Chemical Bonding and Molecular Structure Ch 10 Molecular
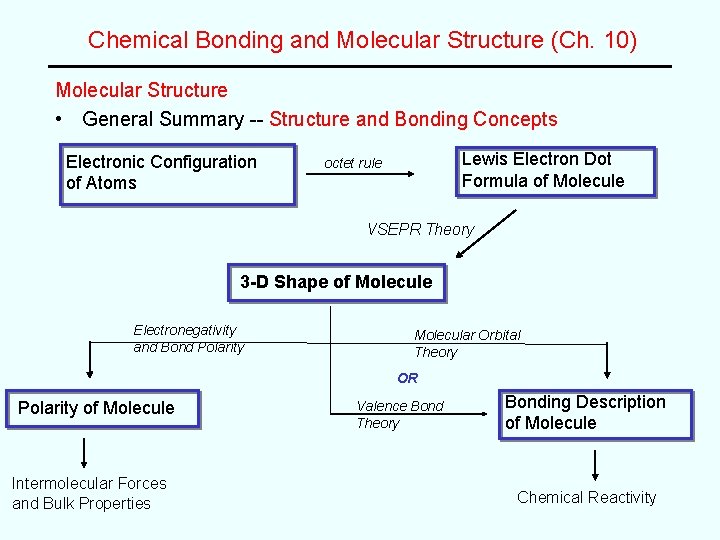
Chemical Bonding and Molecular Structure (Ch. 10) Molecular Structure • General Summary -- Structure and Bonding Concepts Electronic Configuration of Atoms Lewis Electron Dot Formula of Molecule octet rule VSEPR Theory 3 -D Shape of Molecule Electronegativity and Bond Polarity Molecular Orbital Theory OR Polarity of Molecule Intermolecular Forces and Bulk Properties Valence Bond Theory Bonding Description of Molecule Chemical Reactivity
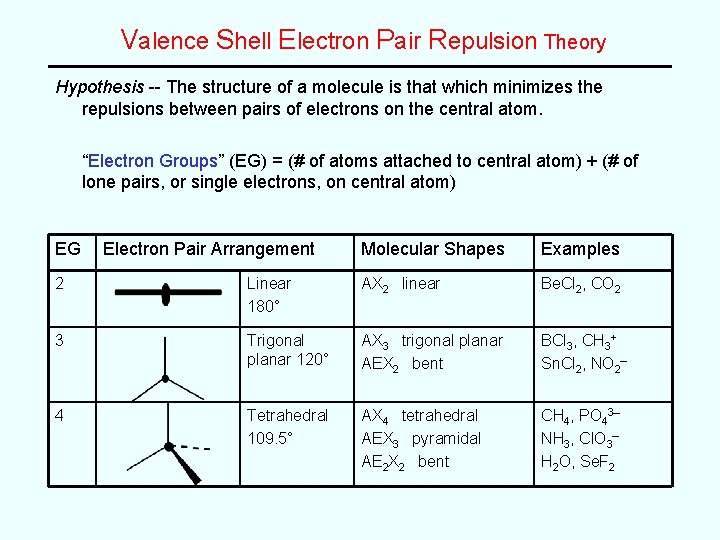
Valence Shell Electron Pair Repulsion Theory Hypothesis -- The structure of a molecule is that which minimizes the repulsions between pairs of electrons on the central atom. “Electron Groups” (EG) = (# of atoms attached to central atom) + (# of lone pairs, or single electrons, on central atom) EG Electron Pair Arrangement Molecular Shapes Examples 2 Linear 180° AX 2 linear Be. Cl 2, CO 2 3 Trigonal planar 120° AX 3 trigonal planar AEX 2 bent BCl 3, CH 3+ Sn. Cl 2, NO 2– 4 Tetrahedral 109. 5° AX 4 tetrahedral AEX 3 pyramidal AE 2 X 2 bent CH 4, PO 43– NH 3, Cl. O 3– H 2 O, Se. F 2

Valence Shell Electron Pair Repulsion Theory (cont. ) EG Electron Pair Arrangement Molecular Shapes Examples PF 5, Se. Cl 5+ SF 4, Br. F 4+ Cl. F 3, Xe. O 32– Xe. F 2, ICl 2– 5 Trigonal bypyramidal 120º & 90º AX 5 AEX 4 AE 2 X 3 AE 3 X 2 trig bypyramid “see saw” T-shaped linear 6 Octahedral 90º AX 6 octahedral AEX 5 square pyramidal AE 2 X 4 square planar SF 6, PCl 6– Br. F 5, SF 5– Xe. F 4, IF 4– (A = central atom, X = terminal atom, E = lone pair) Related Aspects: -- In trigonal bipyramid structures, lone e- pairs adopt equatorial positions (e) -- Order of repulsions: Lp-Lp > Lp - Bp > Bp - Bp (predicts distortions from ideal geometries)

Sample Problems • Use VSEPR Theory to predict (and draw) 3 -D structures of: NH 3 SF 4 Br. F 4–
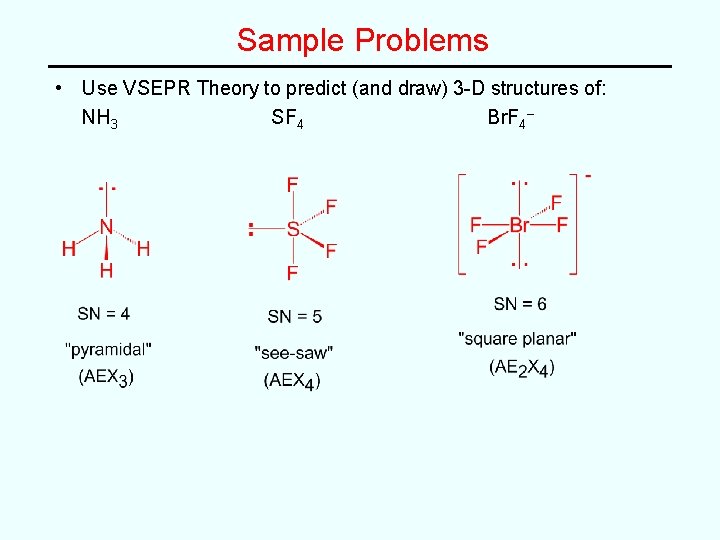
Sample Problems • Use VSEPR Theory to predict (and draw) 3 -D structures of: NH 3 SF 4 Br. F 4–
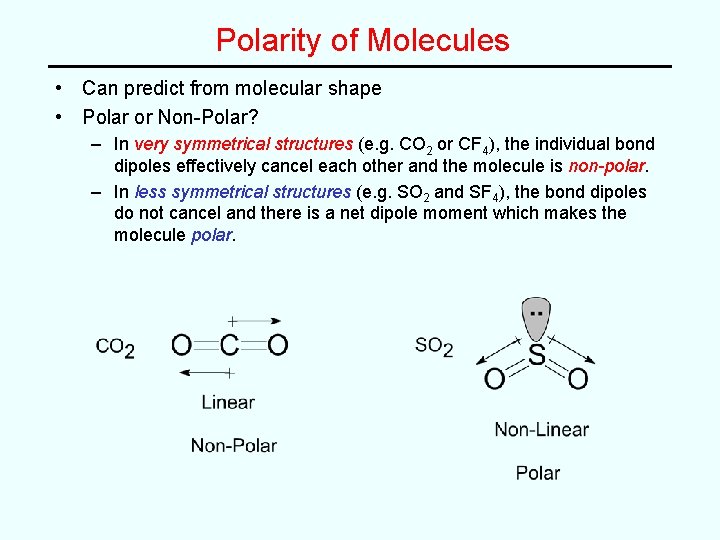
Polarity of Molecules • Can predict from molecular shape • Polar or Non-Polar? – In very symmetrical structures (e. g. CO 2 or CF 4), the individual bond dipoles effectively cancel each other and the molecule is non-polar. – In less symmetrical structures (e. g. SO 2 and SF 4), the bond dipoles do not cancel and there is a net dipole moment which makes the molecule polar.
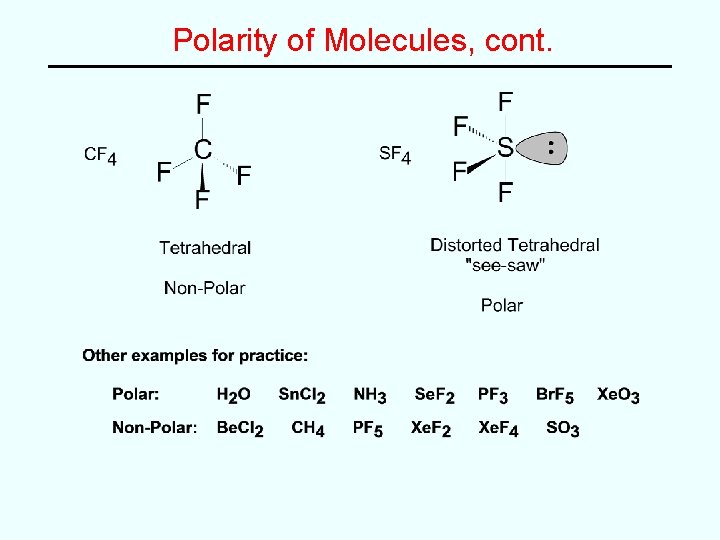
Polarity of Molecules, cont.
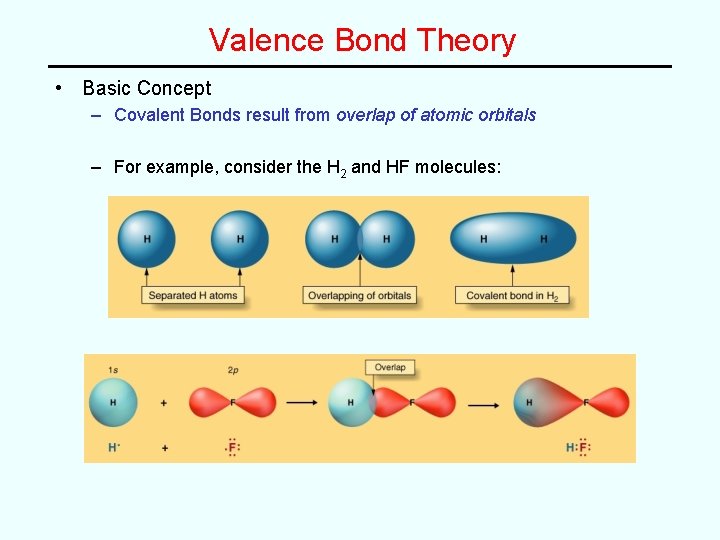
Valence Bond Theory • Basic Concept – Covalent Bonds result from overlap of atomic orbitals – For example, consider the H 2 and HF molecules:
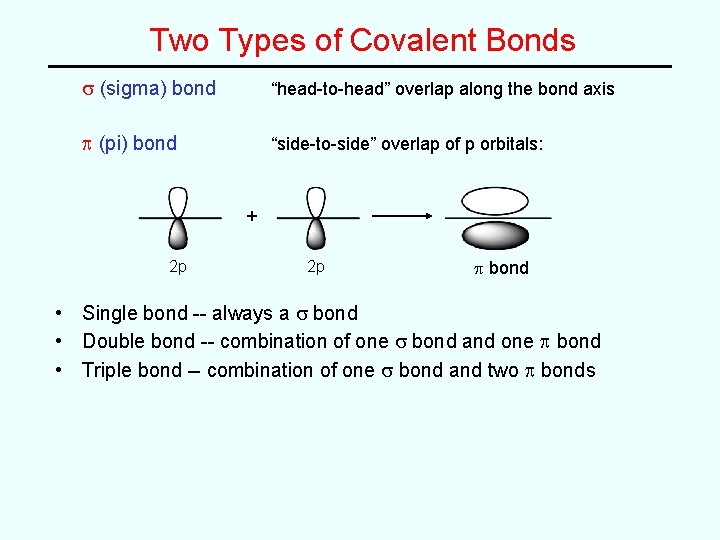
Two Types of Covalent Bonds s (sigma) bond “head-to-head” overlap along the bond axis p (pi) bond “side-to-side” overlap of p orbitals: + 2 p 2 p p bond • Single bond -- always a s bond • Double bond -- combination of one s bond and one p bond • Triple bond -- combination of one s bond and two p bonds
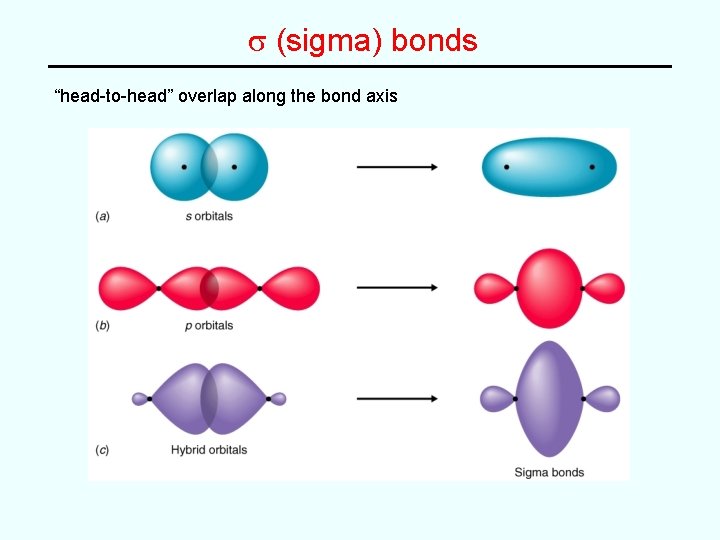
s (sigma) bonds “head-to-head” overlap along the bond axis
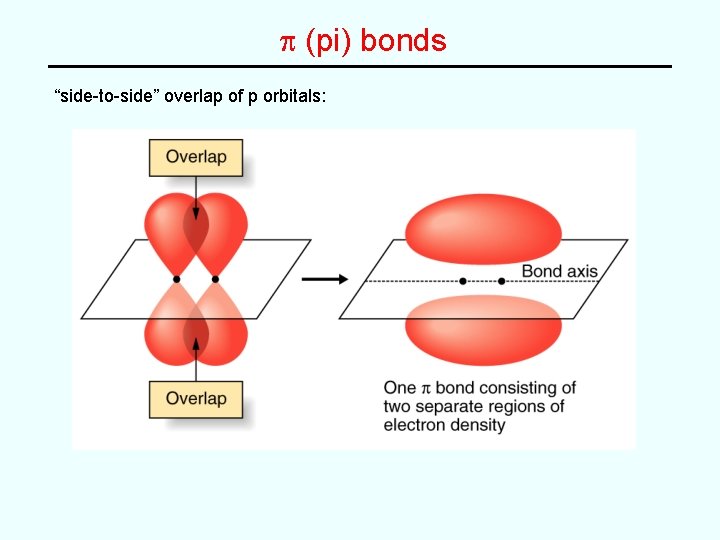
p (pi) bonds “side-to-side” overlap of p orbitals:
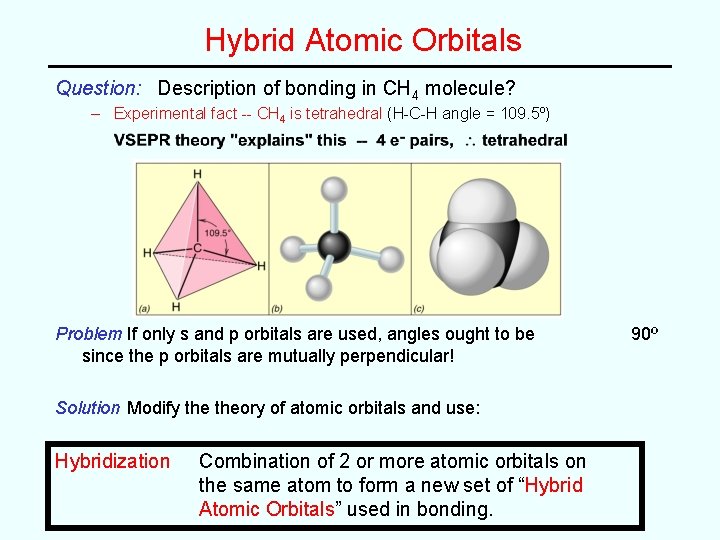
Hybrid Atomic Orbitals Question: Description of bonding in CH 4 molecule? – Experimental fact -- CH 4 is tetrahedral (H-C-H angle = 109. 5º) Problem If only s and p orbitals are used, angles ought to be since the p orbitals are mutually perpendicular! Solution Modify theory of atomic orbitals and use: Hybridization Combination of 2 or more atomic orbitals on the same atom to form a new set of “Hybrid Atomic Orbitals” used in bonding. 90º

Types of Hybrid Orbitals Atomic Orbitals Hybrid Orbitals Geometry Unhybridized p Orbitals one s + one p two sp EG = 2 Linear (180º) 2 one s + two p three sp 2 EG = 3 Trigonal planar (120º) 1 one s + three p four sp 3 EG = 4 Tetrahedral (109. 5º) 0 {Note: combination of n AO’s yields n Hybrid Orbitals) Example: in CH 4, C is sp 3 hybridized: C ground state - valence shell orbital diagram (predicts 90º angles -- wrong!) C hybridized state (predicts 109. 5º angles -- right!)

Examples • Use valence bond theory to describe the bonding in the following (use clear 3 -D pictures showing orbital overlap, etc) H 2 O NH 3 CH 4 PF 3 --simple s bonds and lone pairs H 2 CNH --double bond like H 2 CCH 2 ethene and H 2 CO formaldehyde) HCN --triple bond like HCCH ethyne and N 2 nitrogen)
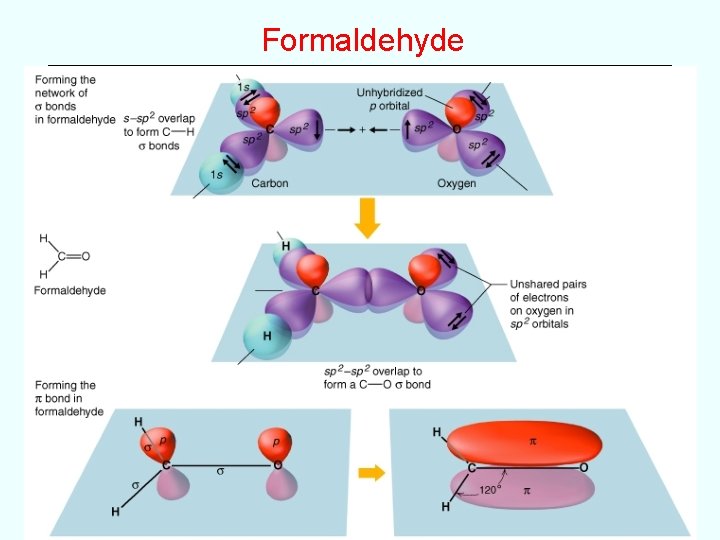
Formaldehyde
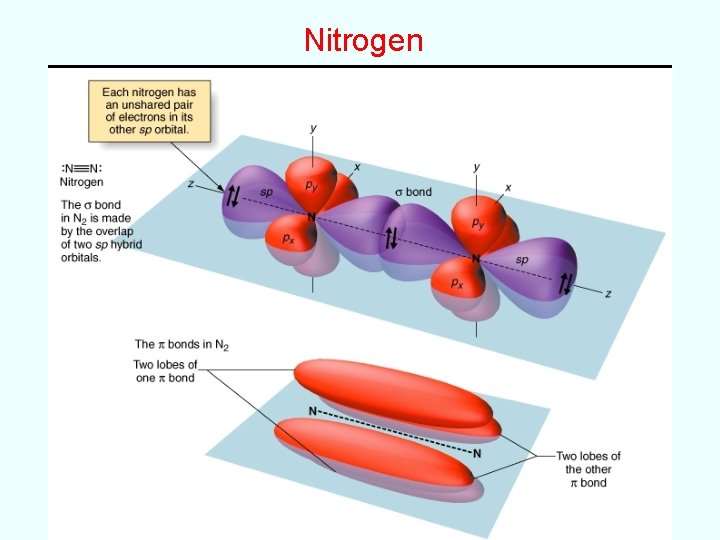
Nitrogen

Comparison of VB and MO Theory • Valence Bond Theory (“simple” but somewhat limited) – e– pair bonds between two atoms using overlap of atomic orbitals on two atoms • Molecular Orbital Theory (more general but “complex”) – All e–’s in molecule fill up a set of molecular orbitals that are made up of linear combinations of atomic orbitals on two or more atoms MO’s can be: “localized” -combination of AO’s on two atoms, as in the diatomic molecules “delocalized” -combination of AO’s on three or more atoms as in benzene (C 6 H 6)
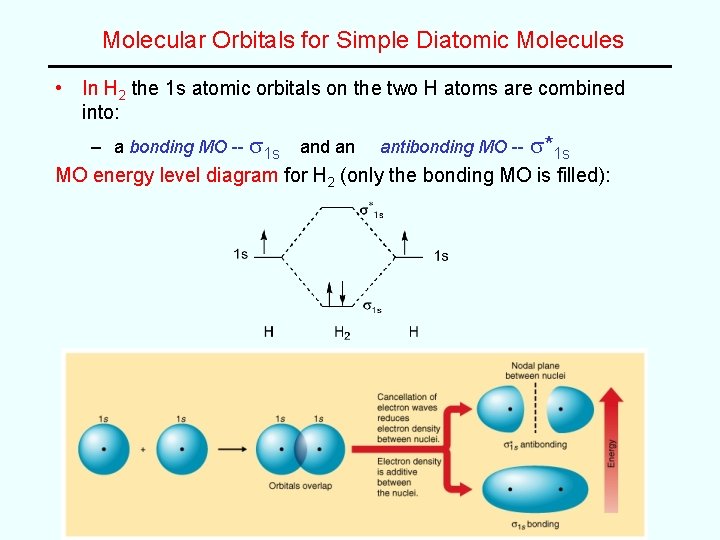
Molecular Orbitals for Simple Diatomic Molecules • In H 2 the 1 s atomic orbitals on the two H atoms are combined into: – a bonding MO -- s 1 s and an antibonding MO -- s*1 s MO energy level diagram for H 2 (only the bonding MO is filled):
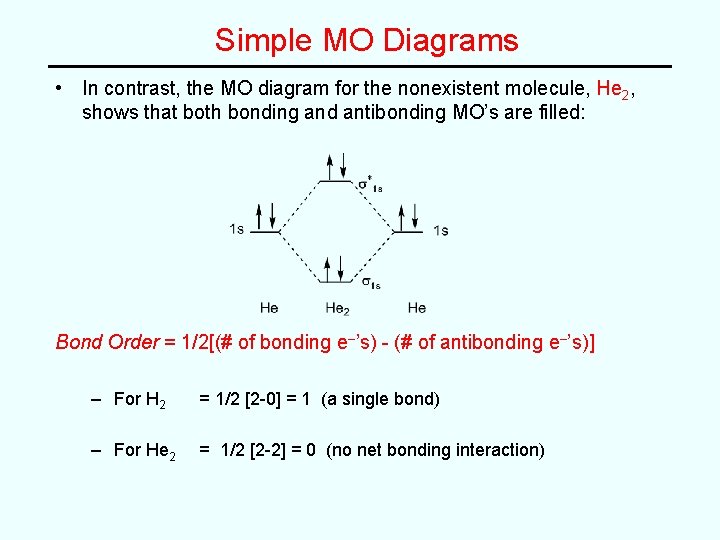
Simple MO Diagrams • In contrast, the MO diagram for the nonexistent molecule, He 2, shows that both bonding and antibonding MO’s are filled: Bond Order = 1/2[(# of bonding e–’s) - (# of antibonding e–’s)] – For H 2 = 1/2 [2 -0] = 1 (a single bond) – For He 2 = 1/2 [2 -2] = 0 (no net bonding interaction)
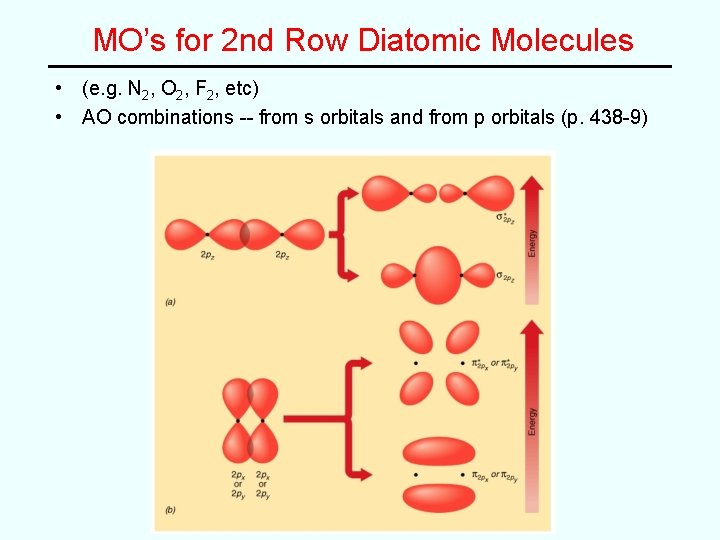
MO’s for 2 nd Row Diatomic Molecules • (e. g. N 2, O 2, F 2, etc) • AO combinations -- from s orbitals and from p orbitals (p. 438 -9)
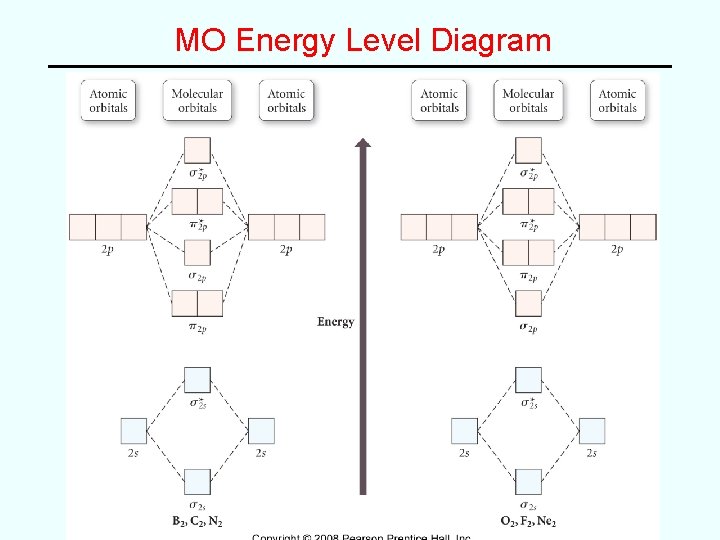
MO Energy Level Diagram

Examples • e. g. Fill in MO diagram for C 2, N 2, O 2, F 2 and Ne 2 and determine bond order for each: Molecule C 2 N 2 O 2 F 2 Ne 2 Bond order 2 2 0 3 1 • General “rules” – Electrons fill the lowest energy orbitals that are available – Maxiumum of 2 electrons, spins paired, per orbital – Hund’s rule of maximum unpaired spins applies* *(accounts for paramagnetism of O 2 (VB theory fails here!)
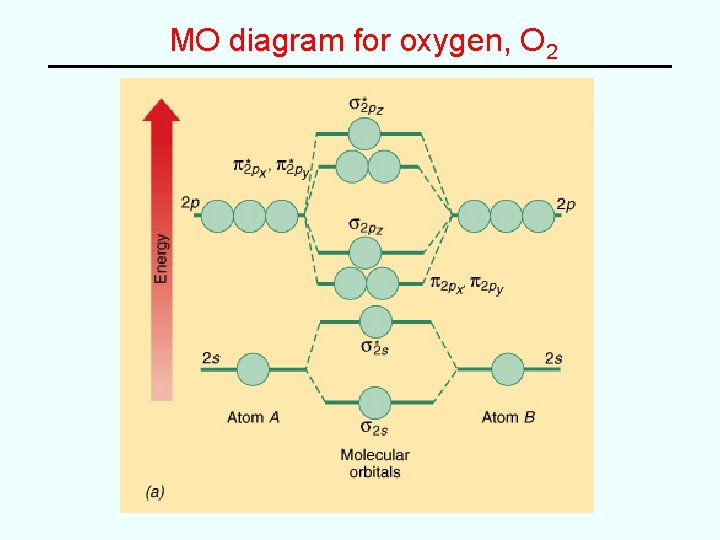
MO diagram for oxygen, O 2
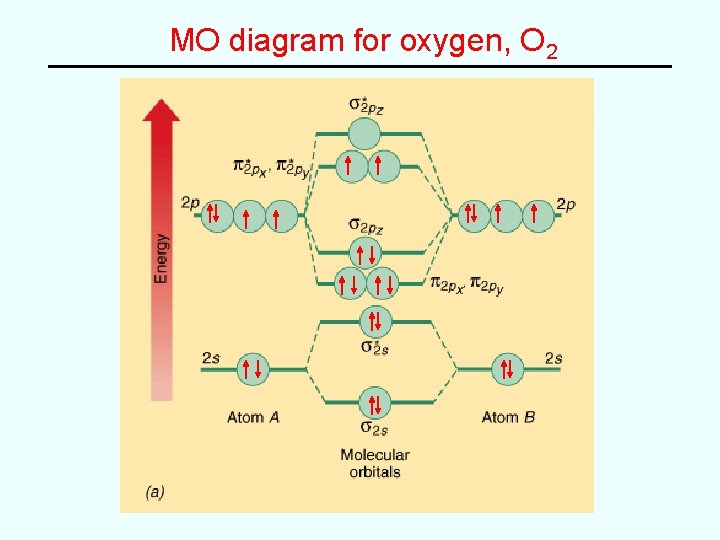
MO diagram for oxygen, O 2
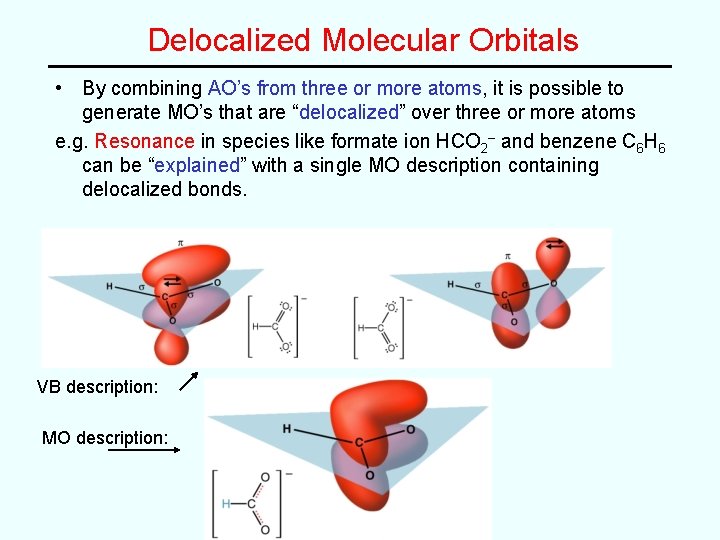
Delocalized Molecular Orbitals • By combining AO’s from three or more atoms, it is possible to generate MO’s that are “delocalized” over three or more atoms e. g. Resonance in species like formate ion HCO 2– and benzene C 6 H 6 can be “explained” with a single MO description containing delocalized bonds. VB description: MO description:
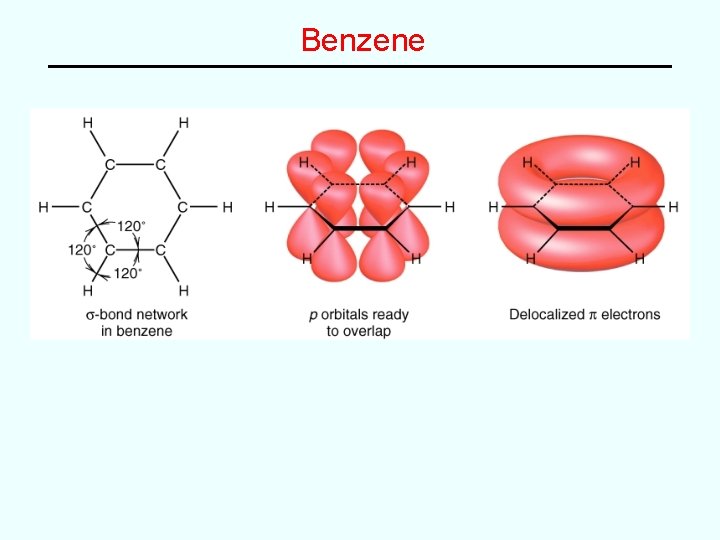
Benzene

Sample Problem • Fully describe the bonding in Na. HCO 3 using valence bond theory.

Sample Problem • Fully describe the bonding in Na. HCO 3 using valence bond theory. Answer: The Lewis structure is (and its resonance equivalent). VSEPR theory gives a trigonal planar structure at carbon (3 EG), and a bent structure at the central O (4 EG). At C, the expected hybridization is sp 2, with 120° bond angles. For the terminal O with a single bond, there will be one s bond between the p orbital (terminal O) and the sp 2 orbital (central C). For the C=O bond, there will be a similar s bond, but also one p bond from the side-on interaction of one p orbital on each atom. continued…

Sample Problem, cont. For the C-OH bond there will be one s bond between the O sp 3 orbital and the C sp 2 orbital. At the central oxygen there is sp 3 hybridization (EG = 4). Each lone pair lies in one sp 3 hybrid orbital. The O-H bond is a s bond between the O sp 3 hybrid and the H 1 s orbital.

Sample Problem • Write the MO diagram for HCl. Predict the bond order and sketch the bonding and antibonding MO’s. [note: H 1 s energy = -13 e. V, Cl 3 s energy = -25 e. V, Cl 3 p energy = -14 e. V]
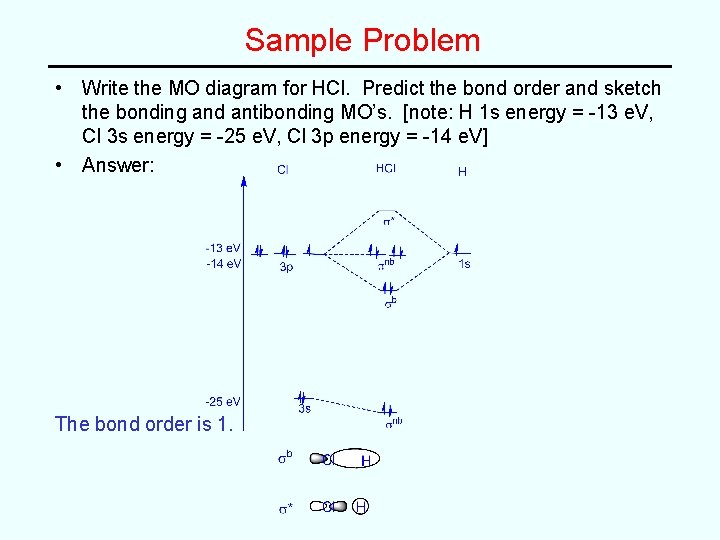
Sample Problem • Write the MO diagram for HCl. Predict the bond order and sketch the bonding and antibonding MO’s. [note: H 1 s energy = -13 e. V, Cl 3 s energy = -25 e. V, Cl 3 p energy = -14 e. V] • Answer: The bond order is 1.
- Slides: 31