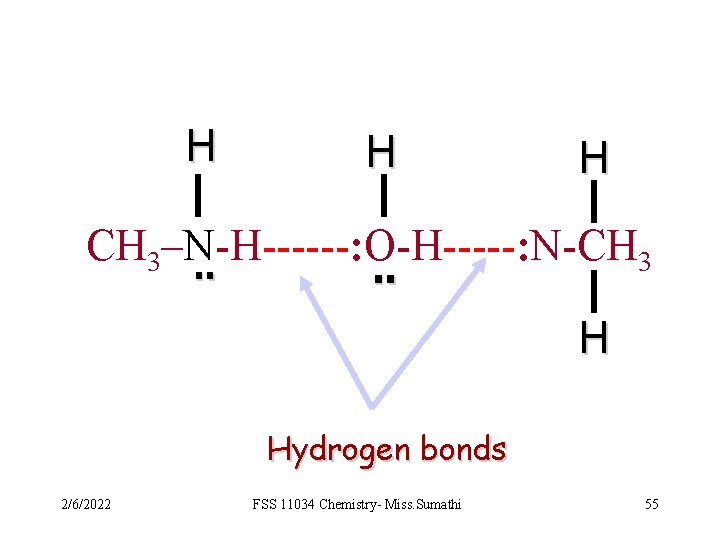
H H H CH 3–N-H------: O-H-----: N-CH 3. . H Hydrogen bonds 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 55

q. Alcohol and carboxylic acids are soluble in water because the -OH groups in the compounds can form H-bonds with the water molecules. However, not all organic compounds that contain -NH 2 group or –OH groups are soluble in water. As the relative molecular mass increases, the non-polar hydrocarbon portion becomes larger. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 56

q. Since hydrocarbons are insoluble in water, the solubility decrease as the relative molecular mass of the organic compound increases 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 57
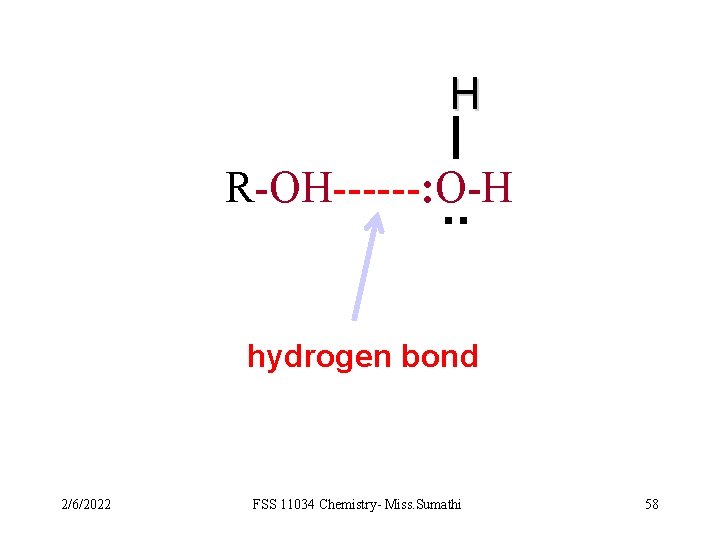
H R-OH------: O-H . . hydrogen bond 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 58
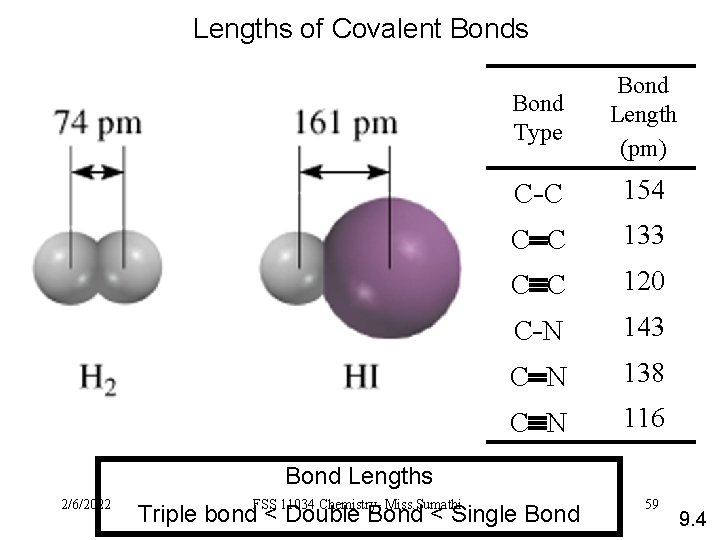
Lengths of Covalent Bonds Bond Type Bond Length (pm) C -C 154 C C 133 C C 120 C -N 143 C N 138 C N 116 Bond Lengths 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi Triple bond < Double Bond < Single Bond 59 9. 4

Electronegativity is the ability of an atom to attract toward itself the electrons in a chemical bond. Electron Affinity - measurable, Cl is highest X (g) + e- X-(g) Electronegativity - relative, F is highest 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 60 9. 5

VIDEO 2 Polarised bond ? Bonds between the atoms that are polarised because of the difference in electronegativities of the atoms. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 61

2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 62 9. 5

VIDEO 3 Polar molecule A dipole molecule in which the positive and negative pole can be distinguished (because of the separation of the charge). Example: + O = C = O polar or not? 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 63
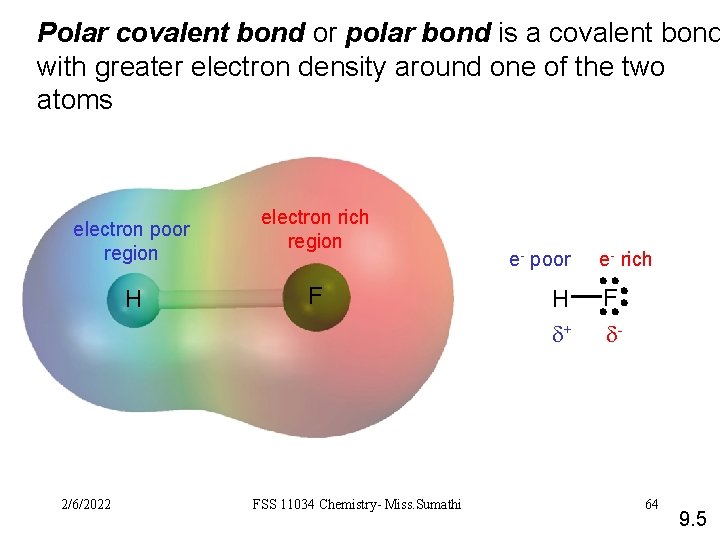
Polar covalent bond or polar bond is a covalent bond with greater electron density around one of the two atoms electron poor region H 2/6/2022 electron rich region F FSS 11034 Chemistry- Miss. Sumathi e- poor H d+ e- rich F d- 64 9. 5

Dipole moment (µ) - A quantitative measure of the polarity of a bond that can only be measured exp. - A polar molecule has µ ≠ 0 Non-polar molecule has µ = 0 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 65
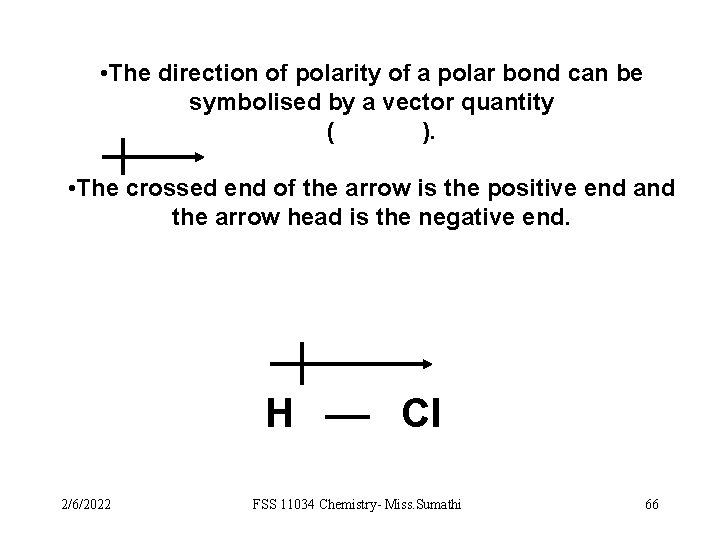
• The direction of polarity of a polar bond can be symbolised by a vector quantity ( ). • The crossed end of the arrow is the positive end and the arrow head is the negative end. H Cl 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 66
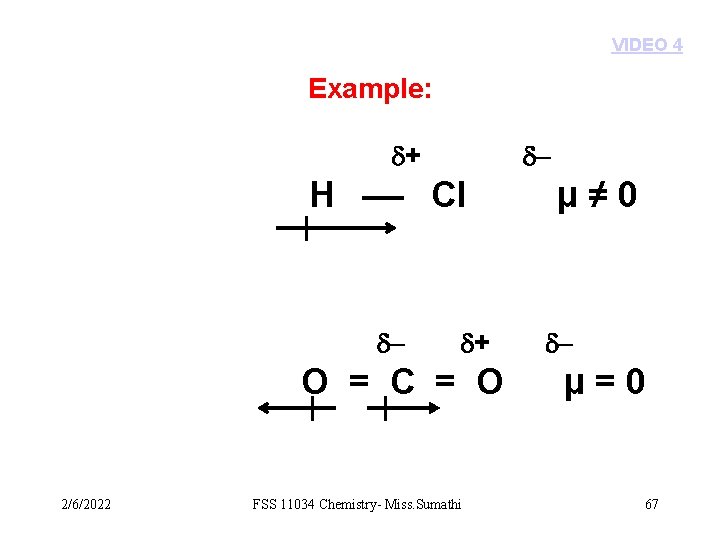
VIDEO 4 Example: + H Cl + O = C = O 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi µ≠ 0 µ=0 67

Determining polar and non-polar molecules Use three-step approach outlined below : Ø Use electronegativity values to predict bond dipoles. Ø Use the VSEPR method to predict the molecular shape. Ø From the molecular shape, determine whether bond dipoles cancel to give a non-polar molecule or combine to produce a (resultant dipole moment) for the molecule. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 68

Molecules with different atoms & asymmetrically arranged are polar. Example: + H Cl 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 69
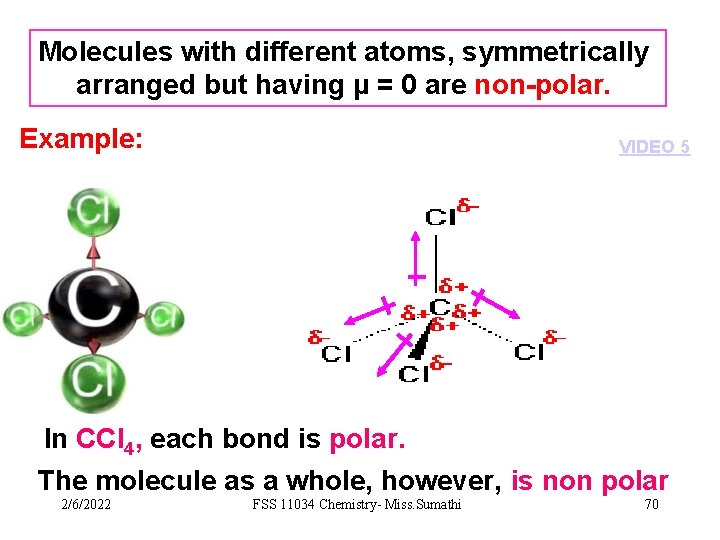
Molecules with different atoms, symmetrically arranged but having µ = 0 are non-polar. Example: VIDEO 5 In CCl 4, each bond is polar. The molecule as a whole, however, is non polar 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 70

Example: In SF 6, each bond is polar. The molecule is non polar 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 71
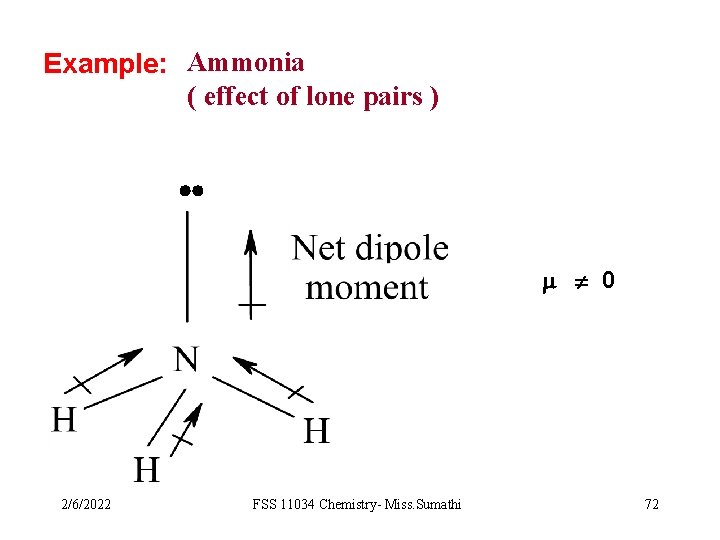
Example: Ammonia ( effect of lone pairs ) 0 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 72
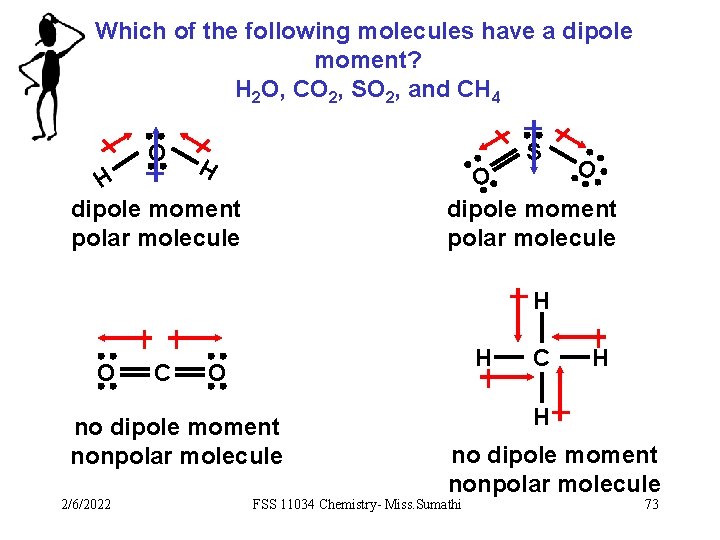
Which of the following molecules have a dipole moment? H 2 O, CO 2, SO 2, and CH 4 O S H O O dipole moment polar molecule H O C H O no dipole moment nonpolar molecule 2/6/2022 C H H no dipole moment nonpolar molecule FSS 11034 Chemistry- Miss. Sumathi 73

Evaluation 2 Predicting the polarity of molecules. State which of them are polar and which are non-polar. a) H 2 O b) NH 3 c) CCl 4 d) CHCl 3 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 74
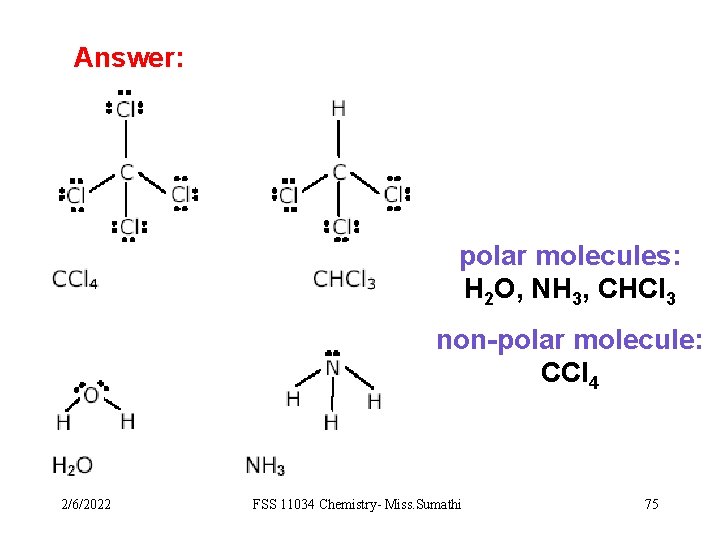
Answer: polar molecules: H 2 O, NH 3, CHCl 3 non-polar molecule: CCl 4 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 75

Classify the following bonds as ionic, polar covalent, or covalent: The bond in Cs. Cl; the bond in H 2 S; and the NN bond in H 2 NNH 2. Cs – 0. 7 Cl – 3. 0 – 0. 7 = 2. 3 Ionic H – 2. 1 S – 2. 5 – 2. 1 = 0. 4 Polar Covalent N – 3. 0 – 3. 0 = 0 Covalent 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 76 9. 5
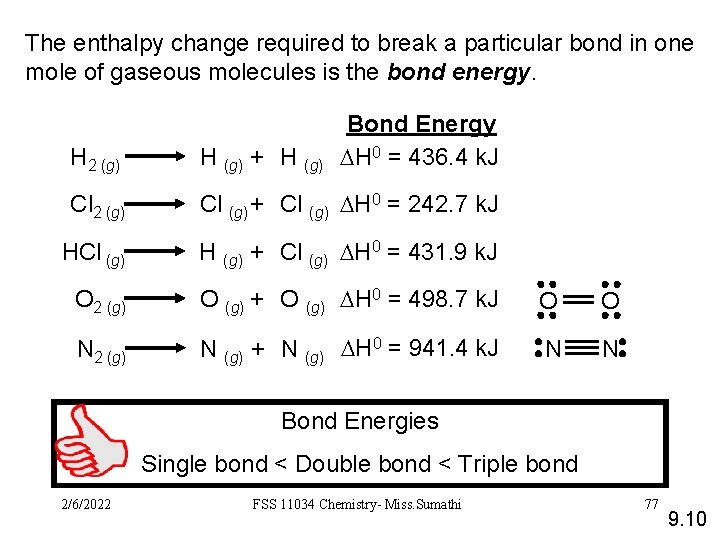
The enthalpy change required to break a particular bond in one mole of gaseous molecules is the bond energy. Bond Energy DH 0 = 436. 4 k. J H 2 (g) H (g) + H (g) Cl 2 (g) Cl (g) + Cl (g) DH 0 = 242. 7 k. J HCl (g) H (g) + Cl (g) DH 0 = 431. 9 k. J O 2 (g) O (g) + O (g) DH 0 = 498. 7 k. J O O N 2 (g) N (g) + N (g) DH 0 = 941. 4 k. J N N Bond Energies Single bond < Double bond < Triple bond 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 77 9. 10
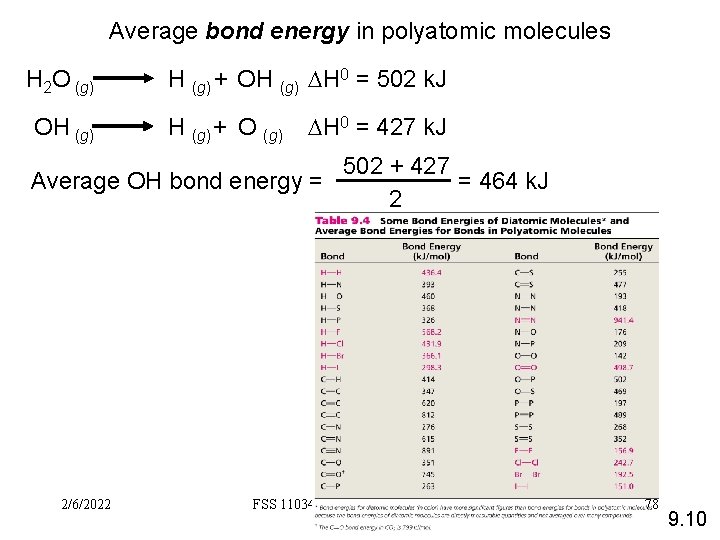
Average bond energy in polyatomic molecules H 2 O (g) OH (g) + OH (g) DH 0 = 502 k. J H (g) + O (g) DH 0 = 427 k. J 502 + 427 = 464 k. J Average OH bond energy = 2 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 78 9. 10
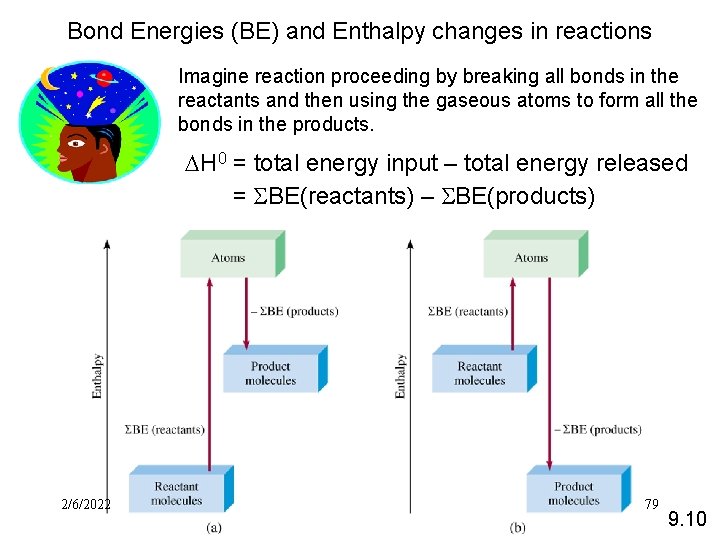
Bond Energies (BE) and Enthalpy changes in reactions Imagine reaction proceeding by breaking all bonds in the reactants and then using the gaseous atoms to form all the bonds in the products. DH 0 = total energy input – total energy released = SBE(reactants) – SBE(products) 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 79 9. 10

Use bond energies to calculate the enthalpy change for: H 2 (g) + F 2 (g) 2 HF (g) DH 0 = SBE(reactants) – SBE(products) Type of bonds broken H H F F Type of bonds formed H F Number of bonds broken Bond energy (k. J/mol) Energy change (k. J) 1 1 436. 4 156. 9 Number of bonds formed Bond energy (k. J/mol) Energy change (k. J) 2 568. 2 1136. 4 DH 0 = 436. 4 + 156. 9 – 2 x 568. 2 = -543. 1 k. J 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 80 9. 10
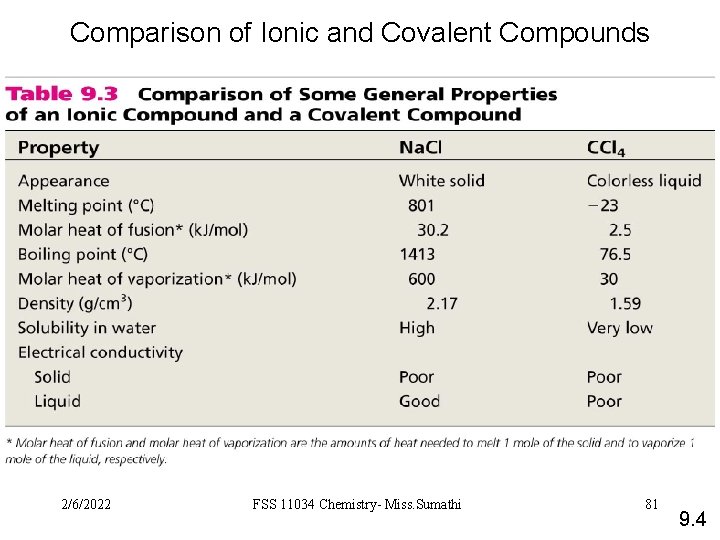
Comparison of Ionic and Covalent Compounds 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 81 9. 4

2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 82 9. 5
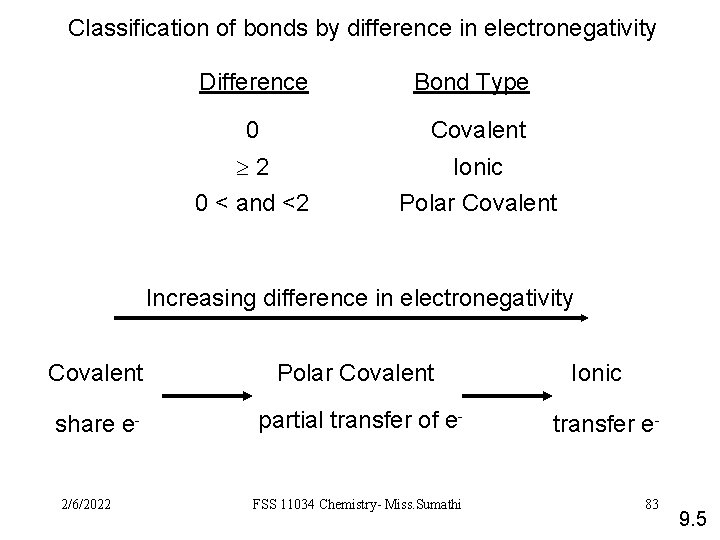
Classification of bonds by difference in electronegativity Difference Bond Type 0 Covalent 2 0 < and <2 Ionic Polar Covalent Increasing difference in electronegativity Covalent Polar Covalent share e- partial transfer of e- transfer e- FSS 11034 Chemistry- Miss. Sumathi 83 2/6/2022 Ionic 9. 5

Metallic Bond • A metallic bond can be defined as the electrostatic force between the positively charged metallic ions and the ‘sea’ of electrons. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 84

Ø In a metallic bond, metals atoms can be imagined as an array of positive ions immersed in a sea of delocalized valence electrons. Ø These delocalized electrons are not bound to individual atoms and they can therefore serve to bind large numbers of metal atoms together. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 85

PHYSICAL PROPERTIES • Metals are shiny solids are room temperature (except mercury), with characteristic high melting points and densities. • Properties of metals: – Able to be deformed without breaking. Malleability is the ability of a metal to be hammered into shapes. – Ductility is the ability of a metal to be drawn into wire. – Because the valence electrons can move freely (or mobility of the delocalized electrons), metals are good heat conductors and electrical conductors. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 86

The change of melting point and boiling points • A melting point of a metal indicates the strength of its metallic bonding in its structure. The strength of metallic bonding 2/6/2022 no. of valence e per atom α radius of the atom FSS 11034 Chemistry- Miss. Sumathi 87

So, the bonding will be weaker in sodium ( 1 valence e) compared to magnesium (2 valence e) and aluminium (3 valence e). 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 88
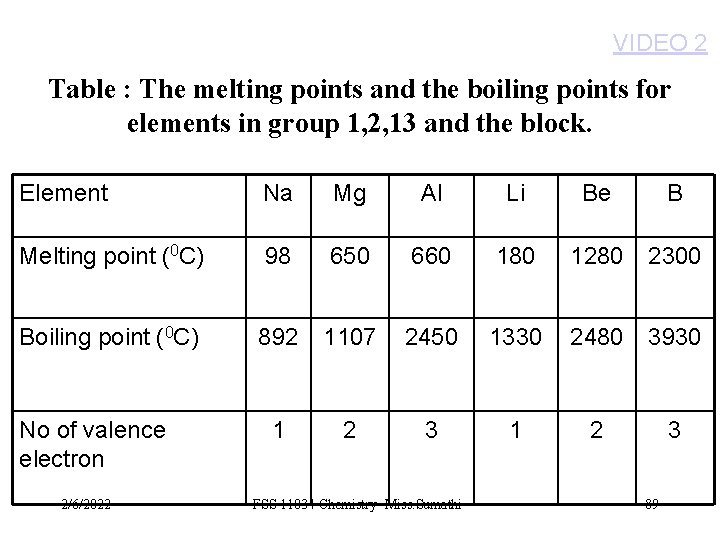
VIDEO 2 Table : The melting points and the boiling points for elements in group 1, 2, 13 and the block. Element Na Mg Al Li Be B Melting point (0 C) 98 650 660 180 1280 2300 Boiling point (0 C) 892 1107 2450 1330 2480 3930 1 2 3 No of valence electron 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 89

- Def: The 3 D arrangement of atoms in a molecule. - affects physical and chemical properties. - predicted by using Valence-Shell Electron Pair Repulsion (VSEPR) model. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 90

VIDEO 2 - The study of molecular geometry. - States “The valence electron pairs surrounding a central atom repel one another. Thus, the orbital containing those electron pairs are oriented to be as far apart as possible”. 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 91

3 types of repulsive forces: Ø Ø Ø between bonding pairs between lone pairs between a bonding pair & lone pair bonding-pair vs. bonding pair repulsion 2/6/2022 lone-pair vs. bonding < pair repulsion FSS 11034 Chemistry- Miss. Sumathi < lone-pair vs. lone pair repulsion 92
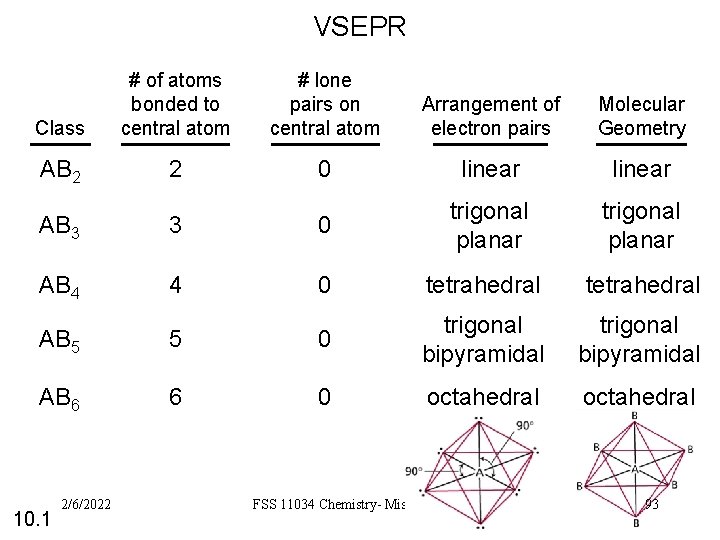
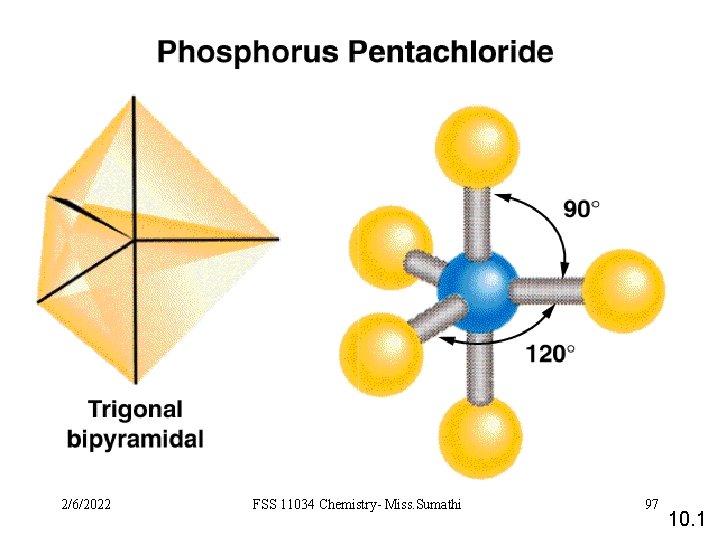
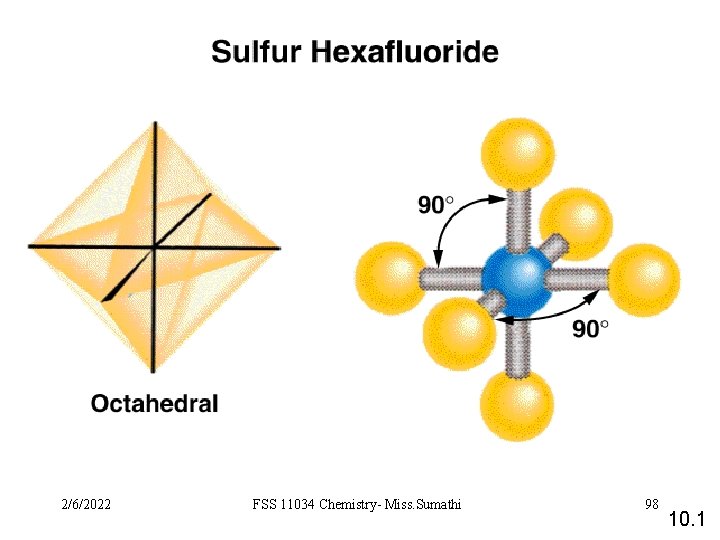
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear trigonal planar AB 3 3 0 trigonal planar AB 4 4 0 tetrahedral AB 5 5 0 trigonal bipyramidal AB 6 6 0 octahedral 10. 1 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 93
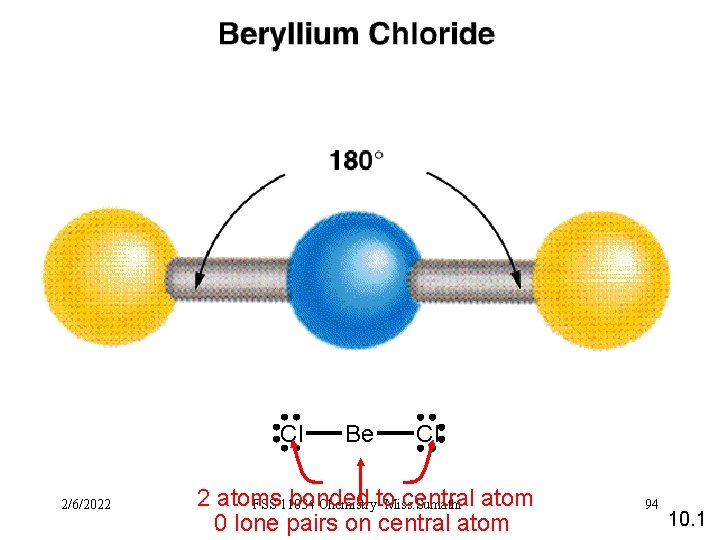
Cl 2/6/2022 Be Cl 2 atoms bonded central atom FSS 11034 Chemistry-to Miss. Sumathi 0 lone pairs on central atom 94 10. 1
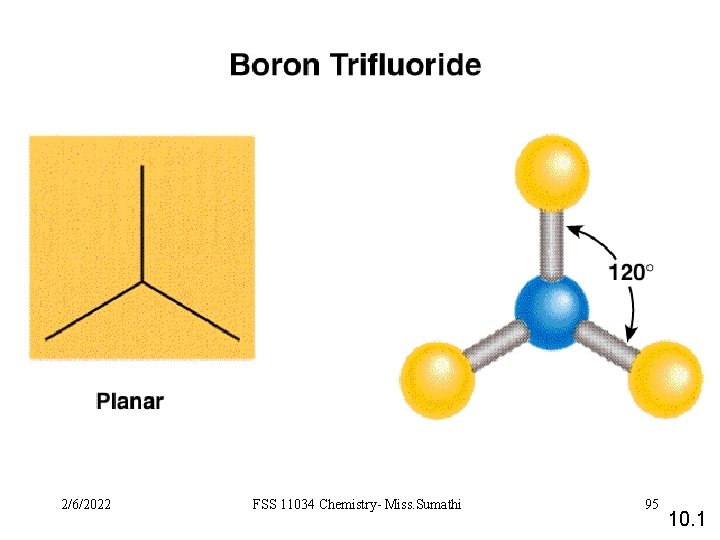
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 95 10. 1
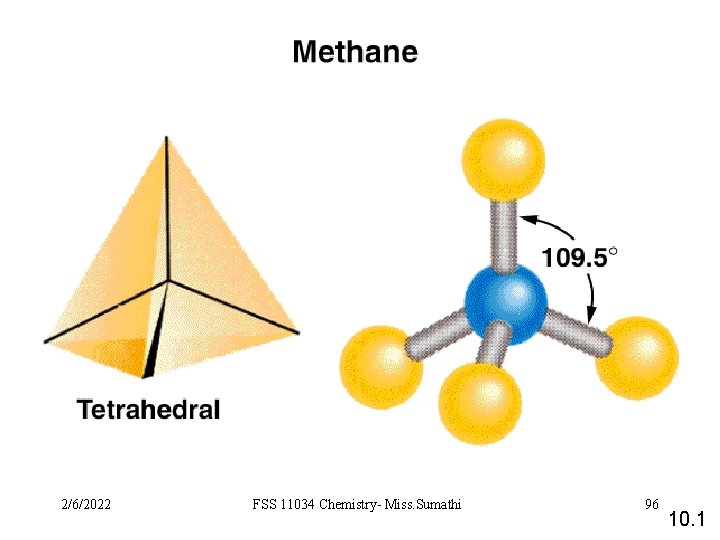
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 96 10. 1
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 97 10. 1
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 98 10. 1
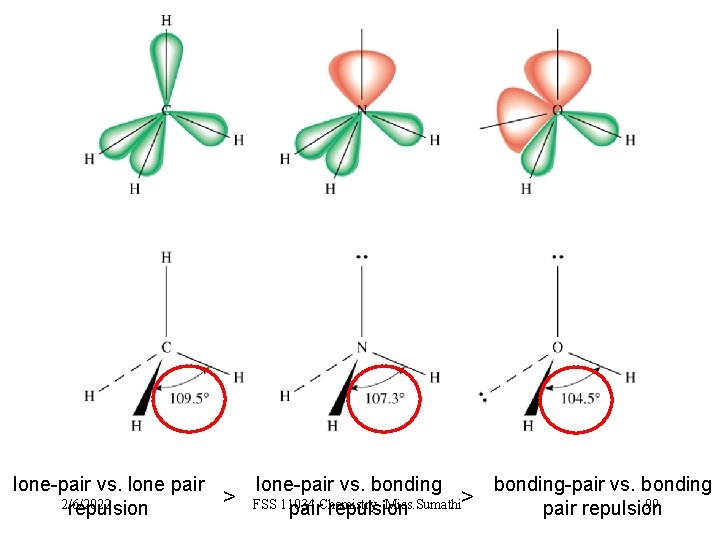
lone-pair vs. lone pair 2/6/2022 repulsion > lone-pair vs. bonding-pair vs. bonding FSS 11034 Miss. Sumathi> 99 pair. Chemistryrepulsion pair repulsion
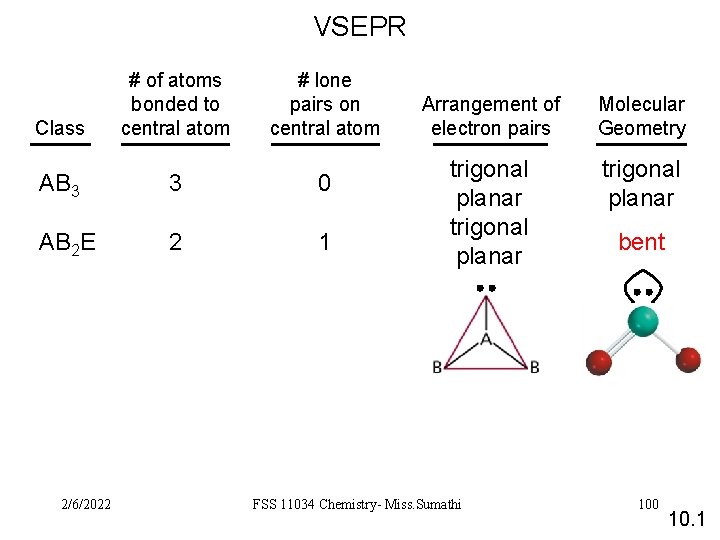
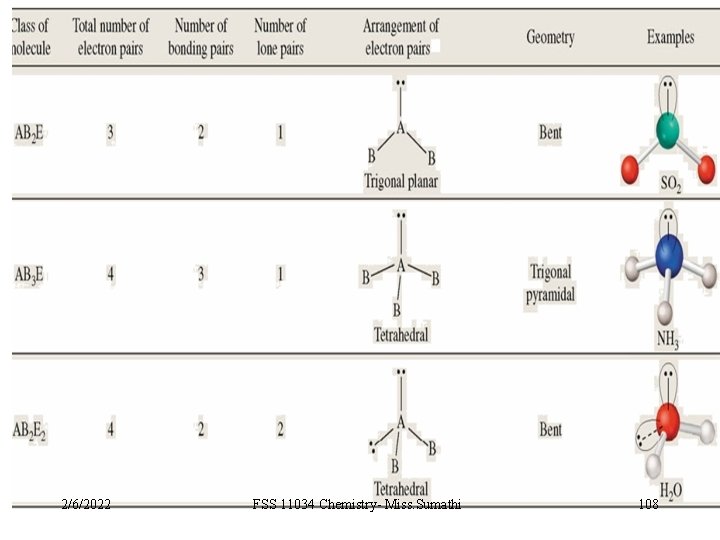
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom AB 3 3 0 AB 2 E 2 1 2/6/2022 Arrangement of electron pairs Molecular Geometry trigonal planar FSS 11034 Chemistry- Miss. Sumathi bent 100 10. 1
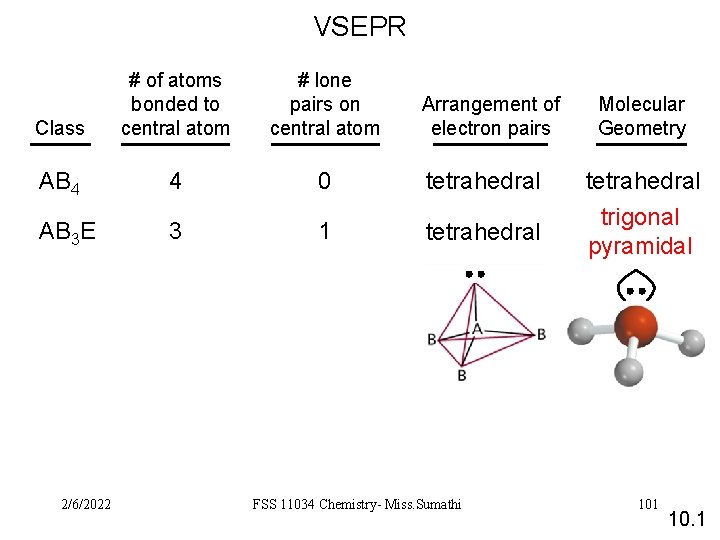
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom AB 4 4 0 AB 3 E 3 1 2/6/2022 Arrangement of electron pairs Molecular Geometry tetrahedral trigonal pyramidal FSS 11034 Chemistry- Miss. Sumathi 101 10. 1
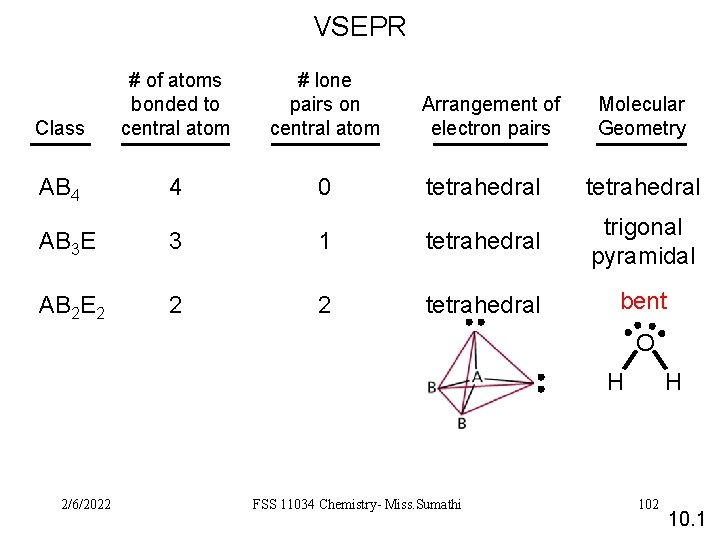
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom AB 4 4 0 Arrangement of electron pairs Molecular Geometry tetrahedral AB 3 E 3 1 tetrahedral trigonal pyramidal AB 2 E 2 2 2 tetrahedral bent O H 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi H 102 10. 1
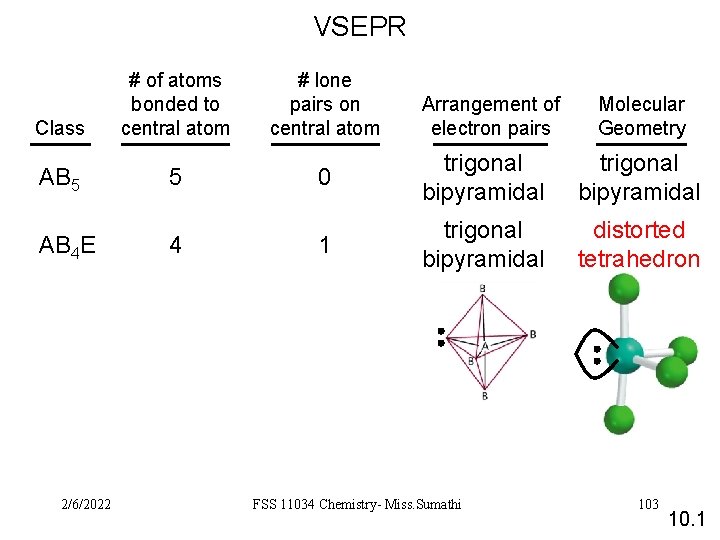
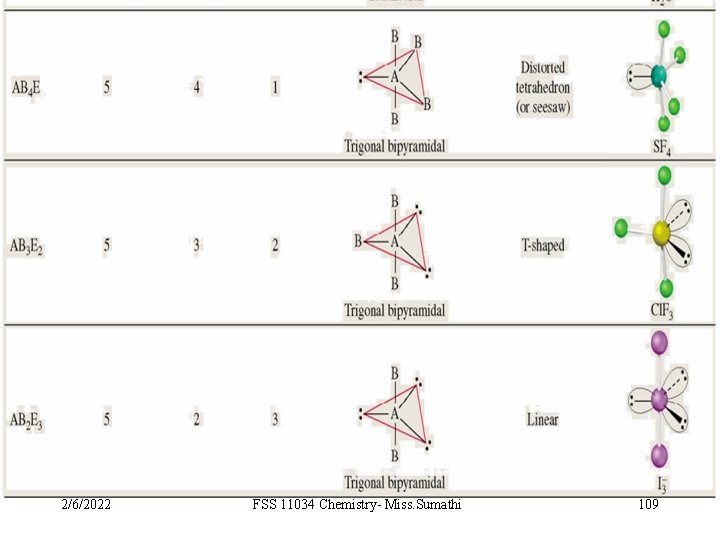
VSEPR Class AB 5 AB 4 E 2/6/2022 # of atoms bonded to central atom 5 4 # lone pairs on central atom Arrangement of electron pairs Molecular Geometry 0 trigonal bipyramidal 1 trigonal bipyramidal distorted tetrahedron FSS 11034 Chemistry- Miss. Sumathi 103 10. 1
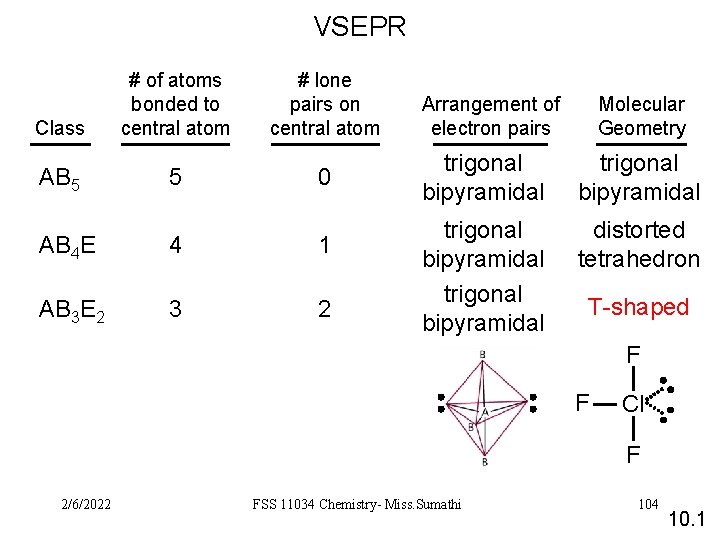
VSEPR Class AB 5 # of atoms bonded to central atom 5 # lone pairs on central atom 0 AB 4 E 4 1 AB 3 E 2 3 2 Arrangement of electron pairs Molecular Geometry trigonal bipyramidal distorted tetrahedron T-shaped F F Cl F 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 104 10. 1
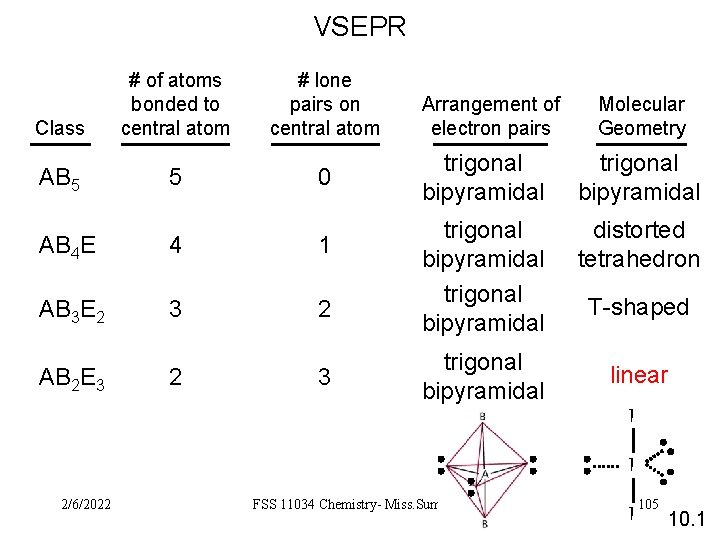
VSEPR Class AB 5 # of atoms bonded to central atom 5 # lone pairs on central atom 0 AB 4 E 4 1 AB 3 E 2 3 2 AB 2 E 3 2 3 Arrangement of electron pairs Molecular Geometry trigonal bipyramidal distorted tetrahedron trigonal bipyramidal T-shaped linear I I 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi I 105 10. 1
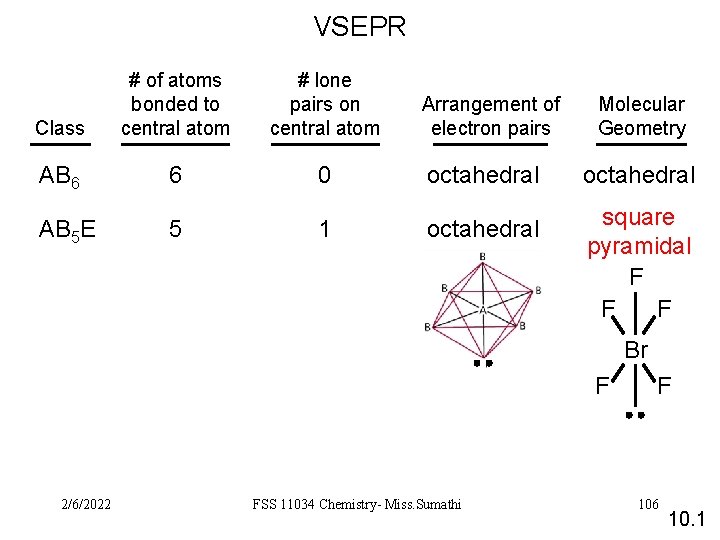
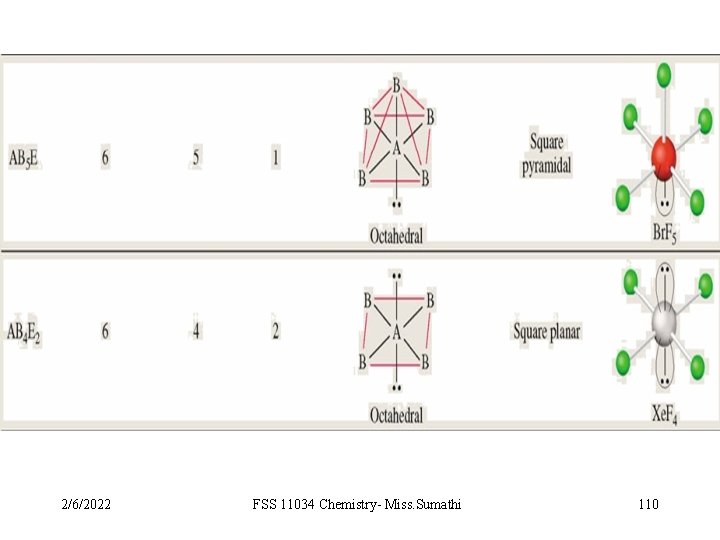
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom AB 6 6 0 octahedral AB 5 E 5 1 octahedral square pyramidal F F F Arrangement of electron pairs Molecular Geometry Br F 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi F 106 10. 1
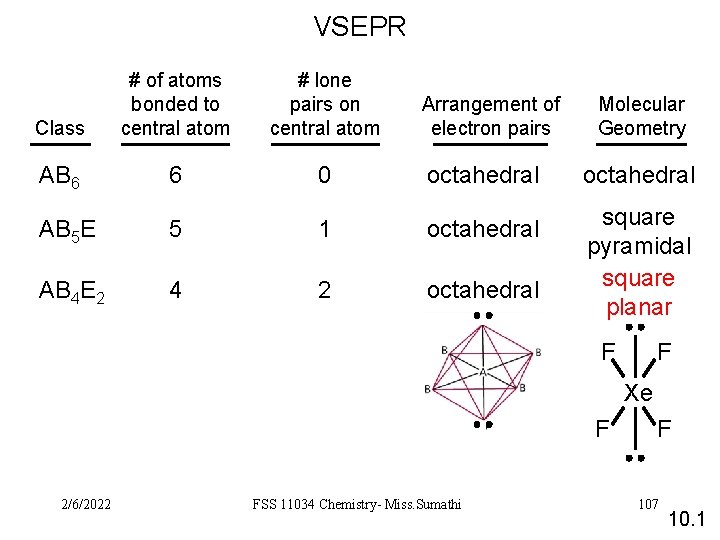
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom AB 6 6 0 octahedral AB 5 E 5 1 octahedral AB 4 E 2 4 2 octahedral square pyramidal square planar Arrangement of electron pairs Molecular Geometry F F Xe F 2/6/2022 FSS 11034 Chemistry- Miss. Sumathi F 107 10. 1
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 108
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 109
2/6/2022 FSS 11034 Chemistry- Miss. Sumathi 110
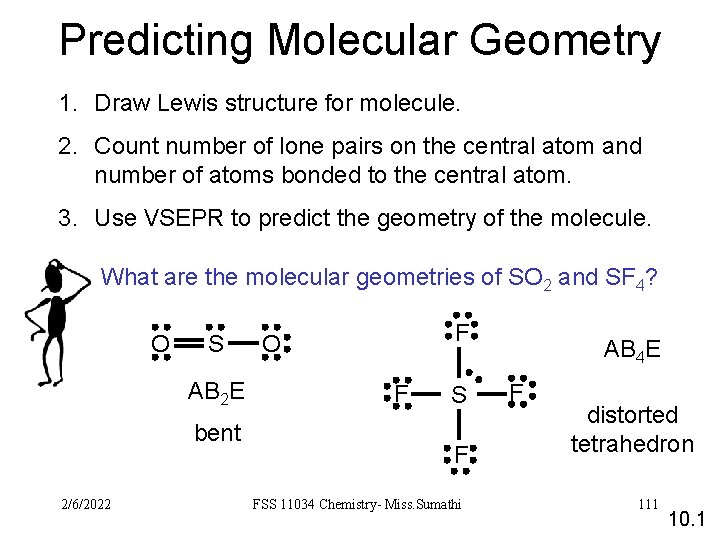
Predicting Molecular Geometry 1. Draw Lewis structure for molecule. 2. Count number of lone pairs on the central atom and number of atoms bonded to the central atom. 3. Use VSEPR to predict the geometry of the molecule. What are the molecular geometries of SO 2 and SF 4? O S AB 2 E bent 2/6/2022 F O F S F FSS 11034 Chemistry- Miss. Sumathi AB 4 E F distorted tetrahedron 111 10. 1

Evaluation : Use the VSEPR model to predict the geometry of the following molecules and ions: 2/6/2022 a) C 2 H 4 b) OF 2 c) Al. Cl-4 d) As. H 3 FSS 11034 Chemistry- Miss. Sumathi 112
- Slides: 111