Chemical Bonding Chapter 20 Stability Achieved by Bonding

Chemical Bonding Chapter 20

Stability Achieved by Bonding • Elements other than Group 18 achieve stability (full set of valence electrons) by bonding and forming compounds • 3 types of chemical bonds • Ionic • Covalent • Metallic

1. Ionic Bonding • Formed between metals and nonmetals • Formed between positive and negative ions • One atom gains electrons from the other atom that loses

Naming Binary Ionic Compounds 1. Name metal first (positive ion) 2. Then add the name of the nonmetal (negative ion) but change the ending to “ide” • Ending is usually the last syllable Example: sodium (Na) and chlorine (Cl) Name: Sodium Chloride Chemical Formula: Na. Cl

Practice Problem 1: Name the ionic compound shown below: Mg. F 2 Magnesium fluoride

Practice Problem 2: Name the ionic compound shown below: Li 2 O Lithium oxide

Writing Chemical Formulas for Binary Ionic Compounds 1. Write the symbol for each element with it’s oxidation number (charge) • Remember: write metal first 2. Chemical formulas of ionic compounds are always neutral (so oxidation numbers must balance) • • Add subscripts to the symbols for the ions until the sum of the charges is equal to zero No subscripts are needed if only one ion is present

Example: Calcium Chloride 1. Ca+2 2. Ca. Cl 2 Cl-1

Practice Problem 3: Write the chemical formula for aluminum bromide.

Practice Problem 4: Write the chemical formula for magnesium nitride.

Practice Problem 5 Write the chemical formula for calcium oxide.

Basic Properties of Ionic Compounds • Electrolytes • Conduct electricity in aqueous solution • Dissolve well in water • Solid crystal at room temperature • Strong interparticle forces

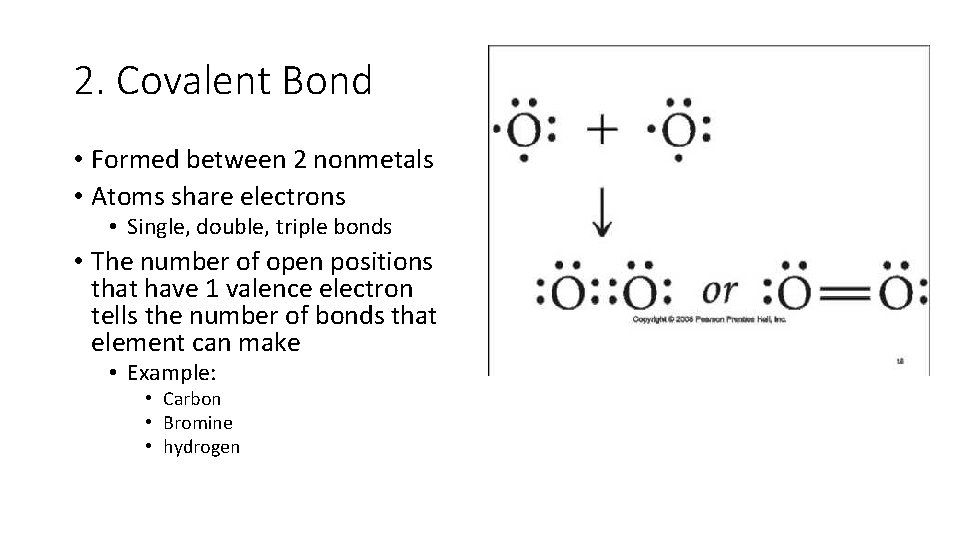
2. Covalent Bond • Formed between 2 nonmetals • Atoms share electrons • Single, double, triple bonds • The number of open positions that have 1 valence electron tells the number of bonds that element can make • Example: • Carbon • Bromine • hydrogen

• To name a covalent compound, same as ionic naming except you use prefixes to represent how many atoms of each element you have • • • 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca

Naming Molecular Compounds 1. write the name of the element that is the least electronegative first. Example: SO 2 sulfur

2. Write the name of the second element and change the ending to “ide” Example: SO 2 sulfur oxide

3. Add a prefix to the beginning of each element’s name to indicate how many of atoms of each there are (exception: you do not write the word mono in front of the first element if there is only one). Example: SO 2 sulfur dioxide

• If the vowel combination o-o or a-o appear next to each other in the name, the first of the pair is omitted Example: CO 1. Carbon 2. Carbon oxide 3. Carbon monooxide carbon monoxide

Practice Problem 1 Name the following molecular compound: CCl 4

Practice Problem 2: Name the following molecular compound: N 2 O 4

Exceptions to the naming rule: • Hydrocarbons/Organic compounds/Alkanes • Compounds made only of carbon and hydrogen and single bonds • • • CH 4 = methane C 2 H 6 = ethane C 3 H 8 = propane C 4 H 10 = butane C 5 H 12 = pentane C 6 H 14 = hexane C 7 H 16 = heptane C 8 H 18 = octane C 9 H 20 = nonane C 10 H 22 = decane
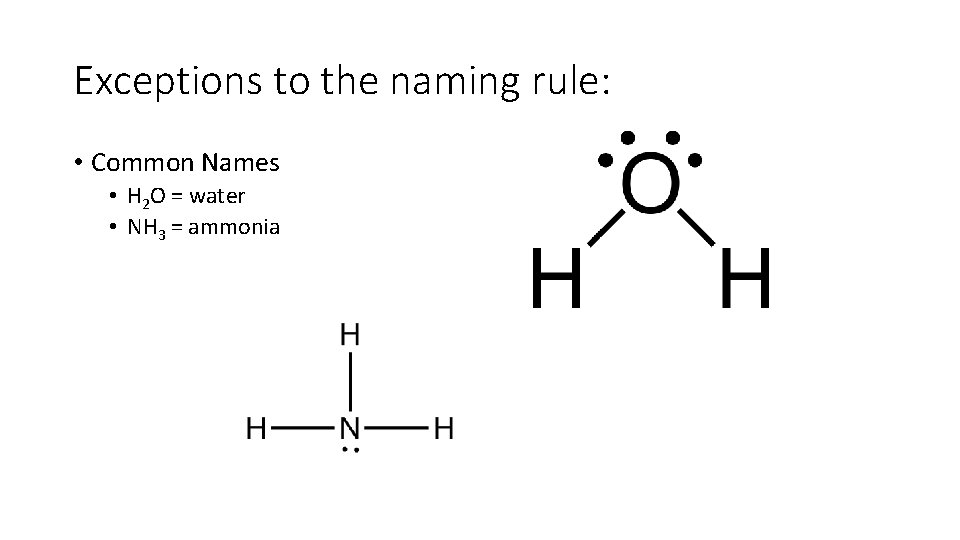
Exceptions to the naming rule: • Common Names • H 2 O = water • NH 3 = ammonia

Writing Molecular Formulas • Name tells you the elements present and the number of each atom to write as subscripts. Example: nitrogen trifluoride

Practice Problem 3: Write the molecular formula for tetraphosphorus decoxide.

Practice Problem 4: Write the molecular formula for dinitrogen monoxide.

Properties of Covalent Compounds • Lower melting point than ionic compounds • Most are liquids or gases • If solid, not as hard as ionic compounds • Weak interparticle forces between molecules • Do not conduct electricity or heat

3. Metallic Bonds • Formed between atoms of metals only • Metal cations and mobile electrons • Occurs in pure metals (Fe)
- Slides: 28