Chemical Bonding Chapter 11 Electrons and Chemical Bonding

Chemical Bonding Chapter 11

Electrons and Chemical Bonding Have you ever stopped to consider that by using the 26 letters of the alphabet, you make all of the words you use every day? Although the number of letters is limited, you combine them in different ways to make a huge number of words. This is the same with elements. We combine them to make different substances.

Combining Atoms Through Chemical Bonding Chemical Bond - is the joining of atoms to form new substances The properties of these new substances are different than the properties of the original substance. When chemical bonds form, electrons are lost, gained, or shared

Electron Number and Organization To understand how atoms form chemical bonds, you need to understand how electrons are arranged in an atom. The number of electrons in an atom = the number of protons. The number of protons can be determined by the atomic number. The electrons are organized in energy levels. 1 st Energy Level: 2 2 nd Energy Level: 8 3 rd Energy Level: 8 Diagram Fluorine:
Outer-Level Electrons and Bonding Not all electrons in an atom take part in a bonding. Most bonds form using the outer most electrons called valence electrons. Valence electron - an electron in the outermost energy level of an atom.

To bond or Not to Bond Not all atoms bond the same way. The number of valence electrons determines if an atom will form a bond. Atoms want to be like noble gases (have a full outer shell of electrons). 8 valence electrons 2 valence electrons like He Atoms bond by gaining, losing, or sharing electrons to have a full outer most energy level like the noble gases.

Complete with your partner 1. How many valence electrons does C have? 2. How many valence electrons does Ca have? 3. How many valence electrons does S have? 4. Why do elements form bonds?

Covalent Compounds Covalent Bonds are formed when two nonmetals share electrons. Covalent Bond – a bond that forms when atoms share one or more pairs of electrons

Covalent Compounds Examples of covalent compounds: 1. ammonia 2. water 3. carbon dioxide Properties 1. low melting points 2. low boiling points 3. Brittle as a solid Substances containing covalent bonds consist of individual particles called molecules. molecule - a neutral group of atoms that are joined together by one or more covalent bonds.

Covalent Compounds Simple Covalent Compounds When found in nature as pure elements, these seven elements exist only as two atoms covalently bonded. H 2 O 2 7 N 8 O N 2 Cl 2 Br 2 I 2 F 2 1 H 9 F 19 Cl 35 Br 53 I

Covalent Compounds How to draw covalent molecules: using electron dot diagrams 1. Write the symbol of the element and place one dot around the symbol for every valence electron in the atom. Examples: Carbon Oxygen Krypton Hydrogen

Covalent Compounds 2. Combine the elements where they can share electrons so each have a full outer shell. (The shared electrons are the covalent bond) Examples: Hydrogen + Chlorine Fluorine + Fluorine Water Oxygen + Oxygen

Complete with your partner Draw the Lewis Structures for each of the following CH 4 CO 2

Covalent Compounds Naming Covalent Compounds Rules: Prefixes Prefix + name One = mono Two = di Three = tri Four = tetra Five = penta Six = hexa Seven = hepta Eight= octa Nine = nona Ten = deca Prefix + name –ide Rule: Do NOT use mono- on the first element Examples: CO 2 C 2 H 4 P 2 O 5 N 3 P 6
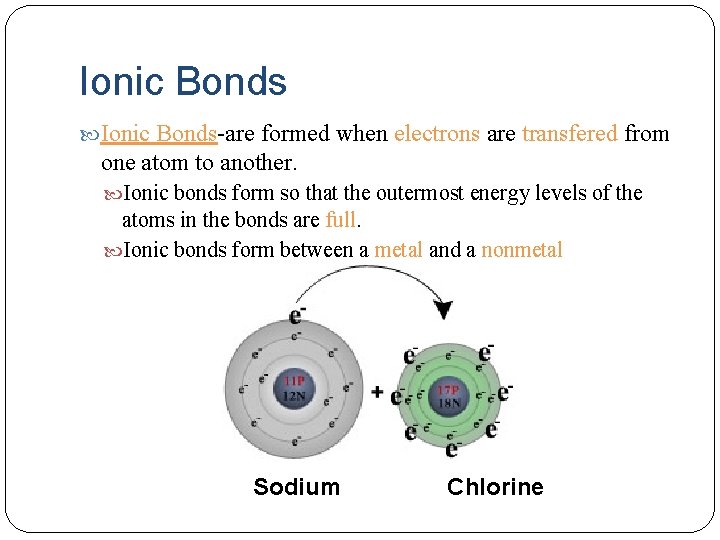
Ionic Bonds-are formed when electrons are transfered from one atom to another. Ionic bonds form so that the outermost energy levels of the atoms in the bonds are full. Ionic bonds form between a metal and a nonmetal Sodium Chlorine

Ionic Bonds Charged Particles (Magnets) A transfer of electrons between atoms changes the number of electrons in each atom but the number of protons stay the same. The negative charges and positive charges no longer cancel out and the atom becomes an ion. Ion- an atom that gained or lost electrons

Ionic Bonds Charged Particles Continued An atom cannot lose electrons without a nearby atom gaining electrons and visa versa. Metals – tend to lose electrons and become a positive ion (cation) Nonmetals – tend to gain electrons and become a negative ion (anion) -ide- the ending that is given to atoms that gain electrons Oxygen = _____ Fluorine = _____ Sulfur = _____

Ionic Compounds Metals tend to lose a certain number of electrons depending on their number of valence electrons and become positively charged Nonmetals tend to gain a certain number of electrons depending on their number of valence electrons and become negatively charged

Ionic Compounds When ionic bonds form, the number of electrons lost by the metal equals the number of electrons gain by the nonmetal atoms. The ions that bond are charged but the compounds formed are neutral because the charges on the ions cancel out.

Complete with your partner What types of elements form Ionic compounds? Why?

Ionic Compounds Writing Ionic Compounds ( metal + nonmetal) Write the metal with its’ charge Write the nonmetal with its’ charge Criss- Cross the charges Only bring down the #, NOT the charge Examples: Sodium and Chlorine Barium and Chlorine Potassium and Oxygen Lithium and Sulfur

Complete with your partner Write the formula for the ionic compound that forms: 1. Potassium + Sulfur 2. Magnesium + nitrogen 3. Aluminum + phosphorous

Ionic Compounds Naming Ionic Compounds Name of Metal + nonmetal w/ ending –ide Examples: Sodium and Chlorine Barium and Fluorine Potassium and Oxygen Lithium and Sulfur
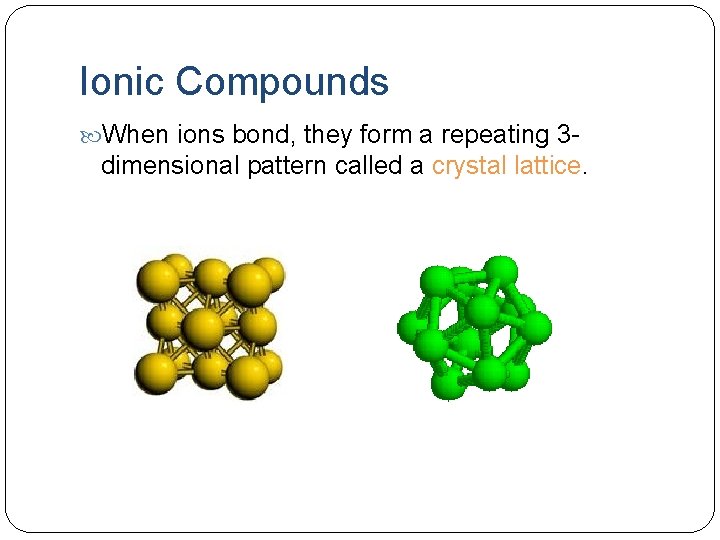
Ionic Compounds When ions bond, they form a repeating 3 - dimensional pattern called a crystal lattice.

Metallic Bonds Properties of Metals 1. malleable 2. ductile 3. conduct heat/electric well Metals are malleable and ductile because of the presence of a metallic bonds. Metallic Bond – a bond between metals of the same atom
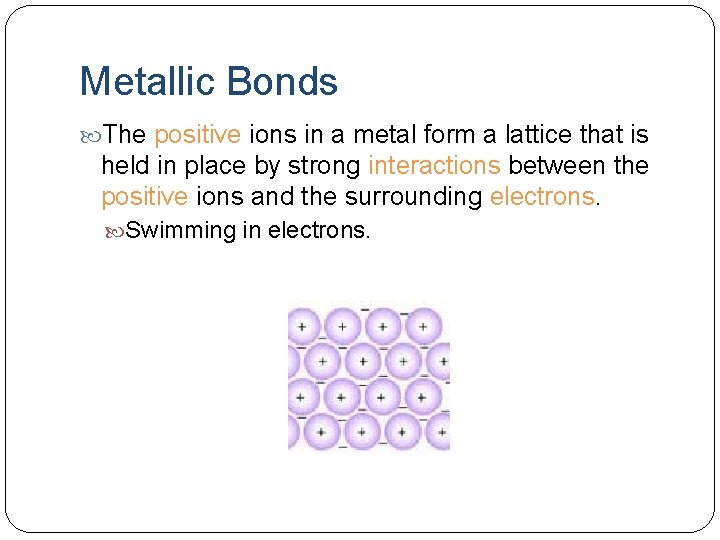
Metallic Bonds The positive ions in a metal form a lattice that is held in place by strong interactions between the positive ions and the surrounding electrons. Swimming in electrons.

Complete on your own Write a paragraph about the 3 types of bonds and the types of elements that are involved in each.
- Slides: 27