Chemical Bonding and Extended Systems p EDA and

Chemical Bonding and Extended Systems - p. EDA and p. EDA-NOCV Marc Raupach, raupach@scm. com Vrije Universiteit Amsterdam, 20 April 2016, © SCM
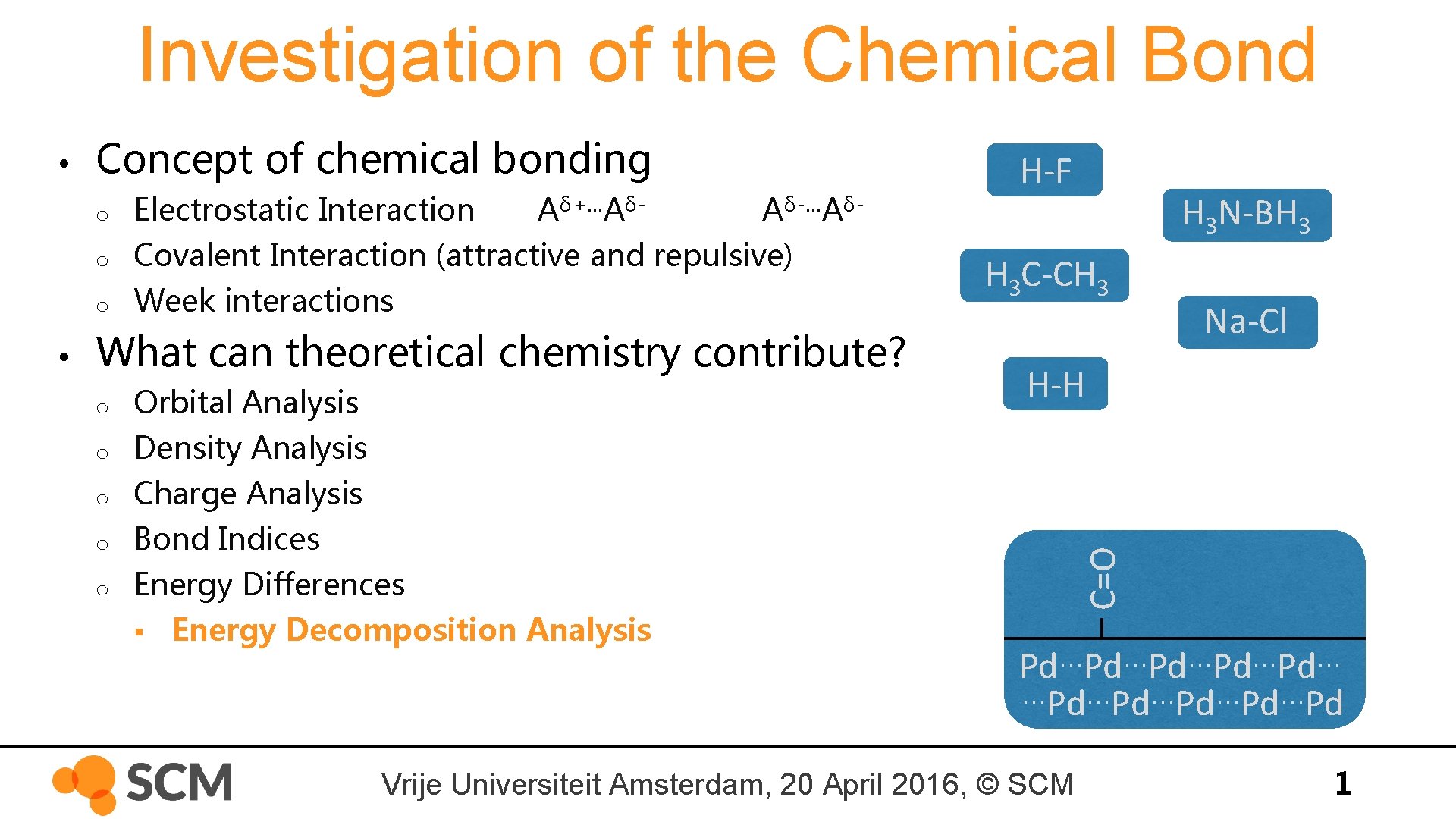
Investigation of the Chemical Bond Concept of chemical bonding o o o • Electrostatic Interaction Aδ+…AδAδ-…AδCovalent Interaction (attractive and repulsive) Week interactions What can theoretical chemistry contribute? o o o Orbital Analysis Density Analysis Charge Analysis Bond Indices Energy Differences § Energy Decomposition Analysis H-F H 3 N-BH 3 H 3 C-CH 3 Na-Cl H-H C=O • … … … Pd Pd Pd …Pd…Pd…Pd Vrije Universiteit Amsterdam, 20 April 2016, © SCM 1
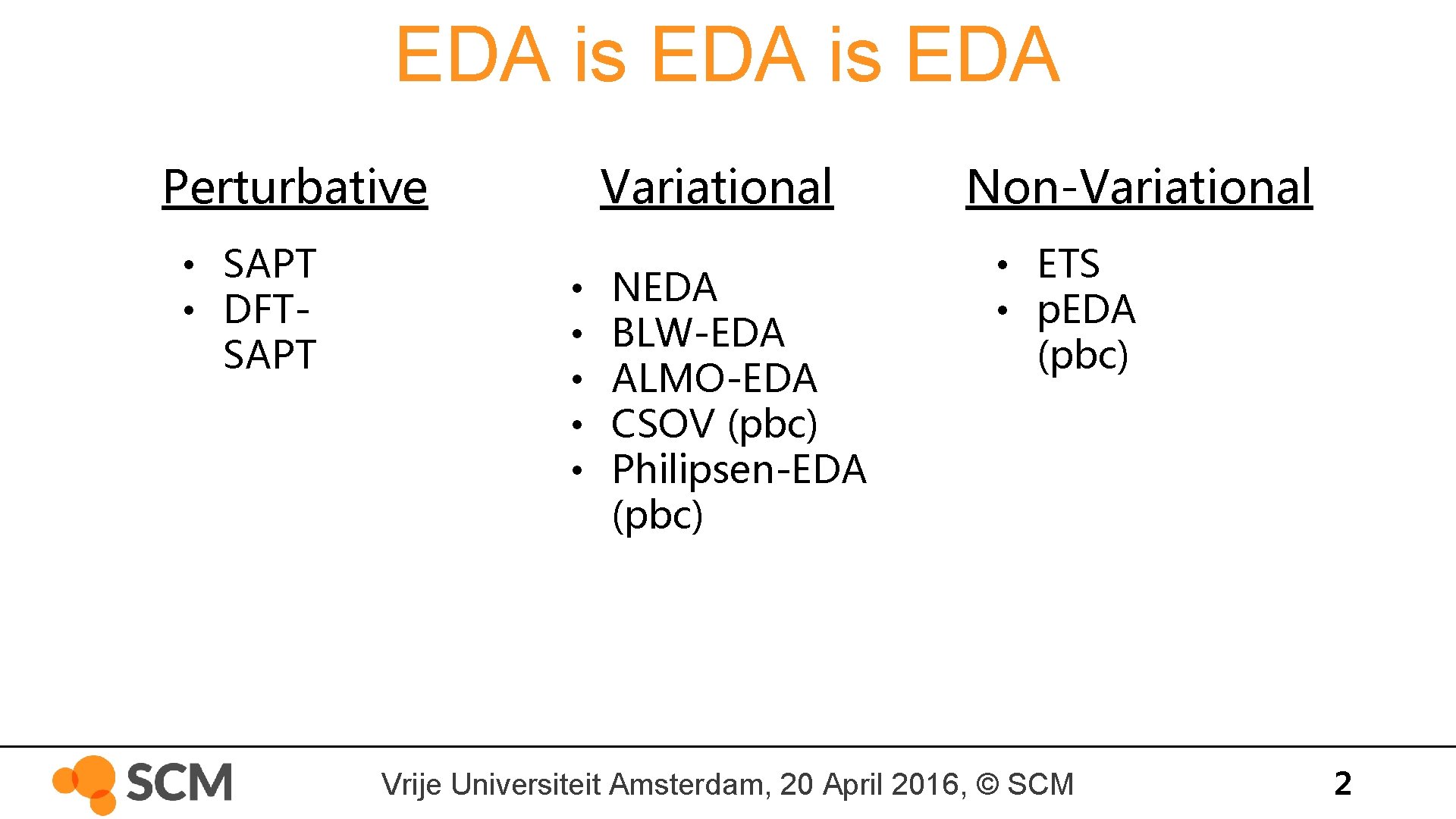
EDA is EDA Perturbative • SAPT • DFTSAPT Variational • • • NEDA BLW-EDA ALMO-EDA CSOV (pbc) Philipsen-EDA (pbc) Non-Variational • ETS • p. EDA (pbc) Vrije Universiteit Amsterdam, 20 April 2016, © SCM 2

History of EDA in ADF Extended Transition State (ETS) method by Ziegler and Rauk Natural Orbitals of Chemical Valency (NOCV) by Mitoraj and Michalak Combined ETS and NOCV method by Mitoraj, Ziegler and Michalak Ziegler and Rauk, Inorg. Chem. 18, 1558 (1979). Mitoraj, Michalak and Ziegler, JCTC 5, 962 (2009). Vrije Universiteit Amsterdam, 20 April 2016, © SCM 3

To Infinity and Beyond? • Real Space Reciprocal Space H -a H 0 H +a H Vrije Universiteit Amsterdam, 20 April 2016, © SCM 4

Periodic Energy Decomposition Analysis Vrije Universiteit Amsterdam, 20 April 2016, © SCM 5
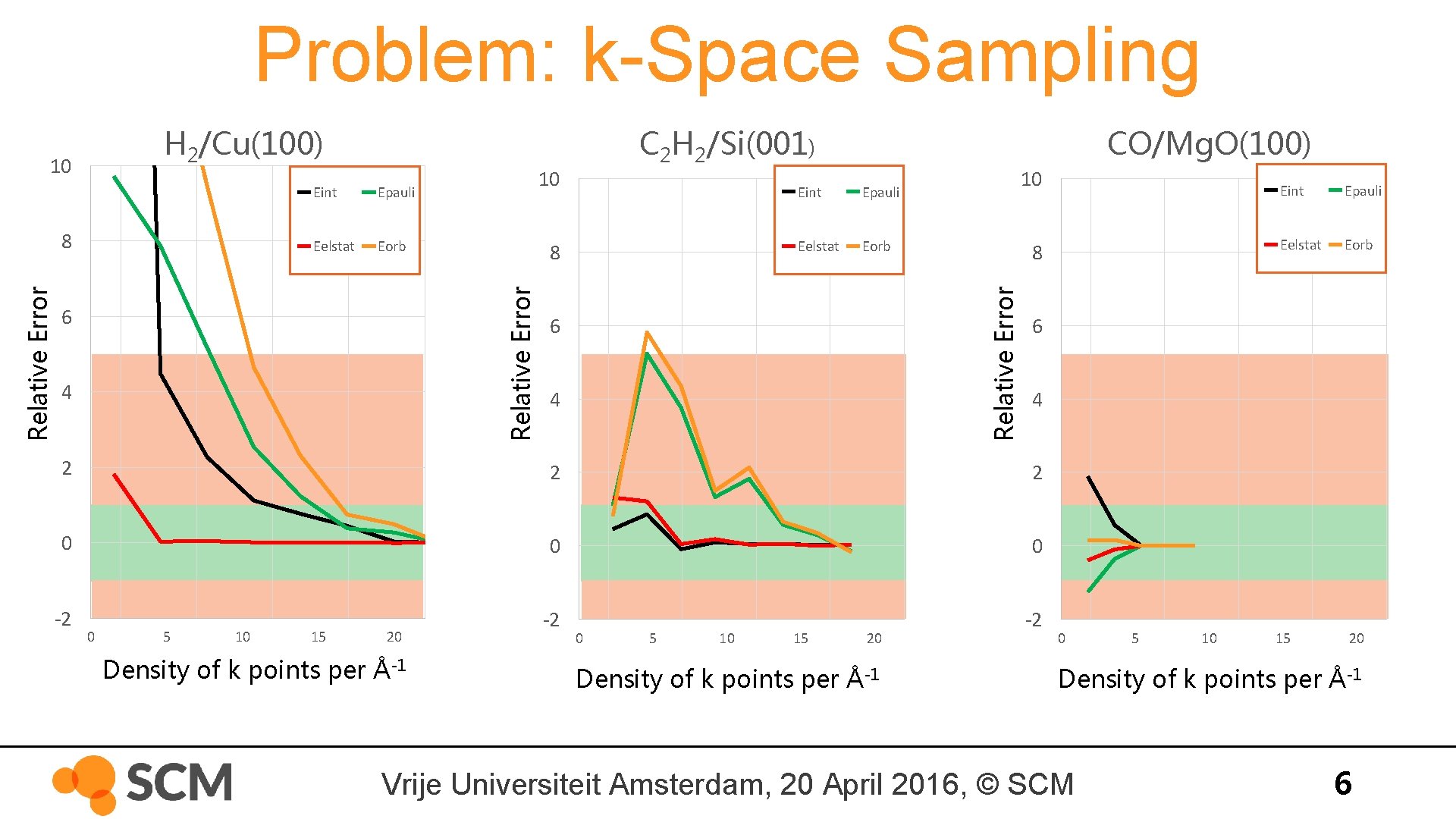
Problem: k-Space Sampling H 2/Cu(100) Epauli Eelstat Eorb 10 8 Relative Error 8 Eint 6 4 CO/Mg. O(100) Eint Epauli Eelstat Eorb 10 8 Relative Error 10 C 2 H 2/Si(001) 6 4 2 0 0 0 -2 -2 -2 10 15 20 Density of k points per Å-1 0 5 10 15 20 Density of k points per Å-1 Eelstat Eorb 4 2 5 Epauli 6 2 0 Eint 0 5 10 15 20 Density of k points per Å-1 Vrije Universiteit Amsterdam, 20 April 2016, © SCM 6
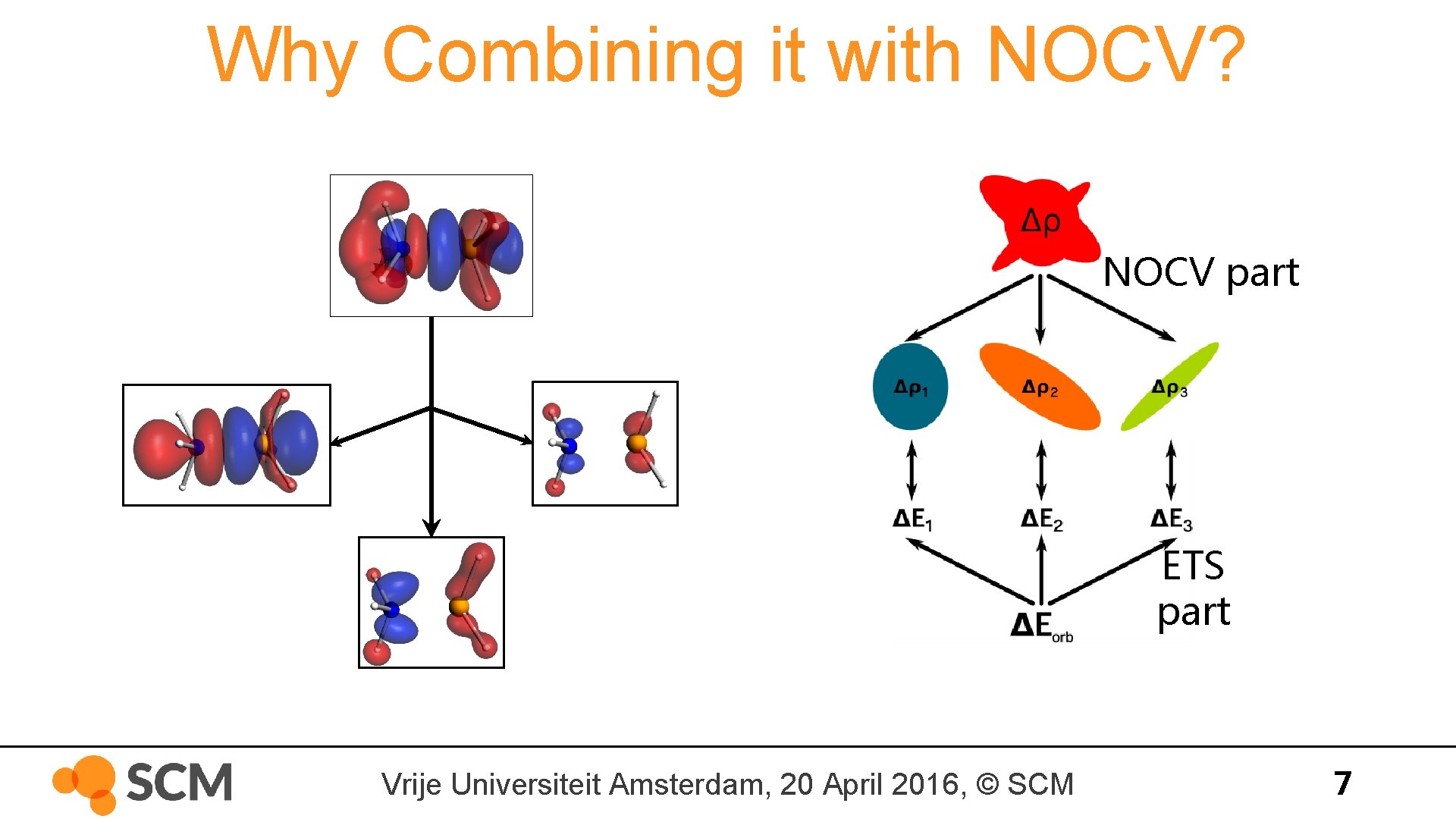
Why Combining it with NOCV? NOCV part ETS part Vrije Universiteit Amsterdam, 20 April 2016, © SCM 7

p. EDA-NOCV: Some equations I • Vrije Universiteit Amsterdam, 20 April 2016, © SCM 8

p. EDA-NOCV: Some equations II • Vrije Universiteit Amsterdam, 20 April 2016, © SCM 9
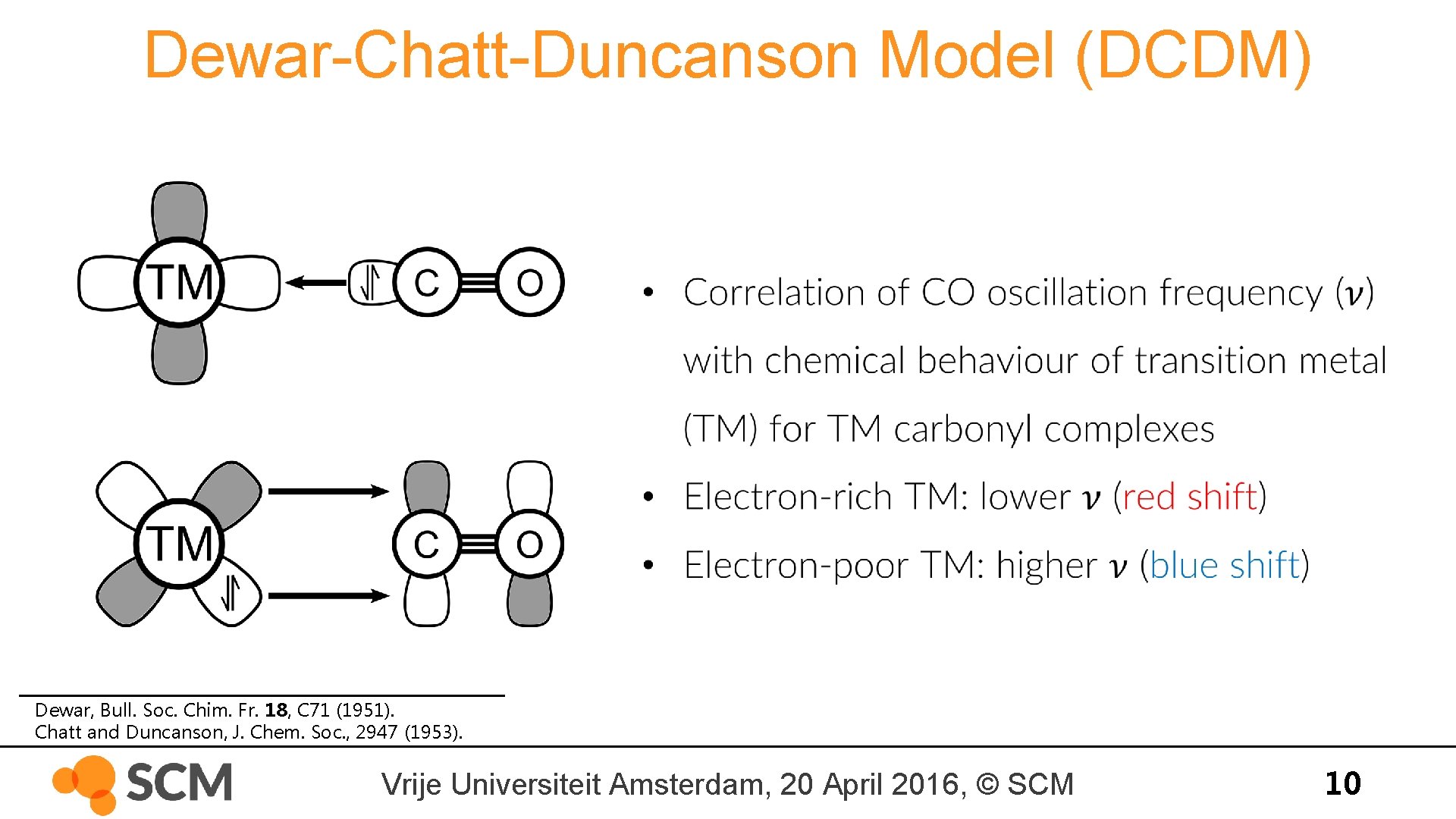
Dewar-Chatt-Duncanson Model (DCDM) Dewar, Bull. Soc. Chim. Fr. 18, C 71 (1951). Chatt and Duncanson, J. Chem. Soc. , 2947 (1953). Vrije Universiteit Amsterdam, 20 April 2016, © SCM 10
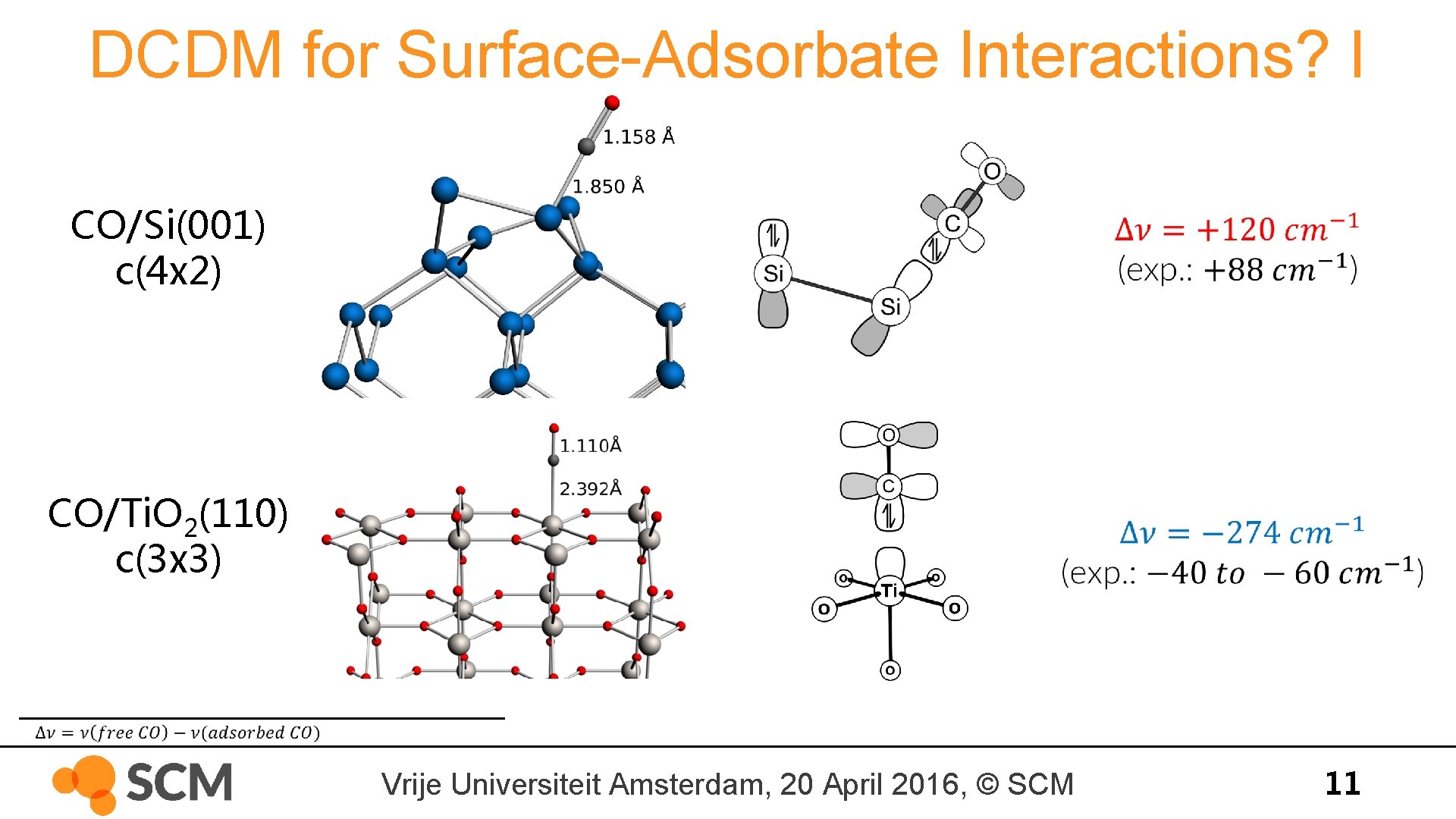
DCDM for Surface-Adsorbate Interactions? I CO/Si(001) c(4 x 2) CO/Ti. O 2(110) c(3 x 3) Vrije Universiteit Amsterdam, 20 April 2016, © SCM 11
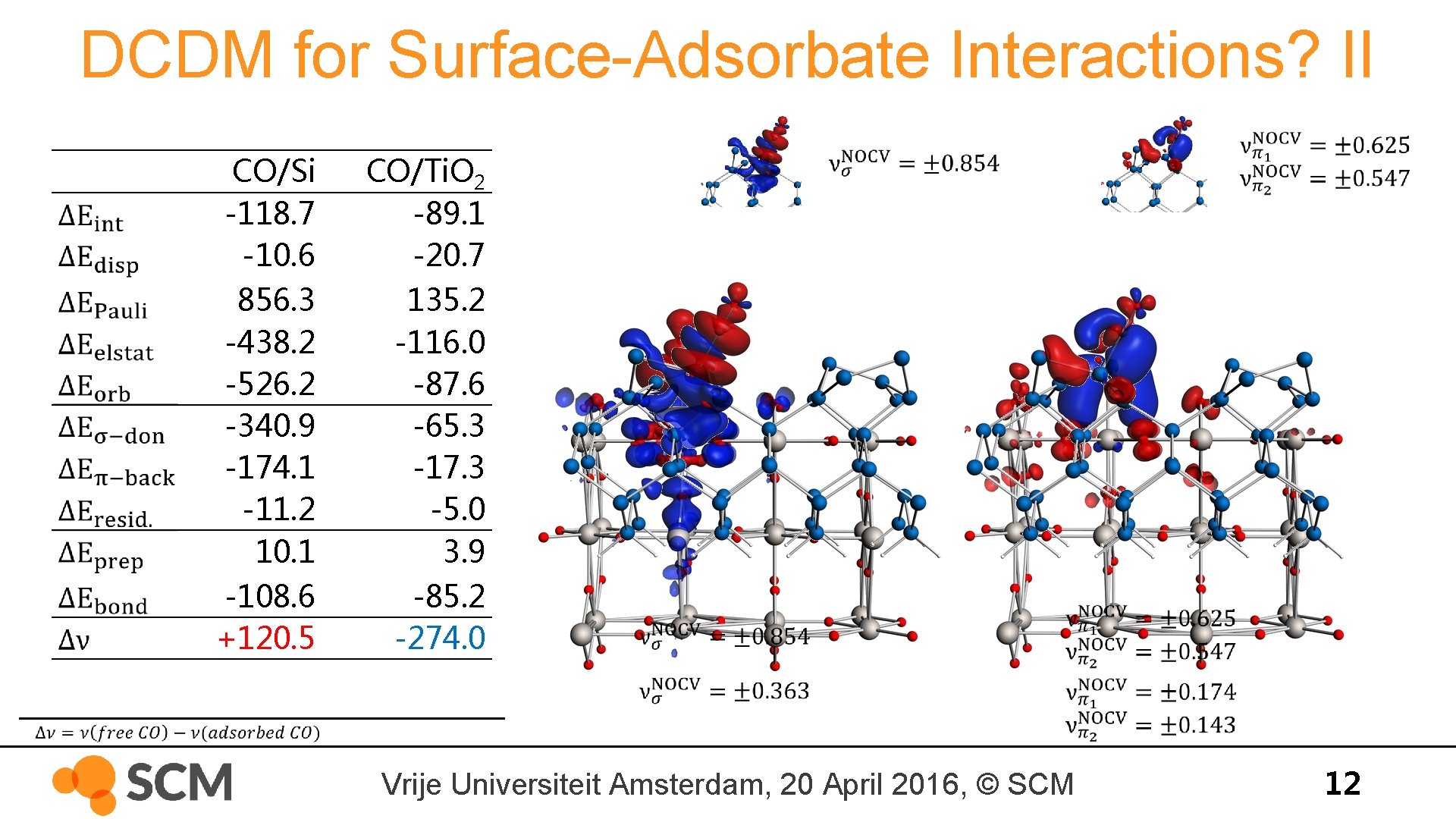
DCDM for Surface-Adsorbate Interactions? II CO/Si -118. 7 -10. 6 856. 3 -438. 2 -526. 2 -340. 9 -174. 1 -11. 2 10. 1 -108. 6 +120. 5 CO/Ti. O 2 -89. 1 -20. 7 135. 2 -116. 0 -87. 6 -65. 3 -17. 3 -5. 0 3. 9 -85. 2 -274. 0 Vrije Universiteit Amsterdam, 20 April 2016, © SCM 12
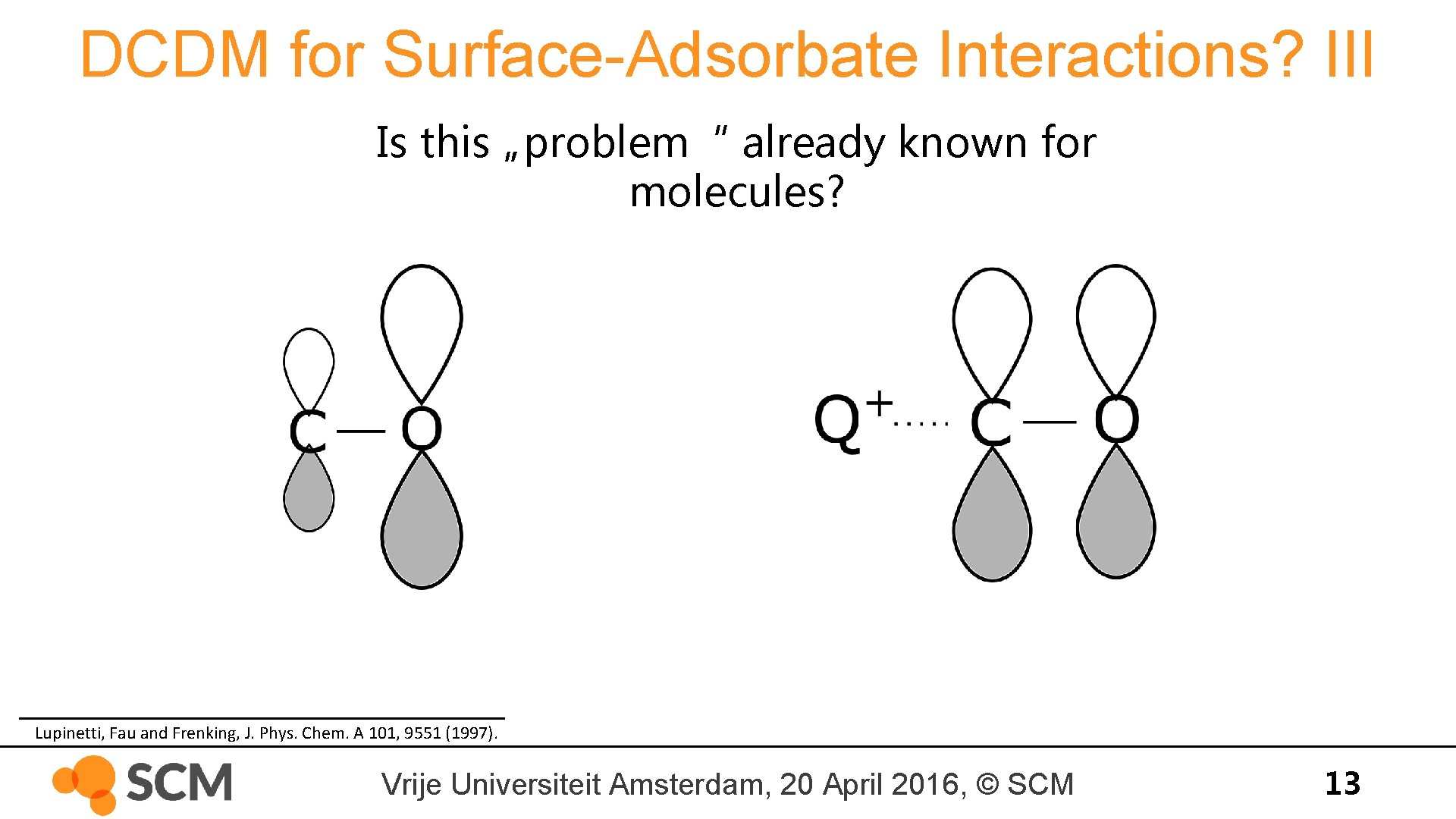
DCDM for Surface-Adsorbate Interactions? III Is this „problem“ already known for molecules? Lupinetti, Fau and Frenking, J. Phys. Chem. A 101, 9551 (1997). Vrije Universiteit Amsterdam, 20 April 2016, © SCM 13

Summary p. EDA: • Decomposition into „standard“ EDA terms for extended systems • For metals and non-metals • Usage of spin-resolved fragment electronic structure p. EDA-NOCV: • Detailed discussion of covalent interactions for extended systems • Γ-only → metallic materials not advisable Vrije Universiteit Amsterdam, 20 April 2016, © SCM 14
- Slides: 15