CHEMICAL BONDING 1 Why do elements form compounds

CHEMICAL BONDING 1

Why do elements form compounds? To achieve an octet (noble gas configuration/full outer shell) To be at their lowest energy To have the most stable arrangement of electrons 2

How do elements form compounds? Sharing Gaining Losing VALENCE ELECTRONS 3

Chemical Bond A mutual attraction between nuclei and valence electrons of different atoms that bind atoms together. 4

TYPES OF CHEMICAL BONDS IONIC - transfer of electrons COVALENT - sharing of electron pairs Nonpolar covalent - equal sharing Polar covalent - unequal sharing 5


Properties of Ionic Compounds 1. Crystalline solids (made of ions) 2. High melting and boiling points 3. Many are water soluble 4. Conduct electricity in molten state 5. Conduct electricity in aqueous solutions 7


Properties of Covalent Compounds (Molecules) 1. Gases, liquids, and solids (made of molecules) 2. Low melting and boiling points 3. Poor electrical conductivity in any phase 4. Many soluble in nonpolar liquids, not water 9
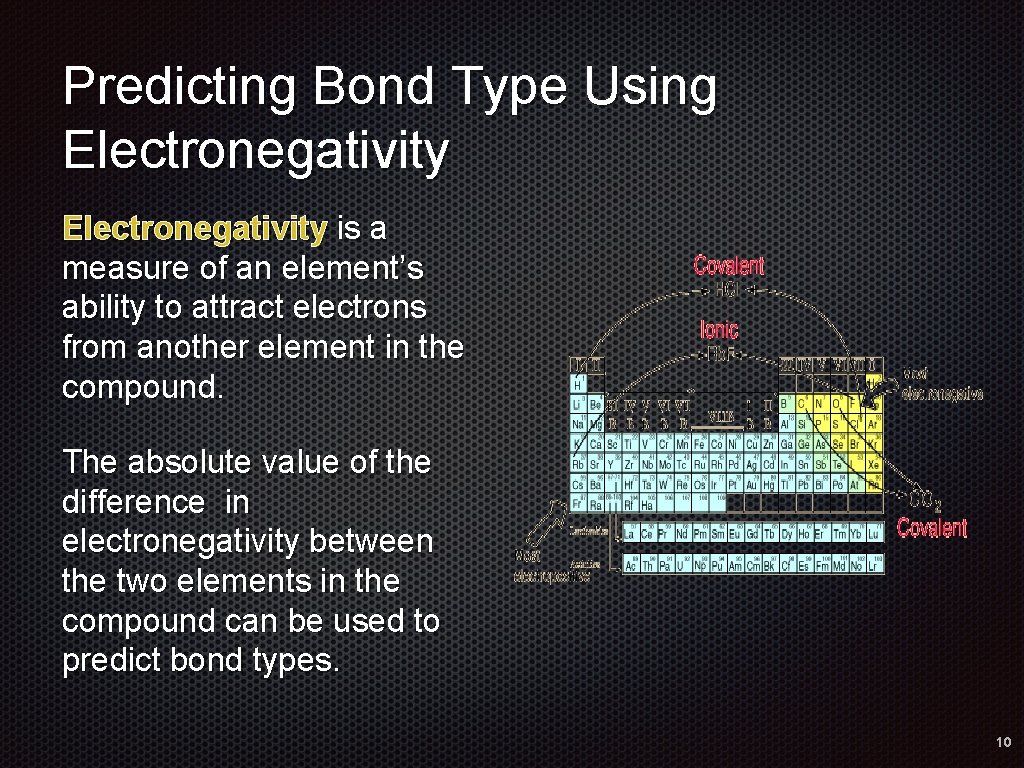
Predicting Bond Type Using Electronegativity is a measure of an element’s ability to attract electrons from another element in the compound. The absolute value of the difference in electronegativity between the two elements in the compound can be used to predict bond types. 10

Electronegativity Difference Bond Type 0 to 0. 4 Nonpolar covalent Greater than 0. 4 to 1. 7 Polar covalent Greater than 1. 7 Ionic 11
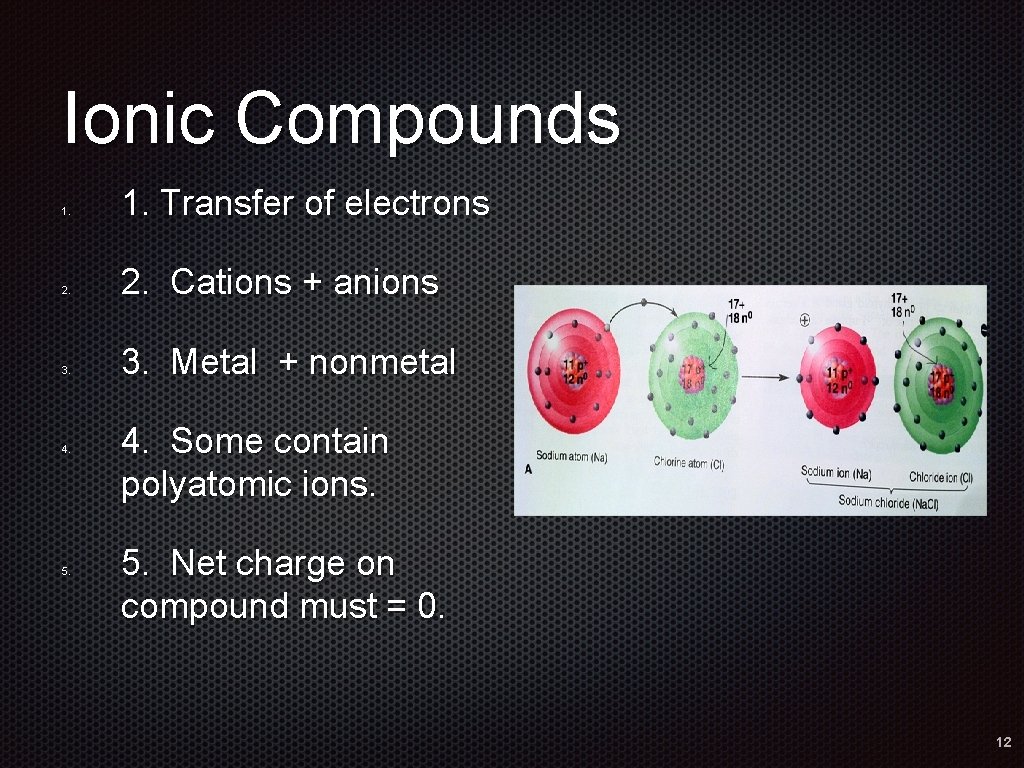
Ionic Compounds 1. 2. 3. 4. 5. 1. Transfer of electrons 2. Cations + anions 3. Metal + nonmetal 4. Some contain polyatomic ions. 5. Net charge on compound must = 0. 12

Binary Ionic Compounds 1. 2. 3. 1. Formed from monatomic cation + monatomic anion. 2. All end in “ide” 3. Group B metals use a Roman numeral with the exception of Silver, Zinc, and Cadmium. The Roman numeral indicates the charge on the cation.

Ternary Ionic Compounds 1 2 3 Made up of at two ions, with at least one ion being a polyatomic ion. Most end in ate or ite. Use parentheses if there is more than one of the same polyatomic ion.


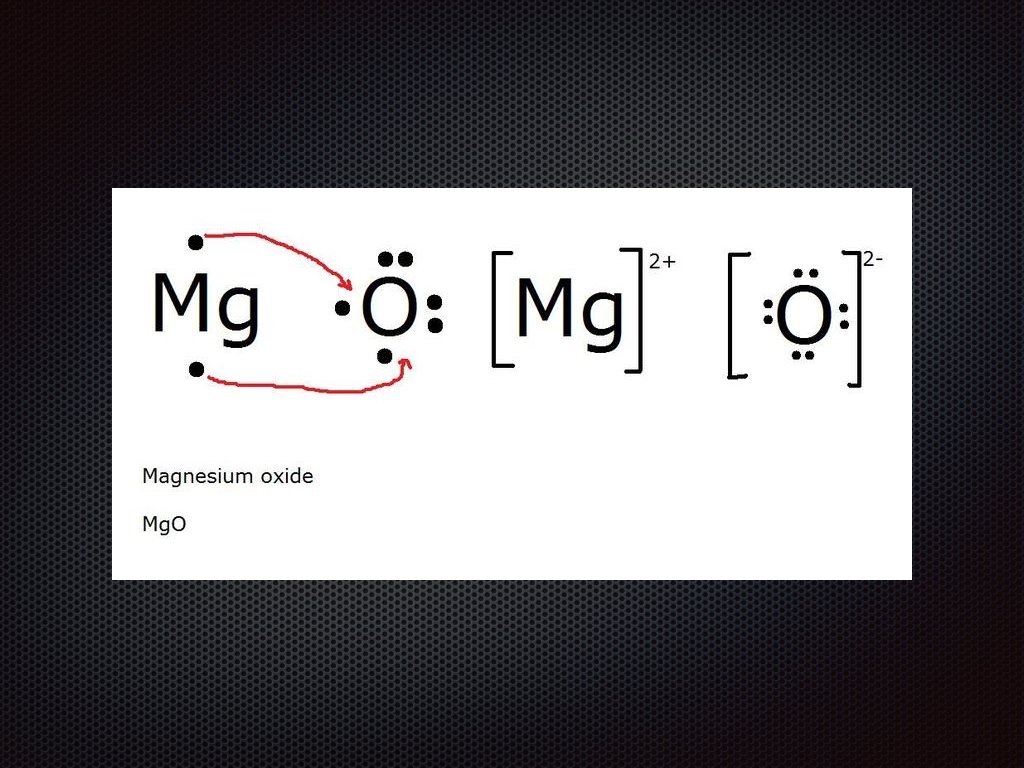
Lewis Dot Structures for Ionic Compounds

Covalent Compounds Also called molecular compounds or molecules. Composed of two or more nonmetals. Electrons are shared.

Covalent Compounds Also called molecular compounds or molecules. Composed of two or more nonmetals. Electrons are shared.

Binary Molecular Compounds All end in “ide” Use prefixes when naming compounds. Do not use parentheses in formulas. Names do not use Roman Numerals.

Prefixes Number Prefix 1 Mono 2 Di 3 Tri 4 Tetra 5 Penta 6 Hexa 7 Hepta 8 Octa 9 Nona 10 Deca
- Slides: 21