Chemical Bonding Atoms Bonding Ionic Bonds Covalent Bonds
Chemical Bonding Atoms & Bonding Ionic Bonds Covalent Bonds Additional Info: Metallic Bonds

I really hate to tell you this but you have a lot of notes to take today over some stuff that can get confusing. You really need to pay attention and ask Mrs. Taylor questions if you don’t understand.

19. 1 Atoms & Bonding What does it mean to “bond”? Think about the types of “bonds” that you might be familiar with, for example: Together we I am bonded to my husband by are one. LOVE I am bonded to my paintball buddies by a sense of camaraderie, together we are one team. 2008_149[1]. JPG 2008_150[1]. JPG. When you use glue to hold two things together they are bonded. How does this idea relate to “Atoms & Bonding”?

A Little Review You should remember the parts of an atom. The proton +p The neutron n The electron -e Of these three particles, which is the only one that has the ability to move from one place to another? The -e You should also recall that atoms are the smallest particle of an element and a molecule is the smallest particle of a compound.

An Important Piece of Information You need to always remember this. Each energy level will hold only a certain number of electrons. Count the levels from the nucleus out. There a total of 7. You need to remember also n 2 8 18 32 that an energy level must be full before the next one will hold -e You need to remember 2 – 8 – 18 (only the first 3 )

Chemical Bonds Means you should write this in your notes. When atoms chemically combine, they create a chemical bond. A chemical bond is an attractive force that holds atoms or ions together to form a molecule. Of the three main parts of an atom, only the –e is involved in forming the bond. Remember, it is the only one Why is this true? that can move. When chemical bonding occurs, electrons from one atom may be transferred to another atom or they may be shared by atoms.

Why does transfer or sharing of –e occur? In order for an atom to be “satisfied” in nature, it always tries to achieve having 8 –e in its outer energy level. This is called “stability” YOU MUST ALWAYS REMEMBER THE NUMBER 8, EIGHT, & VIII. In order to achieve “stability” atoms will gain, lose, or share the electrons in their outer energy level only. The -e in levels below the outer cloud are not involved in chemical bonding.

The three types of bonds: 19. 2 Ionic Bonds What is an “ion”? Remember from previous lessons that atoms appear in nature as neutral particles. This means that even though their outer energy levels may not have 8 –e and they are not stable, they do have the same number of +p in the nucleus as they have –e in their electron clouds. For each + charge in the nucleus they have a charge in the clouds to balance out the charge, thus they are neutral. - If an atom loses or gains electrons in its OEL it will now have an imbalance of charges, either + or -. When the charges are not balanced the atom is called an ION.
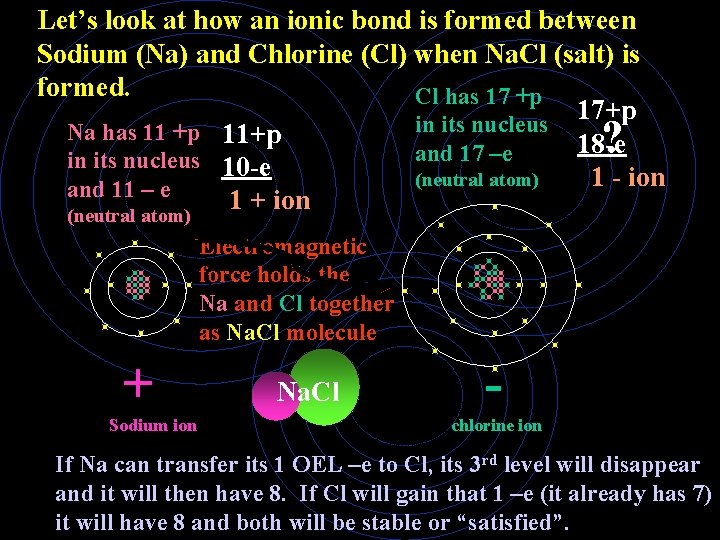
Let’s look at how an ionic bond is formed between Sodium (Na) and Chlorine (Cl) when Na. Cl (salt) is formed. Cl has 17 +p 17+p in its nucleus Na has 11 +p 11+p ? 18 -e and 17 –e in its nucleus 10 -e (neutral atom) 1 - ion and 11 – e 1 + ion (neutral atom) Electromagnetic force holds the Na and Cl together as Na. Cl molecule + Sodium ion Na. Cl chlorine ion If Na can transfer its 1 OEL –e to Cl, its 3 rd level will disappear and it will then have 8. If Cl will gain that 1 –e (it already has 7) it will have 8 and both will be stable or “satisfied”.
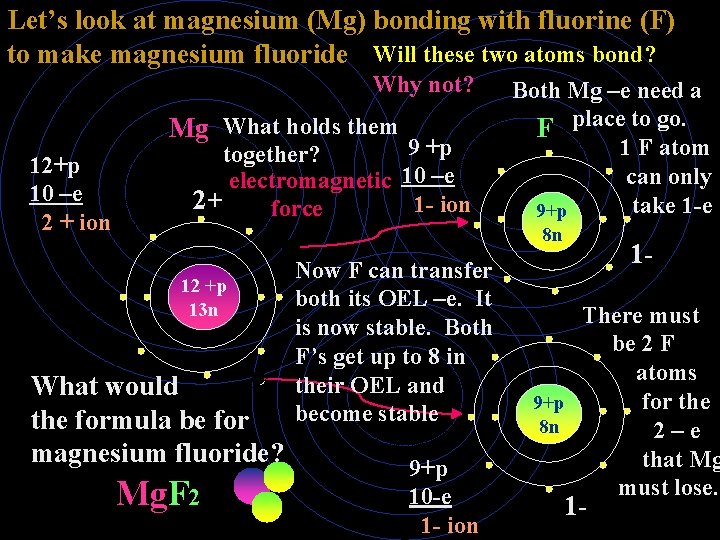
Let’s look at magnesium (Mg) bonding with fluorine (F) to make magnesium fluoride Will these two atoms bond? Why not? Mg What holds them 12+p 10 –e 2 + ion 9 +p together? electromagnetic 10 –e 2+ 1 - ion force Both Mg –e need a F place to go. 1 F atom can only take 1 -e 9+p 8 n 12 +p 13 n What would the formula be for magnesium fluoride? Mg. F 2 Now F can transfer both its OEL –e. It is now stable. Both F’s get up to 8 in their OEL and become stable 9+p 10 -e 1 - ion 1 - There must be 2 F atoms 9+p for the 8 n 2–e that Mg must lose. 1 -
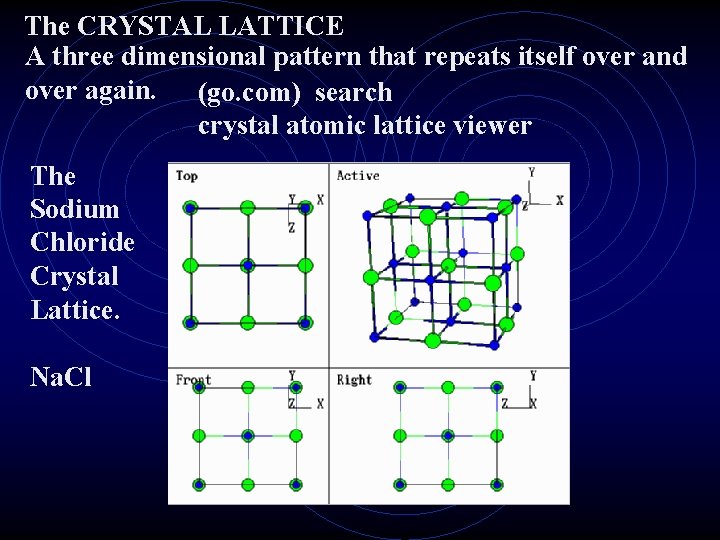
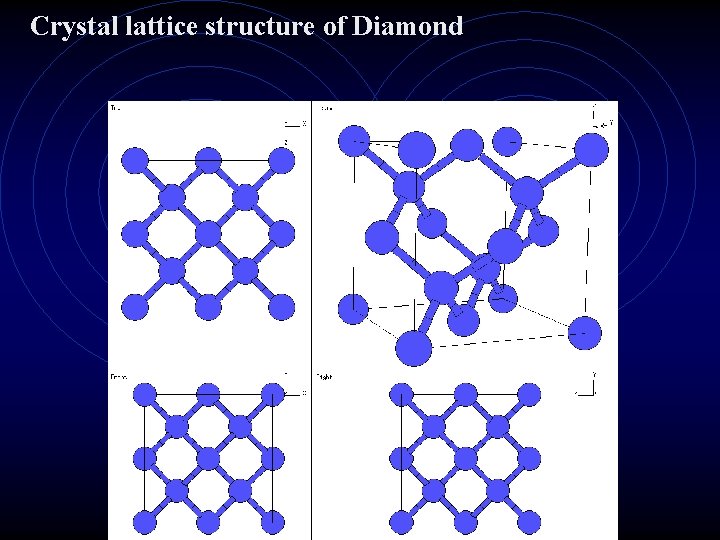
The CRYSTAL LATTICE A three dimensional pattern that repeats itself over and over again. (go. com) search crystal atomic lattice viewer The Sodium Chloride Crystal Lattice. Na. Cl
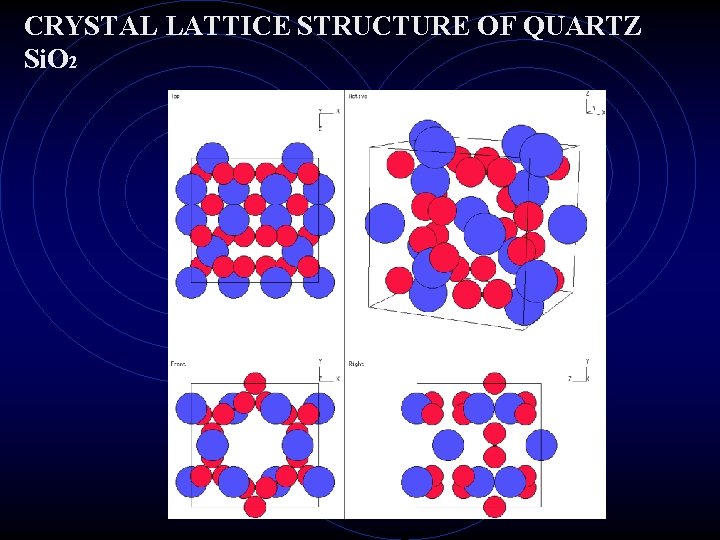
CRYSTAL LATTICE STRUCTURE OF QUARTZ Si. O 2
Crystal lattice structure of Diamond

Just checking to see how you are doing. Get ready for the hard stuff.

IONS Ion is a charged atom. Remember, if an atom loses –e It becomes a + ion, if it gains –e it becomes a – ion. Metals tend to lose electrons and form + ions Non metals tend to gain electrons and form – ions. Ionic bonds are formed between metals and nonmetals. Electromagnetic force (+/- attraction) holds two ions together in a molecule of an ionic compound. Crystals are formed by ionic compounds. The shape of the crystal is determined by how the ions arrange themselves in the crystal lattice.

Covalent Bonds Unlike ionic bonds, atoms involved in covalent bonding do not transfer their OEL –e to other atoms to form a chemical bond. What does the prefix “co-” mean? Most colleges are “coeducational”, what does that mean? That both males and females go there, or more than one gender go to school there. So, “co-” means more than one. “valent” refers to the OEL –e or valence electrons. (those located in the outer energy level)

In a covalent bond, -e in the OEL of atoms are SHARED between two atoms. The simplest covalent bonds happen between atoms That appear in nature as diatomic molecules. Diatomic molecules are two atoms of the same element that are Bonded together. IMPORTANT INFORMATION THAT YOU WILL NEED TO KNOW FOR THE REMAINDER OF THIS CLASS These elements always appear in their pure form as diatomic molecules: H 2 - hydrogen, O 2 - oxygen, N 2 - nitrogen, F 2 -fluorine, Cl 2 - chlorine, Br 2 -bromine, I 2 -iodine, At 2 - astatine.
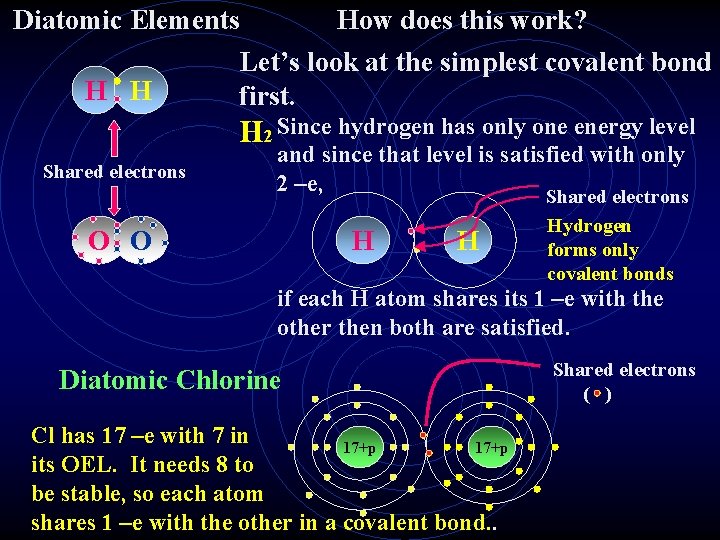
Diatomic Elements H H How does this work? Let’s look at the simplest covalent bond first. H 2 Since hydrogen has only one energy level Shared electrons and since that level is satisfied with only 2 –e, O O H H Shared electrons Hydrogen forms only covalent bonds if each H atom shares its 1 –e with the other then both are satisfied. Diatomic Chlorine Cl has 17 –e with 7 in 17+p its OEL. It needs 8 to be stable, so each atom shares 1 –e with the other in a covalent bond. . Shared electrons ( )

The Metallic Bond How are the atoms held together in a piece of metal such as a copper wire? Metal atoms have a tendency to lose their OEL –e’s. Remember the –e are the only particles that can move so they are free to travel anywhere in the piece of metal. n -e -e -e n -e -e n -e -e -e - e -en -e -e -e -e n n -e -e -e n-e -e n -e -e -e -e n n n -e -e When metals form a “metallic crystal” they simply dump their valence –e into a “sea of electrons”to be shared by all the nuclei of all the atoms in the metal n

Bonds and Properties The type of bond formed between atoms of different elements determine many of the properties of the compound formed. IONIC BONDS are the strongest bonds because of electromagnetic force. This bond accounts for: high melting point hardness brittleness shape (crystals) COVALENT BONDS are the weakest bonds. low melting points dull finish METALLIC BONDS ductility conductivity malleability luster

Predicting Types of Bonds By type of element: Metal + Nonmetal Ionic Bond Nonmetal + Nonmetal Covalent Bond Metal + Metallic Bond By location on PT Left + Right Ionic Bond Right + Right Covalent Bond Left + Left Metallic Bond By lose/gain –e Lose + Gain Lose + Lose Ionic Bond Covalent Bond Metallic Bond All of this is important

REVIEW Chemical bonding – the attractive force that holds atoms together when they combine to form a compound. Only –e are involved in forming chemical bonds. Why? Ion – a charged atom How is a + ion formed, a – ion ? Metals form +ions when they lose –e’s. Nonmetals form – ions when they gain –e’s. Electromagnetic force Ionic bonds are held together by _________. Ionic bonds are the strongest bonds. Atoms become “stable” (satisfied) when they have ____ -e’s in their OEL. transferred Ionic bonds are formed when –e’s are ______ from one atom to another. 8

share COVALENT BONDS are formed when atoms ____ -e’s. Covalent bonds are the weakest type of bond. Diatomic molecules are two atoms of the same element bonded together by a covalent bond. Diatomic elements are: H 2, O 2, N 2, F 2, Cl 2, I 2, At 2 The simplest covalent bond is the hydrogen bond in a diatomic molecule of hydrogen - H 2. Why? dump Metallic bonds are formed when metal atoms _______ their valence –e’s into a “sea of electrons”. The type of bond accounts for properties. IONIC – high melting points, crystals, hardness, brittleness, luster COVALENT - low m. p. , dullness METALLIC – malleability, ductility, conductivity, luster

QUIZ A HARD ONE!! 1. An atom is stable when it has ______ -e in its OEL. 2. When –e are transferred to another atom, a(n) ______ bond is formed. 1. If a metal atom loses –e it forms a _______ion. 2. When two atoms of the same element are bonded together you have a _______ molecule. (ex: H 2) 1. In covalent bonds, -e are _______ between two atoms. 2. The simplest covalent bond is between two atoms of_____. 3. In metallic bonds –e are _______. 4. True / False The first energy level can hold up to 8 –e’s. 5. Ionic bonds result in _______force holding the atoms together. 1. If a sulfur atom gains 2 –e’s, it will form a ______ ion. (use a number and a sign to answer 10)

The Answers 1. 8 2. Ionic 3. Positive 4. Diatomic 5. Shared 6. Hydrogen 7. Dumped 8. False 9. Electromagnetic 10. 2 -
- Slides: 25