Chemical bond a force that holds atoms or


� Chemical bond – a force that holds atoms or ions together as a unit � Chemical formula – notation that tells what elements are in a compound and how many of each element there are

� Atoms like to be stable by having a full valence orbital �Stable atoms do not react �Unstable atoms react easily � Atoms gain or lose electrons based on their oxidation number to reach a full/empty shell � Stability can be seen using Lewis Dot Diagrams

� Ionic � Covalent � Metallic

� http: //tinyurl. com/jkm 2 v 2 n � Which type of elements easily lose electrons? Which easily gain electrons? � Why does the sodium atom attract the chlorine atom? � What structure is formed by multiple ion pairs sticking together?

� Bonding of two atoms through the transferring of electrons � Between a metal and a nonmetal � Transferring electrons forms two ions �Cation – positive charge – gives away electrons �Anion – negative charge – steals electrons
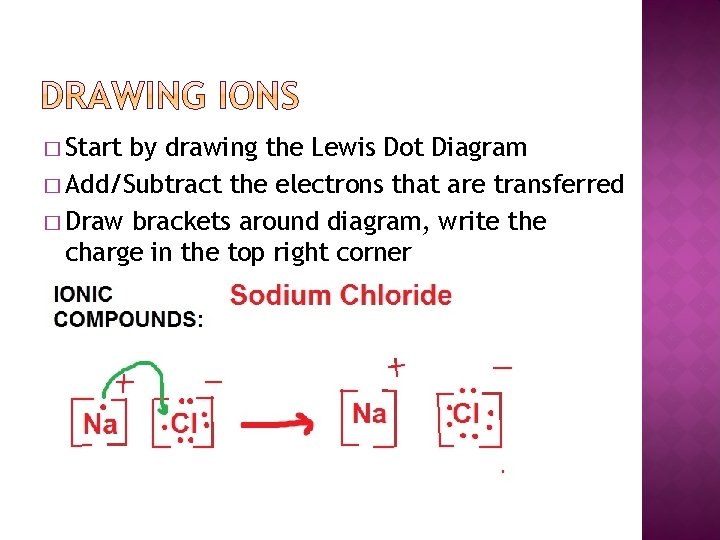
� Start by drawing the Lewis Dot Diagram � Add/Subtract the electrons that are transferred � Draw brackets around diagram, write the charge in the top right corner

� Practice: Draw the following ions �Sodium, lost 1 electron �Oxygen, gained two electrons �Magnesium, lost two electrons
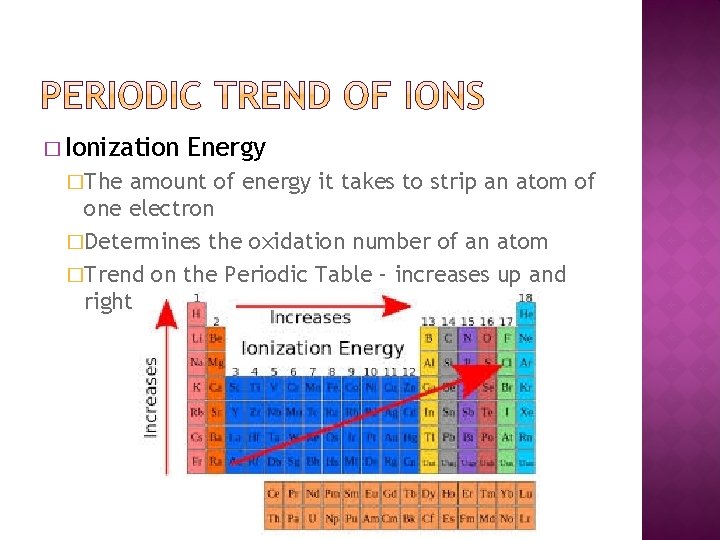
� Ionization �The Energy amount of energy it takes to strip an atom of one electron �Determines the oxidation number of an atom �Trend on the Periodic Table – increases up and right

� The lower the ionization energy, the more reactive an element is � https: //www. youtube. com/watch? v=Hv. VUtp d. K 7 xw&t=15 s � High ionization energies do not always mean highly reactive

� Compounds that contain ionic bonds � Form ionic crystals as solids � Ionic compounds shatter when struck � Poor conductors as solids, good conductors in solutions

� Binary ionic compound - compound with only two elements Name the cation 2. Name the anion 1. 2. � Replace the ending with “-ide” Example: oxygen -> oxide, chlorine -> chloride Examples: Na. Cl � Mg. Br 2 �

� Metals with multiple ions �Many transition metals have more than one ion, which is why we couldn’t say they have a specific number of valence electrons �These metals have multiple names �Examples: Copper (I) and Copper (II) Iron (II) and Iron (III) Lead (II) and Lead (IV) �The number after the element indicates how many electrons it is able to lose

� Polyatomic ions – multiple elements bonded together that have a net charge � Naming the works the same as binary compounds � Common Polyatomic ions:
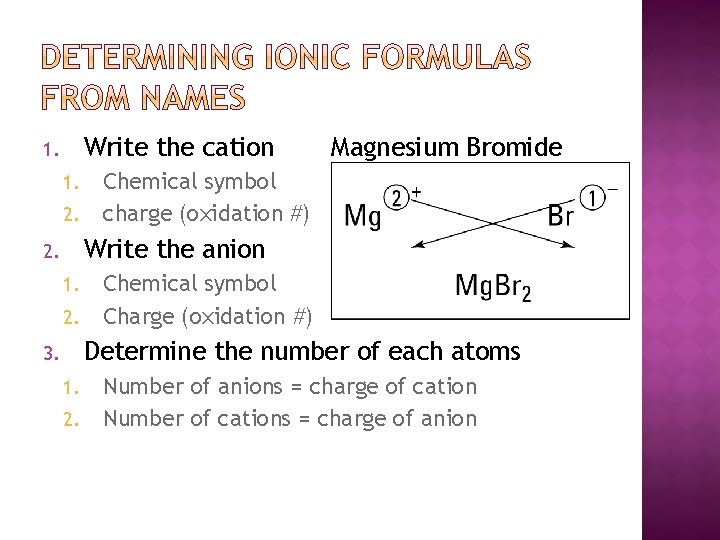
Write the cation 1. 1. 2. Magnesium Bromide Chemical symbol charge (oxidation #) Write the anion 2. 1. 2. Chemical symbol Charge (oxidation #) Determine the number of each atoms 3. 1. 2. Number of anions = charge of cation Number of cations = charge of anion

� Correction �Naming from yesterday: ionic compounds: Greek prefixes are not used � Worksheet �#3 -5 from yesterday
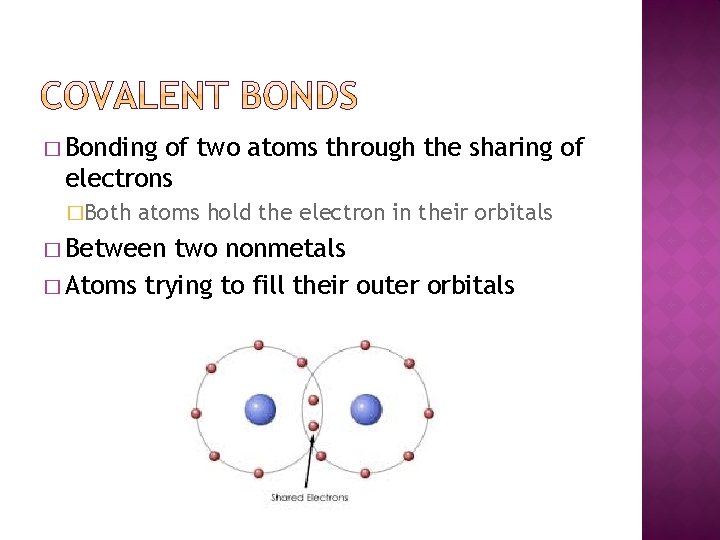
� Bonding of two atoms through the sharing of electrons �Both atoms hold the electron in their orbitals � Between two nonmetals � Atoms trying to fill their outer orbitals

� Molecule – neutral group of atoms that are help together by covalent bonds � Types of covalent bonds forming molecules �Single bond – between 2 electrons, one from each atom �Double bond – between 4 electrons, two from each atom �Triple bond – between 6 electrons, three from each atom
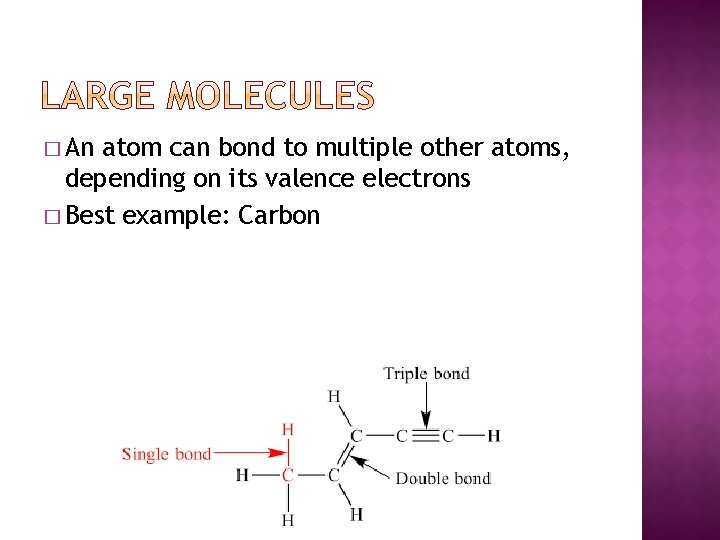
� An atom can bond to multiple other atoms, depending on its valence electrons � Best example: Carbon

� Polar covalent bond – a covalent bond in which electrons are not shared equally �Electrons are pulled harder by some atoms than by others � Electronegativity – how strongly an atom attracts electrons

� Polar bonds form polar molecules �Molecules that have a negative charge on one end a positive charge on the other end � Polar molecules attract each other
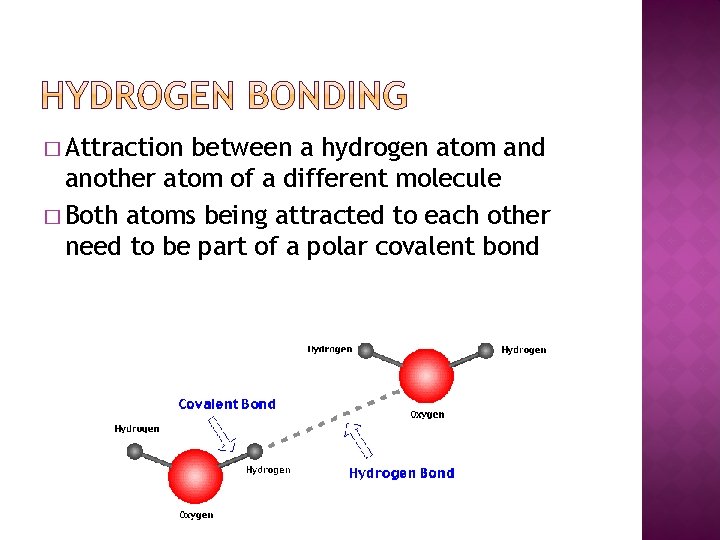
� Attraction between a hydrogen atom and another atom of a different molecule � Both atoms being attracted to each other need to be part of a polar covalent bond

� Atoms are listed in order of increasing electronegativity � The last element in the molecule ends with “ -ide” � Greek prefixes indicate how many of each atom are in the molecule

� Summarize notes about ionic and covalent bonds � Write two questions � 1 level 1 question � 1 level 2 question
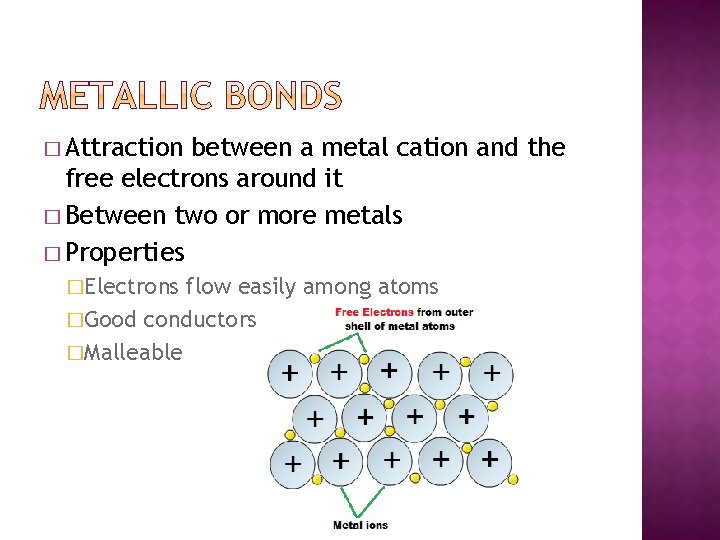
� Attraction between a metal cation and the free electrons around it � Between two or more metals � Properties �Electrons flow easily among atoms �Good conductors �Malleable

� Two or more elements that form metallic bonds � Alloys have properties that are different from all elements in them

� Bronze made from Copper and Tin �Copper and Tin are both very soft �Bronze is much stronger and harder �Used in ancient swords and shields � Brass made from Copper and Zinc �Softer and more malleable than brass �Used in decorations and instruments

� Steel made from Iron and Carbon �Stronger that iron because carbon holds the iron together �Many different alloys of steel that contain different elements, such as chromium or manganese � Alloys are made to have specific properties for specific functions
- Slides: 29